Research Article
Control Strategies of Stored Product Pests
Department of Pests and Plant Protection, National Research Centre, El-Behoose St., P.O. Box 12622-Dokki, Cairo, Egypt
For thousands of years, insects have been a problem associated with storing surplus dried food, with evidence from archaeological deposits (Buckland, 1981) and written records (Levinson and Levinson, 1994) of the storage and infestation of cereals stretching back to 3000 BC. There is a need to protect stored food from attack by insects because they can destroy large quantities, particularly during long-term storage (Pimentel, 1991). Allergic reactions to stored food arthropods, including Sitophilus granarius (L.), have been reported (Van Lynden-van Nes et al., 1996) and they can cause taint and contamination of grain with their excreta, cast skins and dead bodies (Scott, 1991).
In developed countries even the mere presence of a few insects in a bulk, at densities of considerably less than one insect per kg grain, can cause a serious loss in its market value. In some developed countries grain can be downgraded or rejected completely if even a single live insect is found (Pinniger et al., 1984).
In the UK the Food Safety Act (Anonymous, 1990) was amended recently; now all stores containing grain which might be destined for human consumption are treated as food premises and subject to inspection to ensure that food is not contaminated.
Early attempts to control stored grain pests relied on methods such as mixing dry soil and wood ash with the grain causing lethal dehydration of insects and the fumigant action of certain plant materials (Levinson and Levinson, 1989). In one form or another, the use of admixture and fumigation for the control of storage pests has continued to the present day. However, as our knowledge of the side effects of modern day, broad-spectrum pesticides and fumigants has increased, scientists have begun to explore new approaches to pest control. Concerns over pesticide operator safety and residues in our food and the environmental have led to a review of all organophosphorus pesticides by the Pesticide Safety Directorate in the UK; current advice is for farmers to avoid their use altogether unless there is no alternative (Rooker, 1999). Also, in Egypt, Ministry of Agriculture passed legislation to reduce agricultural pesticide usage by at least 50% by the year 2000.
The control of pests in commodity storage (farm and commercial) and food plants (food manufacturing processing and warehousing) requires a high degree of professionalism combined with experience and knowledge.
Pest control strategies must utilize combinations of techniques that are effective, economical and emphasize prevention of food product contamination. These strategies must be directed at immediate pest problems; at preventing future infestation problems; and must respond to routine daily needs, yet be flexible to meet emergency pest control situations. Stored-product pest control strategies tend to emphasize the non-chemical aspects of pest control with the judicious use of pesticides.
Unsatisfactory control of pests results in contaminated products that can cause health, financial, legal and aesthetic problems. Financial losses can result from (1) presence of live or dead insects in products and containers; (2) presence of odors, webbing and frass in products and containers; (3) loss in faith in the company by the consumer because of these conditions and (4) direct loss in weight resulting from insect feeding.
Control of stored-product pests is necessary to prevent contamination/adulteration of human foods. Persons involved in commodity/food storage, handling and/or processing have the responsibility to prevent food adulteration. Failure to do so can result in human illness and/or death, the violation of laws, loss of good will and resulting loss of revenue.
INVERTEBRATE STORED PRODUCT PESTS
A variety of pests may be found in stored commodities and food processing facilities, depending upon geographic location, physical nature of the facility and the type of food being processed.
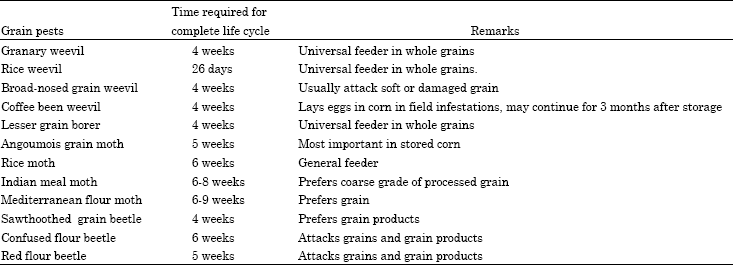
Insects: (Beetles, weevils and borers): Stored grain of almost any kind is subject to attack by insects. Pests which attack whole grain usually develop and feed inside the kernels of grain. These pests are not usually capable of existence outside the grain kernel as immature insects. Examples of whole grain pests are the rice weevil, the granary weevil, the lesser grain borer and the angoumois grain moth.
Other insects which attack grain are usually unable to penetrate whole grain. These insect pests however, can attack grain after it has been either mechanically broken or attacked by whole grain insects. Examples of these secondary pests are the confused and red flour beetles, Indian mealmoth, Mediterranean flour moth and the sawtoothed grain beetle (Table 1, Fig. 1).
