Research Article
Evaluation of Microorganisms from Cassava Waste Water for Production of Amylase and Cellulase
Department of Microbiology, Federal University of Technology, P.M.B. 704, Akure, Nigeria
Cassava (Manihot esculanta Crantz) is a root tuber crop that is widely cultivated in the tropical regions of the world (Iyayi and Losel, 2001; Oboh and Akindahunsi, 2003a). As a shrubby perennial that grow to a height of 6-8 ft, it is usually propagated by planting short section of the stem (O’Hair, 1995; Oboh, 2005). Suffix to say that different cultivars of cassava which abound worldwide mature at different rates. However, certain varieties contained large amount of cyanogenic glycosides (linamarin and lotaustralin) which can hydrolysed to hydrocyanic acid (HCN) by their endogenous enzyme (linamarase) when the plant tissue is damaged during harvesting, processing or other mechanical processing (Oboh and Akindahunsi, 2003b). The protein content of cassava products can be increased by adding protein to the deficient food in a way that will not alter the organoleptic qualities of the original food (Oboh, 2005). Also, through controlled fermentation, microflora could be made into large numbers in the mash (Raimbault, 1998; Oboh et al., 2002; Oboh and Akindahunsi, 2003a), thus increasing the protein content of cassava products.
Oboh (2005) identified two important wastes that are generated during the processing of cassava tubers to include cassava peels and the liquid squeezed out of the mash. The bioconversion of the cassava wastes have been documented (Antia and Mbongo, 1994; Okafor, 1998; Raimbault, 1998; Tweyongyere and Katongole, 2002; Oboh, 2005). The (waste water) contains heavy loads of microorganisms, lactic acid, lysine (from L. coryneformis), amylase (from L. delbruckii) capable of hydrolyzing the glycosides (Raimbault, 1998; Akindahunsi et al., 1999). The industrial applications of amylases as additives in detergents for the removal of starch from textiles, liquefaction of starch and proper formation of dextrin in baking have been reported (Shaw et al., 1995). In addition, amylases are used in high fructose corn syrup preparation, saccharification of starch for alcohol production and in brewing (Uzochukwu et al., 2001; Aiyer, 2004). Cellulase is added to crush apples to increase juice yield and prevent contamination (Bhat, 2003). Cellulases are utilized in the textile industry for colour brightness (Csizer et al., 2001) and for stone wash look in jeans (Haki and Rakshik, 2003), paper processing, production of ethanol for fuel from the non-edible portion of corn and wheat (Iogen Corporation, 2003). Therefore, the industrial applications of these enzymes (amylases and cellulase) cannot be underscored.
Nigeria is the largest world largest producer of cassava and it constitutes the most economic sources of starch (Daramola and Osanyinlusi, 2006). During the processing of cassava tubers in various products, liquid waste waters generated was reported to cause serious havoc to vegetation, houses and bring about infection. This no doubt have being causing serious environmental pollution as a result of the indiscriminate discharge. The liquid squeezed out can be dried and used as animal feeds (Okafor, 1998; Oboh and Akindahunsi, 2003a).This work focus on the isolation, characterization and identification of microorganisms that may be present in cassava waste water for the production of amylase and cellulase which could be utilized in an industrial process.
MATERIALS AND METHODS
Source of Samples
Cassava waste water was obtained from five different cassava processing factories locations in Akure, Ondo State, Nigeria. The samples were collected in clean sterile bottles and transported to the microbiology laboratory of the Federal University of Technology, Akure,Nigeria in an ice bags for analysis. Stock samples were maintained at 4°C in the refrigerator.
Isolation and Identification of Associated Microorganisms
The associated microorganisms of the samples were isolated using serial dilution pour plate of Akerele (1990). In this case, 0.1 mL of dilutions x10-5 and x10-2 of the samples were inoculated on nutrient agar (NA) and potato dextrose agar (PDA) for bacteria and fungi, respectively. The inoculated media were incubated at 37°C for 24 h for bacteria and at 30°C for 3-5 days for fungi. Discrete colonies that developed on the plates were counted and recorded as colony forming unit per milliliter (cfu mL-1). The pure cultures of bacteria obtained by sub culturing were identified using the methods of Holt et al. (1994), while the fungal isolates were identified by Barnett and Hunter (1972).
