Research Article
Effect of Different Carbon Sources on the Growth and Polygalacturonase Activity of Aspergillus flavus Isolated from Cropped Soils
Department of Microbiology, Federal University of Technology, P.M.B. 704, Akure, Nigeria
Pectinases are enzymes that are capable of degrading pectin materials. These pectinases produced by fungi are mainly made up of those enzymes, which act on pectinic acids and those which act on pectic acid (pectinlyase and polygalacturonase respectively) (Eludire et al., 2003). The action of these enzymes could be random cleaving or at the extremities (White and Kennedy, 1998). The hydrolytic action of these group of enzymes have been used for their classification which include polygalacturonase, pectin esterase, pectin lyase and pectate lyase (Singh et al., 1999).
Microorganisms producing pectinases have been widely identified to be mostly pathogenic fungi (Cleveland and Cotty, 1991; Greens and Clausen, 1999). The major characteristics of most of the microbes attacking plant tissues is the elaboration of cell wall degrading enzymes which are non-specific with respect to their carbohydrate requirements for growth (Akinyosoye and Oboh, 2001, 2004).The cell walls of plant contains many different kinds of polysaccharides like cellulose, various hemicellulose and pectin (Akinyosoye and Oboh, 2004). However, there are many non-pathogenic species of bacilli, clostridia and Aspergillus, Penicillium, Fusarium and Phoma which pectinases (Akinola and Onaolapo, 2003; Eludire et al., 2003; Akinyosoye and Oboh, 2004). Stochinkaya et al. (1990) documented the fact that the symbiosis between the bacteria and the legume was made possible by the pectinases, thus accounted for the presence of pectinolytic enzymes in nodule bacteria. Studies had revealed bacteria to produce the pectolytic enzyme as much as fungi, but only the fungal enzymes particularly those from the species of Aspergillus have been generally employed for commercial purposes (Ismail, 1996).
Pectic substances are so ubiquitous in the plant kingdom and their efficient utilization could enhance the economic competitiveness of bioconversion process intended to compete with conventional industrial processes (Kapoor et al., 2000, 2001). Therefore, the recent interest in the degradation of pectic substances cannot be undermined which is evident from the vast range of industrial applications (Kapoor and Kuhad, 2002). These applications ranged from degumming of bast fibre (Bruhlmann et al., 1994; Kapoor et al., 2001), treatment of alkaline pectic waste water (Tanabe et al., 1988), improvement of wood texture prior to preservation (Endress, 1996), enzymatic extraction of oil from oil seeds (Buenrostro and Lopez-Munguia, 1986) and extraction, clarification and depectinization of fruit juices (Alkorta et al., 1998; Akinola and Onaolapo, 2003).
However, isolated plant cell walls have been used as carbon sources for various cell wall degrading enzymes of which polygalacturonase use to be the first enzyme elaborated with cellulose being the last (Akinyosoye and Oboh, 2004). Polygalacturonase which is the enzyme of interest, however, is a pectic or pectolytic enzyme with wide applications. This study investigates the effect of different carbon sources on the growth and polygalacturonase activity of Aspergillus flavus isolated from cropped soils.
Source of Materials
Pure strains of Aspergillus flavus was isolated from the cropped soils of the Federal University of Technology Teaching and Research Farm, Akure, Nigeria. The strains was maintained on potato dextrose agar (PDA) oxoid at 4°C. The chemicals used were analytical grade, while distilled water was employed.
Determination of Growth
Fifty milliliter portion of the basal medium ( modified) contained peptone, 1 g; KH2PO4, 1.05 g; NaNO3, 4 g; MgSO4.7H2O, 0.1 g; NaHPO4, 2 g; carbohydrate source (raw and commercial), 20 g and distilled water 1000 mL, while the carbon free medium serves as control. The pH of the broth medium was usually adjusted to 6.30 with either 0.1M NaOH or 0.1M HCl as appropriate. The various carbon sources used were ripe and unripe banana peels, ripe and unripe plantain peels, orange bagasse, cassava and potato peels, soluble starch and sucrose. The sterilized media at 121°C for 15 min in an autoclave were inoculated with 1 mL of the spores suspension of Aspergillus flavus according to Akinyosoye and Akinyanju, (1989). The cultures were incubated at 30°C on a rotatory shaker at 1000 rpm 72 h, subsequently harvested and analysed for polygalacturonase activity, while the growth was determined using the Narasimha et al. (2006).