Grain mite (Acarus siro L.): Mites will attack not only grain but cheese, flour, pet food, oilseeds, medicinal herbs, hay, deep litter or poultry houses, abandoned beehives and just about any food used by man.
Control: During heavy infestations cast skins and dead bodies accumulate into fluffy masses that can be blown around. It is a common inhabitant of house dust and is strongly allergenic. Vacuuming is required if areas are heavily infested. These signs indicated that storage conditions are too damp. Infested materials in homes should be discarded and the infested areas thoroughly cleaned.
 | |
| Fig. 1: | Principal stored grain insects, (For safe and effective use of insecticides, always identify the problem correctly) |
| Table 1: | List of stored grain insects and some biological aspects |
 | |
In commercial facilities infestations must be fumigated to gain control over these pests. Heavy infestations have a characteristic sweet or minty odor which usually necessitates the food be destroyed (Mason, 2004).
MONITORING FOR STORED-PRODUCT INSECTS
Stored-product insects cause significant losses of stored grains, processed foods, fibers and animal products. In general, losses can be minimized when infestations are quickly identified and appropriate control measures implemented.
Monitoring methods: Current monitoring methods for insect infestation vary and depend on the type of insect, commodity and/or storage. They include:
Bulk commodity storage:
| • | Observation of the commodity surface and over space for insects and/or evidence of their presence, i.e., webbing, cast skins, dust, odor, etc. |
| • | Examination of commodity samples obtained by various means, i.e., probe sampling, turning of the commodity, etc. |
| • | Probe (pitfall) traps inserted into the surface of grain masses and left for varying lengths of time have proven useful as a means of early detection of live, free-living insects |
| • | Temperature monitoring in grain masses may detect localized increased in temperature which can indicate the presence of an insect infestation |
Pheromone/food attractant monitoring: Numerous methods for detecting stored-product insects have been developed or are being investigated. The most promising technique that has been developed and continues to be refined, is monitoring populations with insect pheromones and/or food attractants.
The incorporation of monitoring methods into existing stored-product pest management programs can lead to earlier detection of low level infestations and pinpointing location of infestations. Monitoring information can be used to justify reduction in pesticide use or the need for intensified surveillance and pest management procedures. Monitoring results also can serve as an indicator of how well Integrated Pest Management (IPM) program components are functioning (Campbell et al., 2002).
| Table 2: | Status of pheromones for stored-product insects |
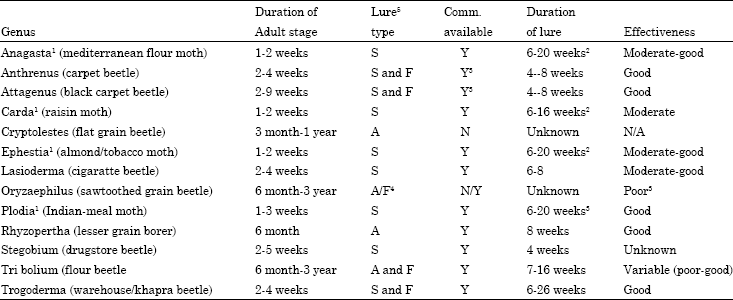 | |
| A: Aggregation pheromone; F: Food attractant; S: Sex pheromone. 1Same lure for all apecies, separate lure for candra under development. 2May be effective for up to 40 weeks based on USDA studies. 3Trogoderma traps utilizing food attractants will catch larve of both attagenus and anthrenus. Commercial sex lures have been marketed (1992) for carpet beetles, their effectiveness is not known. 4Aggregation pheromone under development. 5Food lure results have been erratic. Glue boards currently are almost as effective and more economical. Used with permission: Stored-product pest monitoring methods. Technical information memorandum No. 27 armed forces pest management board, June 1992 | |
Pheromone lures have been developed for several stored-product insects (Table 2). Lures developed for species with short-lived adults have proven more effective. The lure for the lesser grain borer (Rhyzopertha dominica) also has produced good results. Food attractant lures for stored-product insects (predominantly an oil lure consisting of oat oil, wheat germ oil extracts and mineral oil, wheat germ oil extracts and mineral oil) are used for species having long-lived adults and some larvae. These lures may be used with or without pheromones. In general, food attractant lures have a smaller effective range than pheromone lures. They can be used to enhance the effectiveness of pheromone traps for flour beetles (Tribolium) and to attract Trogoderma, Attagenus and Anthrenus larvae (Burkholder and Ma, 1985).
Inefficient trap design. Even when a highly attractive semiochemical is used to draw insects close to the trap, catch may still be low if the trap design fails to encourage the insects to enter the trap. Visual and secondary olfactory cues may be necessary; for example, adding vertical stripes to the outside of funnel traps increased the catch of Ephestia cautella (Walker), by 20 and 45% in moving and still air, respectively, compared with traps without stripes (Quartey and Coaker, 1992).
