Research Article
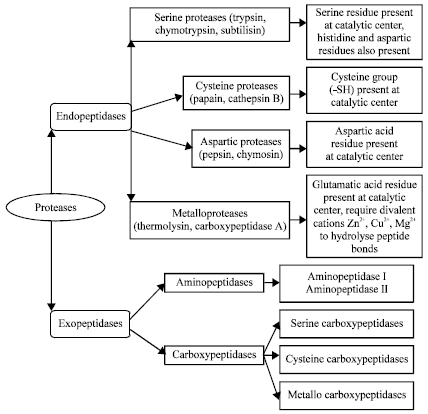
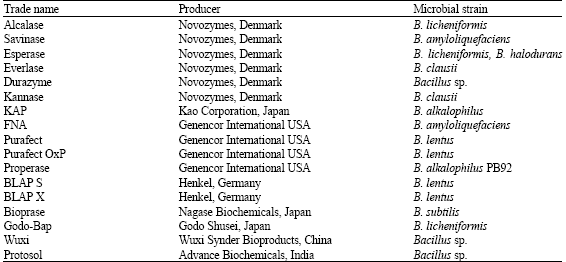
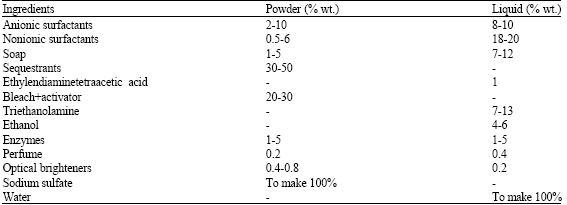
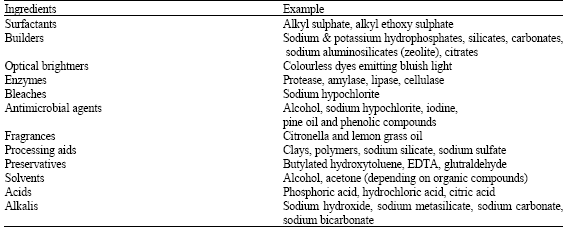
Microbial Proteases and Application as Laundry Detergent Additive
Department of Biotechnology, Abhilashi Institute of Life-Sciences, Tanda, Ner Chowk, Mandi-175008, H.P., India
Savitri
Department of Biotechnology, Abhilashi Institute of Life-Sciences, Tanda, Ner Chowk, Mandi-175008, H.P., India
N. Thakur
Department of Biotechnology, Abhilashi Institute of Life-Sciences, Tanda, Ner Chowk, Mandi-175008, H.P., India
R. Verma
Department of Biotechnology, Abhilashi Institute of Life-Sciences, Tanda, Ner Chowk, Mandi-175008, H.P., India
T. C. Bhalla
Department of Biotechnology, Himachal Pradesh University, Shimla-171005, H.P., India