Research Article
Genotoxicity Assessment of Fresh Khat Leaves Extract in Chinese Hamster Ovary Cell Lines
Department of Laboratory Medicine, Faculty of Applied Medical Sciences, Albaha University, Al-Baha, Kingdom of Saudi Arabia
LiveDNA: 969.454
Chewing khat habit represents a major socio-economic problem in the Southern Arabia and many countries of the Horn of Africa. The leaves of Catha edulis (Celastraceae) (khat) are chewed for its sympathomimetic and stimulating effects. Khat leaves are chewed due to their pleasurable and stimulating effects. It is chewed mainly in countries of Eastern Africa regions and Southern Arabia1-4. The use of khat has spread worldwide, however, it has until recently remained mostly confined to the regions where the plant is planted since only fresh leaves have the potency (of cathinone) to produce the desired pleasurable and stimulating effects. Due to its structural similarity with amphetamine, cathinone is the most important active ingredient of khat, which causes the major pharmacological effects5. Other constituents of khat leave include tannins, amino acids and a significant amount of ascorbic acid, magnesium and beta-carotene1,6. The market value of the leaves correlates with their cathinone contents1. The detrimental and common adverse effects of the active principle of khat on man and animals include analgesia, insomnia, anorexia, irritability, hyperthermia, mydriasis and endocrinological disturbances have been described7-9. Acute autonomic responses, such as elevated blood pressure and tachycardia, have been reported10,11. The chronic use of khat has also been associated with the development of hypertension12.
The major compounds are absorbed in the oral cavity due to the efficient alkaloid extraction by chewing13. Increase in micronucleated buccal cells was observed in khat chewers14. Leaves extract administration was found to inhibit RNA and DNA synthesis in the neurons of chick embryo15 and reduce DNA and RNA contents in liver and brain tissue homogenates in rat16. Meanwhile, cytotoxicity and mutagenicity to mammalian cells have been reported after treatment with methanolic khat leaves extract17. Numerical chromosomal aberrations have been reported to be induced by khat extract in somatic cells of mice18 while structural chromosomal aberrations in rat blood samples were observed by Al-Zubairi et al.19. The cytotoxic effects of whole khat extract on three leukemia cell lines (HL-60, Jurkat and NB4 cells) were reported20 and apoptosis induction through a mechanism involving activation of caspase-1, caspase-3 and caspase-821. Cytotoxic and genotoxic effects on human cell lines CEM has also been reported22.
This study reported the results obtained on the genotoxic effects of fresh khat leaves extract in Chinese hamster ovary (CHO) cell lines, using chromosomal aberrations assay (CAs) and micronucleus (MN) formation as cytogenetic endpoints. This study described the results of those investigations and comments on the in vitro genotoxic effects of fresh khat leaves extract in CHO cell line.
Khat extract: Fresh khat leaves have been obtained from khat sellers in Kuala lumpur. Khat leaves have been washed thoroughly, blended with juicer and the leaves juice has been filtered and used immediately for cell cultures treatment. Khat leaves extract doses were expressed according to starting fresh plant material weight.
Chromosomal aberration assay: In genotoxicity testing, the potential of a chemical to induce chromosomal damage is usually investigated. Therefore, the fresh khat leaves extract was evaluated for its potential to induce chromosome aberrations and micronuclei formation in CHO cell lines.
CHO cell lines: Chinese hamster ovary (CHO) cell lines were purchased from ECACC (UK). Atypical diploid CHO cell contains 21 chromosomes. The cells grow as an adherent monolayer in the tissue culture flask, doubling approximately every 12 h. Cultures were monitored for their health status throughout the experiment by cell counting and microscopic observation. Culture medium used to maintain the cell lines was RPMI 1640 medium (PAA Laboratories GmbH, Germany) supplemented with 10% foetal bovine serum (PAA Laboratories GmbH, Germany) using sterile Nunc tissue culture asks (Nunc, Denmark). The cells were incubated in a humidified tissue culture incubator at 37°C and 5% CO2. Cells were trypsinized with 0.05% trypsin (PAA Laboratories GmbH, Germany) when reach approximately 60-80% confluency, collected by centrifugation and seeded in fresh medium. Each trypsinization step was recorded as the cells have one passage. A continued culture of CHO cell line (passage15) was used. On the day before the experiment, 5×105 cells from a culture with approximately 60-80% confluence were seeded into each T-25 cm2 (Nunc, Denmark) flasks. The cells were incubated overnight in a humidified incubator at 37°C and 5% CO2.