Determination of pH
The pH value of each sample was determined using Extech Instrument pH 100 after standardization with appropriate buffers. The electrode sensor of the pH meter was inserted directly into 20 mL sample in a clean 50 mL glass beaker. The value on the recorder was read and recorded as the pH value.
Determination of Total Titratable Acidity (TTA)
This was determined according to the method of Akharayi and Omoya (2005). Twenty-five milliliters of the samples with 3 drops of 1% phenolphthalein as an indicator was titrated against 0.1 M NaOH. The end point was recorded when the pink colour was noted. The TTA was then calculated as lactic acid thus: volume of 0.1 MNaOHx factor x100 over volume of weight of sample used in titration (mL).
Screening Microbial Isolates for Amylase and Cellulase
The method described by Cowan and Steel (1990) was used for the screening of microbial isolates for the production of amylase. This involved the incorporation of the potato starch into nutrient agar. The mixture sterilized, cooled, inoculated with the isolates and incubated. The plates were then flooded with Lugol’s iodine solution for starch hydrolysis which indicated the production of amylase.
A modified method of Ogundero (1982) was employed for the production of cellulase. The basal medium-carboxymethylcellulose (CMC) consisting of Mg SO4.7 H2O, 0.75 g; NaNO3, 2.5 g; KH2PO4, 1.75 g; CaCl2.H2O, 2 g; CMC, 10 g as carbon source, agar-agar and 1000 mL distilled water. The mixture warmed, autoclaved and poured into already sterilized Petri dishes. The bacterial isolates were streaked on the plates according to the standard method and incubated at 37°C for 48 h, while the fungal isolates were inoculated on the plates by the method of Arotupin and Akinyosoye (2001) and incubated at 30°C for 3-5 days. Formation halo of zone around the colony after flooding with 0.1% congo red solution and washed with 0.1 M NaCl confirmed the production of cellulase.
Table 1 showed the total microbial counts from each of the factories. Factory 5 recorded the highest number of microbial population, while factory 2 recorded the least microbial counts. The high microbial counts may be due to lack of efficient control measures in the discharge of the waste water into the environment. Uzochukwu et al. (2001) reported that high level of cassava waste water are produced daily and drained onto roads, streets, rivers and agricultural lands in gari producing communities of Nigeria. These singular activities tend to expose the waste water to microbial contamination. The reports of Uzochukwu et al. (2001) also revealed cassava waste water to contain fermentable sugars, starch, cellulose, while Oboh and Akindahunsi (2003a) documented it to contain cyanogenic glycosides and essential elements such as Zn, Mg, Fe, Ca, Na and K. This nutrients and chemical composition of the waste water posited its ease of colonization, hence may account for the high microbial counts. The low microbial counts in factory 2 may be due to the regular sanitation, disinfection and fumigation of the environment as confirmed by the factory manager.
Microbial isolates of Cassava Waste Water (CWW) consisted of five bacteria, five moulds and two yeasts. The bacterial isolates were Aerococcus viridens, Bacillus substilis, Bacillus sp. Corynebacterium manihot and Lactibacillus acidophilus, while the fungal (mould) isolates included Aspergillus fumigatus, A. niger, A. repens, Articulospora inflata and Geotrichum candidum. The yeast isolates were Candida utilis and Saccharomyces exguus (Table 2). The results clearly showed that both bacteria and fungi including yeasts are associated with the cassava waste water. However, most of these microbial isolates have been implicated during the processing of cassava tubers into various products (Okafor, 1998; Olowoyo et al., 2001; Akinyosoye et al., 2003).