Assay for Polygalacturonase Activity
The culture filterates served as crude enzyme and the polygalacturonase activity of the crude extract was measured using the method of Miller (1959). The specific activity of the enzyme was subsequently determined by using the protein content of the culture filterate employing the method of Lowry et al. (1951), while the pH values of the culture filterates were determined using Exstik pH meter by the method of Arotupin and Akinyosoye (2001).
Proximate Analysis of the Raw Carbon Sources
The proximate analysis of each of the raw carbon sample was carried out according to AOAC (1990) procedures for ash, moisture, crude fibre, fat and protein content using nitrogen to protein conversion factor of 6.25. Carbohydrate was determined by difference.
Mineral Analysis
The raw carbon substrates were sun dried until they were crispy to touch. The dried samples were grounded into fine powder, sieved with 0.2 mm in diameter sieve and kept in desiccators until required for use. The mineral elements namely Na, K, Ca, Mg, Mn and Fe were determined for each of the sample from their solution obtained by dry ashing according to AOAC (1990) using atomic absorption spectrophotometer (AAS).
The present study shows that the carbon sources supported the growth of Aspergillus flavus and the production of polygalacturonase, although to varying degrees. The growth of the Aspergillus flavus was highest in ripe banana peel with 1023 mg/50 mL, followed by orange bagasse with 978 mg/50 mL, then unripe plantain peel with 932 mg/50 mL, while the least growth was recorded in basal medium without carbon source with 36 mg/50 mL. Potato peel recorded the growth of 896 mg/50 mL, unripe banana, ripe plantain and cassava peels recorded 142, 119 and 101 mg/50 mL, respectively. Also, commercial soluble starch and sucrose recorded the growth of 112 and 67 mg/50 mL, respectively (Table 1). However, the raw carbon substrates (polysaccharides) supported the growth of the fungus better than the commercial soluble starch and disaccharide sucrose.
Aspergillus flavus elaborates polygalacturonase during the period of growth (Table 1). All the carbon substrates supported the production of polygalacturonase to a considerable extent. Among the raw carbon substrates used, potato peel had the best polygalacturonase synthesis followed by the ripe banana peel, then orange bagasse, ripe plantain peel and unripe plantain peel with cassava peel giving the lowest PG production (Table 1). Aspergillus flavus produced PG activity values of 840, 670 and 350 μ mg-1 in soluble starch, sucrose and control, respectively. The pH values of the culture filterates ranged from 6.20 to 6.35. In addition, the enzyme activity was highest in raw polysaccharides compared with soluble starch and sucrose, while the lowest PG activity was in carbon free basal mineral medium.
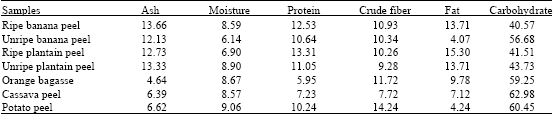
Ash content were within the range of 4.64 to 13.66%, with ripe banana peel having the highest, while orange bagasse had the least. The low moisture content ranged from 6.14 to 9.06%, crude fibres were within the range of 7.72 to 17.24%, while the fat content ranged from 4.24 to 15.30%. Ripe plantain peel had the highest protein content of 13.31%, followed by ripe banana peel 12.53%, then unripe plantain peel 11.05%, unripe banana peel 10.64%, while orange bagasse had the lowest protein content of 5.95%. Potato and cassava peels had protein content of 10.24 and 7.23%, respectively. The carbohydrate content of the raw carbon sources were moderately high ranging 40.57 to 62.98% with cassava peel recording the highest, while ripe banana peel had the least (Table 2).
All the carbon sources were moderately high in Na+, K+, Ca2+ and Mg2+, but low in Fe2+ and Mn2+ (Table 3).
