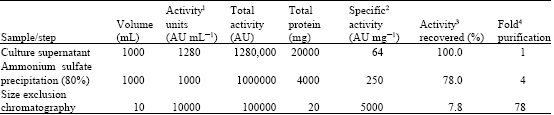
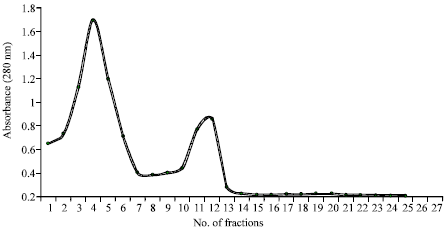
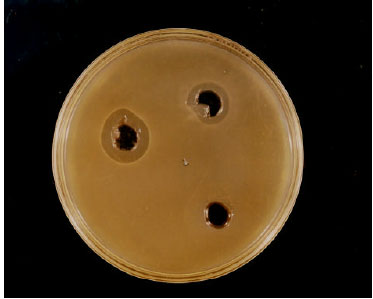
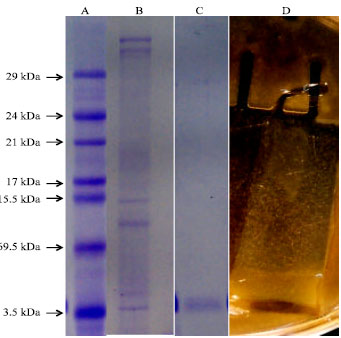
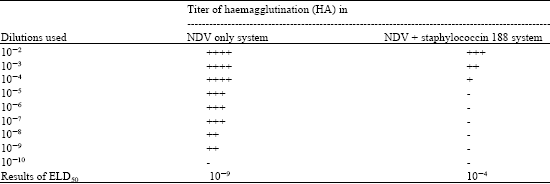
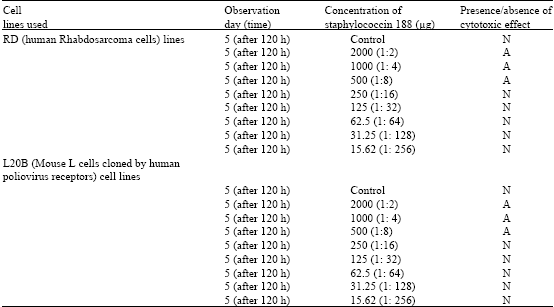
Research Article
Antiviral Activity of Staphylococcin 188: A Purified Bacteriocin Like Inhibitory Substance Isolated from Staphylococcus aureus AB188
Laboratory of Molecular Genetics, Department of Microbiology, University ofKarachi-75270, Pakistan
Sheikh Ajaz Rasool
Laboratory of Molecular Genetics, Department of Microbiology, University ofKarachi-75270, Pakistan
Samia Ahmad
Laboratory of Molecular Genetics, Department of Microbiology, University ofKarachi-75270, Pakistan
Sohail Zahoor Zaidi
Public Health Laboratories, National Institute of Health, Islamabad, Pakistan
Shafqat Rehmani
Sindh Poultry Research Institute, Karachi, Pakistan