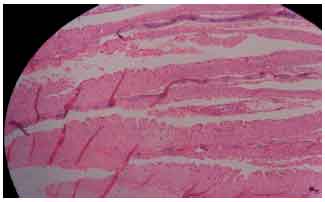
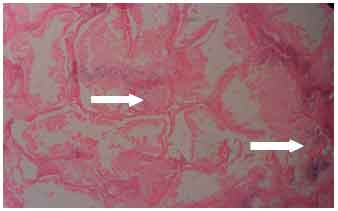
Research Article
Effect of Copper Sulphate on Spawning Success in African Catfish (Clarias gariepinus, Burchell 1822)
Department of Veterinary Public Health and Preventive Medicine, Fish and Wildlife Unit, University of Ibadan, Ibadan, Nigeria
F. Ajani
Department of Animal Science and Fisheries Management, Bowen University, Lwo, Osun State, Nigeria
O.K. Adeyemo
Department of Veterinary Public Health and Preventive Medicine, Fish and Wildlife Unit, University of Ibadan, Ibadan, Nigeria
J.O. Shobiye
Department of Veterinary Public Health and Preventive Medicine, Fish and Wildlife Unit, University of Ibadan, Ibadan, Nigeria