Regular checks reduce moisture risk in stored grain: To maintain grain quality, growers are urged to regularly sample the surface of grain bulks to check for insects, rising moisture levels and possible mould damage. The two main causes of moisture damage on the surface of grain bulks are condensation in the headspace (inside the roof of metal storage bins) and moisture migration within the grain bulk.
White painted bins: Heat-reflective white-painted bins reduce heating by sunlight and enhance passive cooling.
The temperature difference at the edges of a grain bulk near the walls and upper surface can be up to 5°C lower in painted bins.
Insulating the outer layers of grain from radiant heating improves the efficiency of aeration-cooling.
Reducing the fluctuations in grain temperature at the edges of a grain bulk between day and night also reduces the effect of night top silo cooling which causes condensation of water from air and wetting of the grain peak.
New method for detection of parasitized stored products with near-infrared spectroscopy: Near-Infrared Spectroscopy (NIRS) has been used in a variety of agricultural and food technology applications (Panford, 1987). Recent applications include the classification of insect species, detection of internal insect pests of wheat (infested or uninfested wheat kernels) (Dowell et al., 1998) and distinguishing between unparasitized weevil larvae in wheat and those parasitized by wasps (Burks et al., 2000). In this technique, the amount of light absorbed by materials is influenced by the number of molecules of specific constituents. Thus, quantitative information indicating the amount of chemical components such as water, oil, starch, sugar, or protein in agricultural products is measurable with NIRS (Murray and Williams, 1987). Fundamental absorptions usually occur in the mid-IR region (2500-15,000 nm), but 1st, 2nd and 3rd absorption overtones occur in the NIR region (700-2500 n). Advantages of measuring absorption in the NIR instead of mid-IR include lower sensor costs and less sample preparation.
Electronic nose technology for early detection of grain spoilage: Combine harvesting of both temperate and tropical cereals are most efficient when the ripened grain is slightly moist. Subsequent drying to safe moisture contents (e.g., for wheat this is 14% = 0.70 water activity) is essential to prevent initiation of fungal activity which can result in heating of the stored grain and ultimately end in spontaneous heating and complete loss of the grain. The activity of spoilage fungi can also result in the production of mycotoxins by certain genera, which prevents such moulded grain from being used for either animal or human consumption. Thus it is very important to detect fungal deterioration in stored cereal grains at an early stage. This would facilitate and improve existing management of grain stores. It would also allow remedial measures to be more effectively implemented, allowing significant losses and grain downgrading to be avoided. Mycotoxins are fungal secondary metabolites that have been associated with severe toxic effects to vertebrates produced by many important phytopathogenic and food spoilage fungi including Aspergillus, Penicillium, Fusarium and Alternaria species. The contamination of foods and animal feeds with mycotoxins is a worldwide problem (Kabak et al., 2006). A wide variety of volatiles are produced by fungi, either in vitro or when growing on agricultural grain substrates. Potential exists for distinguishing between species of fungi based on characteristic volatile patterns, which may be important when key spoilage fungi may be responsible for the production of harmful mycotoxins. It may be possible to use electronic nose systems to try and distinguish between grain colonised by mycotoxigenic and non-mycotoxigenic species and this area needs further investigation.
There is significant interest in methods for the early detection of quality changes in cereal grains. The development of electronic nose technology in recent years has stimulated interest in the use of characteristic volatiles and odours as a rapid, early indication of deterioration in grain quality (Magan and Evans, 2000).
Electronic nose technology coupled with neural network systems are developing very rapidly, both in terms of sensor development, sensitivity and data processing. This would suggest that in the very near future potential exists for the development of on-line systems through which the quality of stored grain could be monitored remotely as required and enable more efficient and effective long term management of grain storages to be carried out.
CONTROL STRATEGIES
Physical control: Insects in stored grain can be controlled by manipulating the physical environment or applying physical treatments to the grain and insects. The variables defining the physical environment that are usually controlled are: temperature, relative humidity or grain moisture content and relative composition of atmospheric gases in the intergranular air. Physical treatments include mechanical impact, physical removal, physical barriers to prevent the entrance of insects, abrasive and inert dusts, ionizing irradiation, light and sound.
