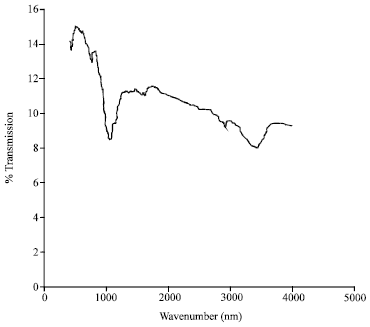
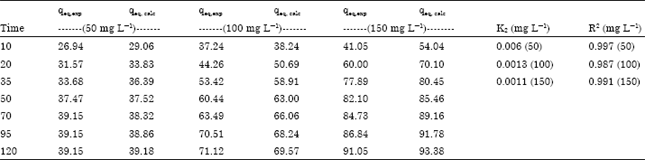
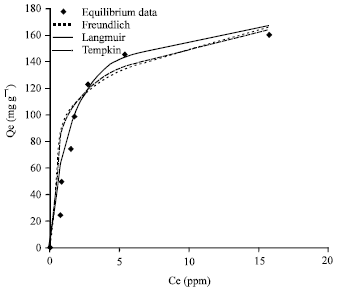
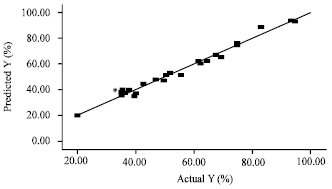
Research Article
Statistical Optimization for the Adsorption of Acid Fuchsin onto the Surface of Carbon Alumina Composite Pellet: An Application of Response Surface Methodology
Department of Chemical Engineering, Indian Institute of Technology, Kharagpur, West Bengal-721302, India
Jayanta Kumar Basu
Department of Chemical Engineering, Indian Institute of Technology, Kharagpur, West Bengal-721302, India