Treatment with fresh khat leaves extract: Fresh khat leaves extract was prepared as 1 mg mL–1 stock solution in absolute ethanol just prior to use and a dosing volume of stock concentration diluted with medium ensured a final ethanol concentration of <1%. Overnight cell cultures were examined under an inverted microscope and appeared healthy, well adherent, evenly distributed and 60-80% confluent. For each extract concentration and controls, duplicate cultures were prepared. Control cultures were handled in the same manner to the treated cultures. Mitomycin-C (Sigma, Germany) was used as a positive control. The treatment medium was 5 mL of the cell culture medium with 10% foetal bovine serum, with the treatment concentration or a control solution and the final concentrations were 0, 5, 50, 500 and 5000 μg mL–1. Cells were cultured in the treatment medium for 24 h.
Cells Harvest and Preparation of chromosome slides: Cells for chromosomal aberration assay were harvested at the end of treatment. Colcemid (PAA Laboratories GmbH, Germany) was added 2 h prior to harvesting, at 0.2 mL to arrest cells in metaphase. Then the medium with suspended cells was transferred into centrifuge tubes. The attached cell monolayer was dissociated with 0.05% trypsin and pooled with the collected cell suspension. After that, the cells were centrifuged at 800 rpm and resuspended in phosphate buffered saline (Sigma), collected, swelled in 0.075 M KCl (Sigma) and fixed in Carny’s fixative (3:1 mixture of methanol and glacial acetic acid) for 3 times. Next, the tubes were centrifuged for 5 min, the supernatant discarded and the cell suspension dropped onto pre-chilled slides previously cleaned with non-toxic detergent and soaked in distilled water at 5°C. Air dried chromosome slides were prepared and stained in 6% Giemsa stain ([CAS 67-56-1] Gibco, Invitrogen, Germany). One thousand cells were examined for each duplicate culture to score mitotic index. The mitotic index (MI) was calculated as the percent of cells at the mitotic stage. Relative mitotic index (RMI) was calculated as Baak23:
![]()
Analysis of chromosome aberrations: All slides were randomly coded and well-spread metaphase cells with 19-23 chromosomes were analyzed for chromosome aberrations as defined. Two hundred metaphase cells from each negative control, positive control and fresh khat leaves extract-treated cultures were scored. The number of each type of aberration and the percent of cells with aberrations were recorded and summarized. The number of chromatid gaps and chromosome gaps were recorded when encountered, but not included in the calculations24. The percentages of cells with aberrations from each concentration were compared to the solvent control values using χ2 analyses.
Micronucleus test (MN)
Cell culture treatment: The CHO cell cultures were prepared as previously described in the chromosomal aberrations assay. After 22-24 h the media was removed and the cells were washed with PBS or media and fresh media containing 6 μg mL–1 Cytochalasin-B (CB [CAS 4930-96-2], Sigma-Aldrich, Germany) was added and incubated for further 22-24 h. One set of negative and positive controls were run for each experimental day.
Harvest and slide preparation: To examine binucleated cells for micronuclei in vitro, the technique utilized was that described by Fenech25. After completing treatment, the cells were trypsinized and suspended in pre-warmed hypotonic solution (0.075 M KCL) and carefully homogenized with a Pasteur pipette. Then the cell suspension was centrifuged again and re-suspended in pre-chilled 10 mL of Carney’s fixative, methanol/acetic acid (3:1 v/v), centrifuged and dropped onto pre-chilled slides. The slides were stained with 6% Giemsa diluted in phosphate buffer (pH 6.8) for 10 min, washed with distilled water, air-dried and examined under the microscope.
Scoring: The MN induction was determined in 1000 binucleated cells with the cytoplasm well preserved and clearly surrounded with nuclear membrane, having an area of less than one third of that of the main nucleus26. Using a Nikon microscope, cells containing 1 micronucleus were scored blindly. For identification of MN, the criterion used was according to Fenech27. In each treatment, for cell cycle kinetic analysis, numbers of mononucleated, binucleated and polynucleated cells per 1000 cells were counted, the nuclear division index or cytochalasin B proliferation index (CBPI) is determined in a blind test. Cells with well preserved cytoplasm, containing 1-4 nuclei, were scored. The CBPI was calculated according to OECD guideline number 48728 using the following formula:
![]()
A minimum of 200 cells per concentration were analysed (N).