| Table 1: | Maximum growth and polygalacturonase activity of A. flavus on different carbon sources |
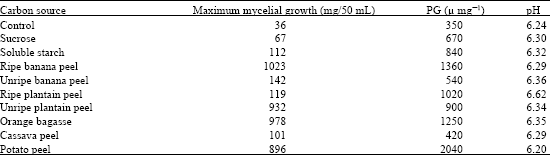 | |
| Control = Carbon free medium, PG= Polygalacturonase | |
| Table 2: | Proximate composition of raw carbon samples in percentage (%) |
 | |
| Table 3: | Mineral composition of raw carbon samples in part per million (ppm) |
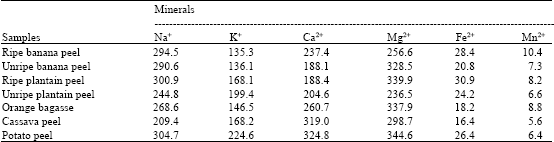 | |
The ability of the Aspergillus flavus to grow on the various carbon substrates is an indication of their utilization by the fungus. The raw carbon sources supported the growth of the fungus more than the commercial soluble starch and sucrose. However, the difference in utilization between the raw and commercial carbon could be due to the proportion of amylase to amylopectin which tend to vary from one starch/polysaccharide sources to another (Akinyosoye and Oboh, 2004). Also, this study revealed that the raw carbon substrates contained essential minerals (Table 3). These minerals together with vitamins, amino acids and other growth promoting substances, which are absent in the commercial soluble starch and sucrose, may also justify the substantial growth of the fungus on the raw carbon substrates (Akinyosoye and Akinyanju, 1989; Akinyosoye and Oboh, 2004). This finding is in agreement with the reports of Akinyosoye and Akinyanju (1989) that starch/complex carbon sources supported the highest fungal growth. In addition, these minerals are essentially required for the substance of normal growth and development of fungi (Griffin, 1981; Moore-Landecker, 1996). Moisture (water) is usually required by all organisms for their chemical reactions for life processes and fungi in particular require moisture or water in their environment for extracellular digestion of nutrient. The low and variations observed in the moisture content of the different raw carbon substrates may not exert any considerable effect on growth of the fungus, since it was grown in broth medium. However, the importance of moisture or water in the cultivation of fungi on solid substrates cannot be underscored (Olutiola et al., 1991; Moore-Landecker, 1996; Silva et al., 2005). In addition, may be the considerable effect of the protein content in the raw carbon substrates. This is in consonance with the reports that organic nitrogen is easily utilizable by fungi and that good growth occurred in complex nitrogen sources than the inorganic nitrogen (Moore-Landrcker, 1996; Vahidi et al., 2004). Although the relationship between lipid content of media and fungal growth is not clear, Nwanze et al. (2005) reported the significance effect of lipids on the mycelial wet and dry weight of Lintinus squarrosulus Mont. and Psathyrella atroumbonata Pelger in submerged liquid cultures. More importantly, another intrinsic parameter that may be responsible for the different growth response observed during the course of this work is the carbon to nitrogen (C/N) ratio. The carbon to nitrogen ratio influence rapidly with a high degree of efficiency on the rate of assimilation of nitrogen into microbial biomass (Zibilske, 1999). All the raw carbon substrates used in this study had reasonable carbon to nitrogen ratio (Table 2). Therefore, the profuse growth of Aspergillus flavus on the raw carbon substrates employed may be as a result of their C/N ratio against what was observed for soluble starch and sucrose substrates. This nature of the culture filterates is in line with the previous observation by Gbolagade (2006). The moderately acidic nature of the culture media, tend to favour the growth of the fungus. The pH range recorded in this study tally with the report of Wubah (1999). Another possible explanation for the low growth of the fungus in sucrose may be due to the interaction between glucose-product of hydrolysis of sucrose and phosphates in the basal medium. Griffins (1981) reported the reaction of phosphates with glucose to form ketoses and other products which may not be easily utilizable by Aspergillus flavus.
Aspergillus flavus produces polygalacturonase during growth on the various carbon substrates and carbon free basal medium (Table 1). The production of polygalacturonase in the carbon substrates by Aspergillus flavus is similar to the production of PG in Phorma sorghina (Akinyosoye and Oboh, 2004) and pectinase production capability of Aspergillus sp and their mutants (Eludire et al., 2003). The higher PG activity in the raw carbon substrates than the commercial soluble starch and sucrose may not be unconnected to the need for the fungus to hydrolyze the complex polysaccharides to simple sugars before utilization by the organism. This will definitely require the elaboration of more enzymes (Oboh and Akindahunsi, 2001; Akinyosoye and Oboh, 2004). The detection of PG in carbon free medium and carbon inclusion media suggests the constitutive and inducible nature of the enzyme in the fungus under investigation. However, the various carbon substrates induced the production of the PG considerably compared with the control-the carbon free medium. This finding corroborates the discoveries of Akinyosoye and Oboh (2004) and Eludire et al. (2003), while working with Phorma sorghina and Aspergillus species respectively. Polygalacturonases are widely used industrially. They are produced by species of bacteria, yeasts and moulds and even plant parasitic nematodes. However, fungi are the major sources of commercial enzymes since the pH optima of such enzymes are within the range naturally found in the materials used for processing (Ismail, 1996). All the carbon substrates used in this study are usually generated in abundance in Nigeria and other parts of Africa as agricultural wastes. In addition, they contained essential minerals and nutrients that can meet the requirement of the fungus which was responsible for the growth recorded. Therefore, these raw carbon substrates could be used in growing microorganisms for an inexpensive production of polygalacturonases.