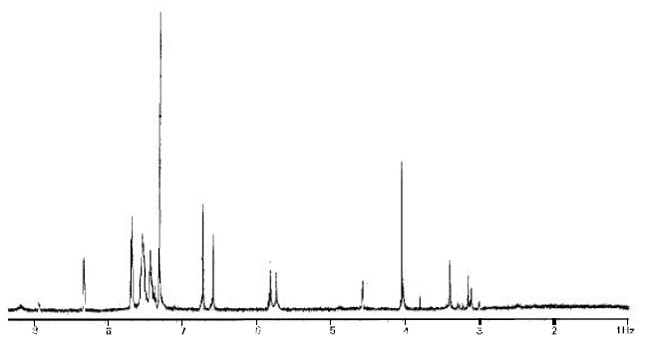
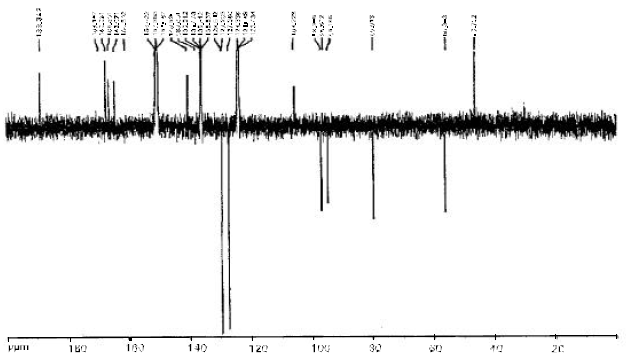
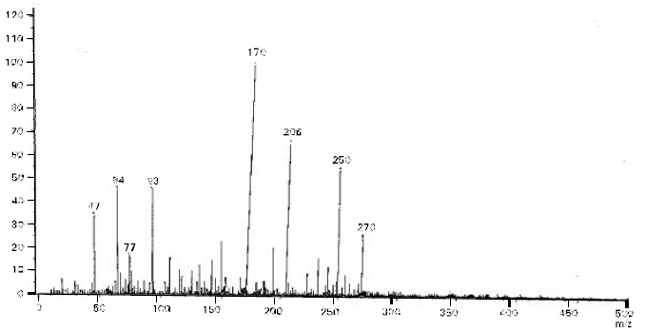
Research Article
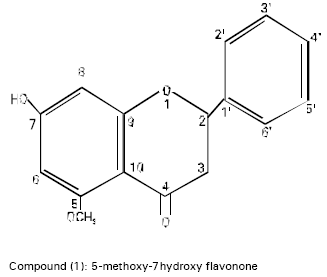
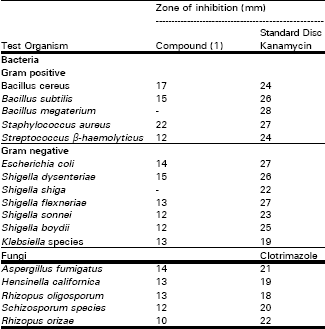
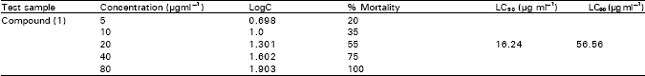
A Flavonone from Leaves of Zanthoxylum budrunga: its In vitro Antimicrobial Activity and Cytotoxic Evaluation
Department of Applied Chemistry and Chemical Technology, University of Rajshahi, Bangladesh, Open School, Bangladesh
Abu Sayeed
Department of Applied Chemistry and Chemical Technology, University of Rajshahi, Bangladesh, Open School, Bangladesh
Md. Anwar -Ul Islam
Open University, Gazipur, Bangladesh
G. R. M. Astaq Mohal Khan
BCSlR, Rajshahi, Bangladesh
M. Helal U. Biswas
Department of Pharmacy, University of Rajshahi, Rajshahi 6205, Bangladesh
M. Shah Alam Bhuiyan
Department of Pharmacy, University of Rajshahi, Rajshahi 6205, Bangladesh