| Table 1: | Total microbial counts of cassava waste water from different factories |
 | |
| F = Factory; cfu mL-1 = Colony forming unit per milliliter, sfu mL-1 = Spore forming unit per milliliter, ND = Not Detectable | |
| Table 2: | Frequency of occurrence of the microbial isolates |
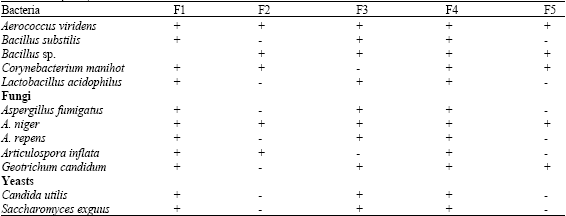 | |
| + = Present, - = Absent | |
| Table 3: | pH values and titratable acidity (TTA) of cassava waste water |
 | |
Okafor (1998) and Oboh and Akindahunsi (2003b) separately reported the bioconversion of agricultural wastes of these microbial isolates of which CWW is not an exception. The growth and survival of these microbial isolates may not be unconnected with the fact that the waste water contained substances that can be utilized by the isolates. Uzochukwu et al. (2001) and Oboh (2005) had earlier reported the composition cassava waste water. These microbial isolates may probably have originated from soil, water and materials used during the processing of cassava, while the variations of the isolates may be due to the handing process and the prevailing environmental conditions. Therefore, the isolates can be said to be transient microorganisms surviving only in the absence or low cyanide and/or other inhibitory substances in cassava (Olowoyo et al., 2001).
The pH values and Titra Table Acidity (TTA) are shown in Table 3. The pH value ranged from 3.55 to 4.02, while the TTA ranged from 9.50 to 13.01%. The observed pH values agreed with the findings of Uzochukwu et al. (2001). They reported pH range of 4.00 to 4.20 of cassava waste water. The acidic nature of the cassava waste water probably favors the growth of the microbial isolates, as these encountered microbial isolates have been reported to be an acid tolerant (Abba-Kareen and Okagbue, 1999; Agarry, 2005; Oboh, 2005). However, the TTA observed were higher than that reported by Uzochukwu et al. (2001). The difference may be as a result of the amount of other organic acids in the waste water, which was probably more in these samples.
All sthe microbial isolates hydrolysed soluble starch with the exception of A. fumigatus and A. repens, while Bacillus substilis and Saccharomyces exguus were unable to hydrolyse carboxymethylcellulose (CMC) (Table 4). The ability of the microbial isolates to hydrolyse soluble starch and CMC suggest the elaboration of both amylase and cellulase which are the enzymes required for the breakdown of these carbon substrates. Those unable to hydrolysed the substrates probably depended on the breakdown products for survival. The presence of sugar and starch (Uzochukwu et al., 2001) and possibly cellulose tend to make the waste water suitable substrates hence, stimulate the production of amylase and cellulase for microbial growth and survival. In addition, species of Bacillus, Lactobacillus, Aspergillus, Candida and Saccharomyces have been reported to be copious producers of amylases and cellulases (Hreggvidsson et al., 1996; Haki and Rakshit, 2003; Kotchoni et al., 2003; Oboh, 2005).
| Table 4: | Screening of microbial isolates for amylase and cellulase production |
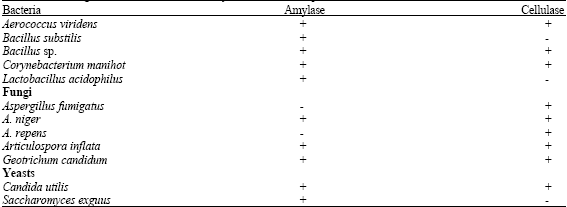 | |
| + = Positive, - = Negative | |
The low pH observed in this study and earlier documented by Oboh (2005) stimulated the production of amylase. He further asserted that amylases are active at low pH, hence can be useful in processes requiring low pH ranges. The squeezed out liquid (waste water) from the fermented cassava pulp have been assessed as an important source of microbial enzymes (Okafor, 1998). Also, Oboh (2005) revealed the presence of amylase in the cassava waste water. The enzymes may have been released into the cassava waste water through the activities of microorganisms associated with the processing methods.
In this study, cassava waste water contained array of microorganisms with potential for the production of amylase and cellulase. These enzymes have direct applications in industrial processes. Therefore, cassava waste water could be converted into economic value through the development of technology for the safe collection of the waste water. This would go a long way in creating safe and friendly environment in communities where cassava tubers are processed into products.
Agwupuye,Christopher Agim Reply
Well written. It worth a scientific article.