Statistical analysis: Data were analyzed for Chi-square analysis using SPSS version 24.0. All statistical tests were performed at the p<0.05 level of significance.
Mitotic index analysis: A two-fold series of 5 concentrations of fresh khat leaves extract were used. The relative mitotic index (RMI) values (Table 1) revealed an increasing inhibitory effect with increasing concentrations of the khat extract as well as for the positive control MMC. The mitotic index of the two higher concentrations was found to be significantly (p<0.001, Table 1). In contrast, the mitotic index of the two lowest concentrations was observed to be non-significantly reduced compared to the control.
Chromosome aberrations (CA) assay: The CA assay, clastogenicity determined was not significantly observed in the untreated control or the solvent treated control.
| Table 1: | Mitotic index and relative mitotic index (RMI) observed for CHO cultures treated with different concentrations of Khat extract and controls |
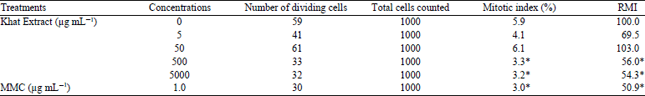 | |
| *Significantly different from the control p<0.05 | |
| Table 2: | Sum of the frequencies of chromosome aberrations (CAs) induced in CHO cultures treated with different concentrations of khat extract and the positive and negative controls |
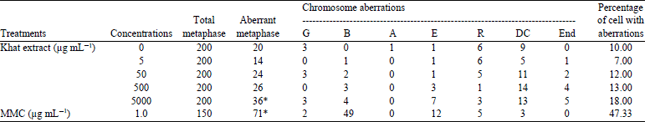 | |
| G: Gap, B: Breaks, A: Acentrics, E: Chromatide exchange, R: Chromosome ring, DC: Dicentric, END: Endoreduplication. *Significantly different from the control p<0.05 | |
| Table 3: | Frequencies of micronucleus (MN) formation and cell cycle kinetics on CHO cultures treated with different concentrations of Khat extract together with MMC treated and untreated cultures |
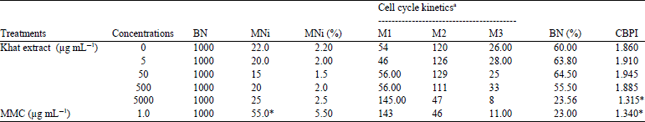 | |
| aThe numbers of mononucleated (M1), binucleated (M2) and polynucleated (M3) cell per 200 cells were quantitated for cell cycle kinetic analysis. *Significantly different from control p<0.05 | |
Positive control (MMC) was significantly shown to induce chromosome aberrations. On the other hand, the clastogenic indices of the highest concentration of fresh khat leaves extract was found to induce chromosome aberrations significantly (p<0.01) as shown in Table 2. Gaps, breaks as well as exchanges, endoreduplications, ring chromosomes and dicentrics were the main types of aberration observed. In treated cultures of CHO cell line the number of breaks was found to be significantly increased in the highest concentration of the khat leaves extract. However, ring chromosomes and dicentrics were found to be non-significantly increased in all treated cultures. Meanwhile, endoreduplications were found to be high in the two highest concentrations. The number of aberrant cells with structural aberrations including or excluding gaps recorded in cultures treated with the highest concentrations (5000 μg mL–1) increased significantly p<0.01 (Table 2) when compared with the untreated groups. Fisher’s exact test was used to test the statistical significance of the CA results.
MN induction: The potential of fresh khat leaves extract to induce micronucleus is shown in Table 3. Treatment of CHO cells with khat leaves extract was to have no effects in the MN induction test. In the cell cycle kinetics analysis or CBPI, treatment with the highest khat extract concentrations (5000 μg mL–1) was found to exert an inhibition of cell proliferation in CHO cells (Table 3), while other concentrations were found to be reduced non-significantly compared to the negative control treated cells.
People of Southern Arabia and East Africa habitually chew khat leaves, which produce amphetamine-like stimulating effects. Habitual khat chewing results in social and public health problems among these populations. It has been reported that khat leaves contain 14.5% tannins, mainly in the form of condensed type tannins29 and other polyphenolic and flavonoid compounds. The mutagenicity of C. edulis leaves extract has been studied previously in bacteria by Hannan et al.30 and rats by Al-Zubairi et al.19. As well as by De Hondt et al.16, who studied the acute effect of khat administration and observed the chromosomal aberrations formation after subcutaneous treatment of rats with methanolic khat extract. In addition, Tariq et al.31 studied the prolonged exposure of mice to khat extract and reported khat extract to induce total and partial sterility in male and post-implantation loss in female and reduced the percent pregnancy rates. Khat extract was also reported to increase the frequency of micronucleated polychromatic erythrocytes, induced bone marrow depression and reduce the mitotic index of the somatic cells18, as well as the sister chromatoid exchange and various chromosomal aberrations in mice bone marrow cells32.
It was found that drying of khat leaves results in loss of some active components like cathinone, therefore, people chewing khat usually prefer to use the green fresh leaves1. In this study it was tried to evaluate for the first time, the genotoxic effects of fresh khat leaves extract in CHO cell lines. This investigation was conducted using the chromosomal aberration assay and micronucleus test as cytogenetic end-points. Fresh khat leaves extract reduced the mitotic index at high extract concentrations and induced higher chromosomal aberrations frequencies as compared to control values as revealed by the CA assay. The percentage of structurally damaged cells in the MMC (positive control) treatment group was statistically significantly increased compared to the solvent control indicating the responsiveness of the cells in this test system. Genotoxic agents have the potential to interact with DNA and may cause DNA damage. Chromosomal aberrations occur in proliferating cells and are regarded as a manifestation of damage to the genome. Chromosomal aberrations assay has been commonly used as a test of genotoxicity in order to evaluate cytogenetic responses to chemical exposure. The results found, at least under the experimental conditions used in the present work, showed the ability of this extract at high concentration, to induce genotoxicity and cytotoxicity in vitro in CHO cells. These results were found to be in agreement with that reported by Barkwan et al.22, who studied the cytotoxic and genotoxic effects of khat extract on human cell lines CEM.
Formation of dicentric and ring chromosomes may suggest telomeric loss or telomeric associations as well as chromatid/chromosome breaks that starts chromosomal breakage-fusion-bridge cycle and finally results in the production of mitotically unstable chromosome aberration, i.e., ring chromosome, dicentric chromosome or telomeric association in metaphase cells33,34. Endoreduplication in eukaryotic cell is a process that involves DNA amplification without cell division35,36. Inhibition of DNA topoisomerase II activity results in the formation of endoreduplication35,37,38 and increase in the number of endoreduplicated cell chromosomes may indicate that the test substance is capable of inhibiting cell cycle progression.
In addition to chromosomal aberration assay, MN induction test is another widely used cytogenetic method to assess in vitro clastogenic potential of substances. The frequency of occurrence of micronuclei in treated cells provides a comparatively rapid and sensitive indication of both chromosomal aberrations and loss that lead to numerical chromosomal anomalies39,40. Micronuclei are chromatin masses formed in the cytoplasm with the appearance of small nuclei result from chromosome fragments at anaphase or from acentric chromosomal fragments. Induction of micronuclei provides a quantifiable measure of recent DNA damage result from acentric fragments or whole chromosomes fragment as a result of an exposure to either clastogenic or an aneuploidogenic effect41,42. The present study failed to show significant effects of exposure of CHO cells to fresh khat leaves extract. However, there were no significant changes in the induction of micronuclei formation between the treated cultures and the negative control cultures. Meanwhile the cell cycle kinetics represented by the CBPI showed that the highest concentration (5000 μg mL–1) of khat leaves extract reduced the CBPI significantly compared to the negative control. This later result corresponds with that of the chromosomal aberration assay, where the highest concentrations of the fresh khat leaves extract induced chromosomal aberrations significantly.
The present results provide evidence that fresh khat leaves extract in high concentrations have a genotoxic effects on cultured Chinese hamster cell line, bringing into highlight the need for further studies to better understand the molecular mechanisms of action of khat leaves extract for a better comprehension. Thus results found, at least under the experimental conditions used in the present work, showed the ability of fresh khat leaves extract to induce genotoxicity in vitro in CHO cells.
This study highlights for the first time, the effect of unprocessed fresh khat leaves extract on the chromosomes of the normal cell lines, CHO cell lines, using the two cytogenetic end points chromosomal aberrations assay and micronucleus test. The results of this study may shed a light on the genotoxicity of the fresh extract of the khat leaves.
The author would like to extent his utmost gratitude and appreciation to Laboratory of Cancer Research-MAKNA, Institute of Biosciences, University of Putra Malaysia for providing the research facilities as well as Dr Ahmad Bustamam, for his help in completing this study.