Inert dusts: Inert dusts are chemically unreactive dusts that have insecticidal capability, killing by physical rather than chemical means. Insects coated in these dusts dehydrate and die. Because the effect is through desiccation, the effectiveness of inert dusts decreases as relative humidity increases. There is renewed interest in technology associated with use of inert dusts in grain storages and a number of reviews have been written (Ebeling, 1971; Banks and Fields, 1995; Golob, 1997; Subramanyam and Hagstrum, 2000; Mahdi and Khalequzzaman, 2006). Inert dusts have been used for centuries by aboriginal people in North America and Africa to control insects in their stored grain. There are five types of inert dusts (Fields and Muir, 1996):
| • | Sands and other soil components are traditional insecticides used by aboriginal peoples as a protective layer on top of stored seed (Golob and Webley, 1980) |
| • | Diatomaceous earth (DE) the fossilized remains of diatoms that are microscopic, unicellular, aquatic plants that have a fine shell made of opaline silica (SiO2 + n H2O). (DE) has extremely low toxicity to mammals (e.g., where DE toxicity for rat LD50 oral >5000 mg kg-1) (Subramanyam and Hagstrum, 1995) and (Anonymous, 1991a) are generally regarded as safe by the USA Environmental Protection Agency. Abd-El-Aziz and Sherief (2010) made chemical modification to the natural Diatomaceous Earth (DE) by different mono-, di-, tri-valent metal hydroxides (MOH, M = Na, Ca, Al) to increase its efficiency. The most effective DE modification was Ca.DE, which has insecticidal, repellent and ovicidal effects against Callosobruchus maculatus and can be used effectively in stored grain integrated pest management program |
| • | Silica aerogel, which is produced by drying an aqueous solution of sodium silicate, is a light powder that is not hygroscopic (Quarles, 1992) |
| • | Non-silica dusts, such as rock phosphate, have been used in Egypt (Fam et al., 1974) and lime (calcium oxide) provides some control (Golob and Webley, 1980) |
| • | Particle films (Kaolin and bentonite clays). The kaolinite-based particle films may also have potential for use as a dry dust material in stored-product environments, especially in specialty organic markets that prohibit the use of diatomaceous earth. Several studies were conducted to evaluate the potential of kaolinite-based particle films to control Tribolium castaneum (Herbst), the red flour beetle and Tribolium confusum (du Val) and the confused flour beetle. These two beetle species are major pests inside milling and processing facilities, food warehouses and food plants. The particle film M-96-018 was effective against both the Triblium species and appears to have a potential for use in management programs to control beetles within storage facilities (Arthur and Puterka, 2002) |
The main advantage of using inert dusts is that they are non-toxic to humans and animals. Diatomaceous earths are registered as a food additive in the USA. Inert dusts in stored grain can provide continuous protection from insect infestations and do not affect the baking quality of wheat.
Inert dusts kill insects by causing moisture to move out of the insects body. The dust either scratches through (diatomaceous earths) or absorbs (silica aerogel) the insect’s waxy coating that normally prevents excessive loss of moisture to the dry grain and intergranular air. The dust, because its mode of action is to dehydrate the insect, decreases in effectiveness as moisture content of the grain increases.
The main problem with inert dusts is that they decrease the bulk density of the treated grain. Because bulk density is presently a major factor in determining grain grades the dust can cause a decrease in the monetary value of the grain even though there have been no changes in its nutritional or functional value. The dust causes an increase in friction between the grain kernels so that the grain does not flow as readily as untreated grain. The angle of repose of the grain is increased and the rate of flow in down-spouts and along hopper-bottom floors will be reduced. The use of inert dust results in dusty conditions during application and grain handling. The dust may cause more wear on equipment.
Ionizing irradiation: Two types of ionizing irradiation have been used to control insects in grain. Gamma radiation can be produced by a radioactive isotope such as cobalt-60. Beta radiation, which is a beam of electrons, can be generated electrically. Irradiation with an electron beam is usually safer and easier to work with because it can be turned on and off while an isotope is always radiating and humans must be shielded from it (Fields and Muir, 1996). Ionizing radiation damages organisms by causing the production of ions or free radicals-charged molecules that are highly reactive. Besides ionization, chemical bonds can also be broken. To cause immediate death to stored product insects may require higher doses. Sterilization of many species of insects can be accomplished at lower doses. Rusty grain beetles are sterilized at only 0.6 kGy but sawtoothed grain beetles and red flour beetles require a 2.0 kGy dose (Banks and Fields, 1995). The grain mite however, requires a much higher dose of 4.5 KGy. Sterilized insects will do little harm to the grain but the grain will continue to appear to be infested and a grain buyer cannot be certain that the insects are sterilized. Following irradiation with gamma rays at 0.5 kGy complete insect mortality occurs in 14 days for rusty grain beetles, 28 days for red flour beetles, 70 days for sawtoothed grain beetles and 200 days for grain mites. Synergistic interaction indicates that microwaves radiation can be used with cold storage for management of Oryzaephilus surinamensis. This treatment could provide an effective and friendly environmental treatment technique in IPM program (Valizadegan et al., 2009).
The advantages of irradiation with beta rays are:
| • | A system can be set up to have a high throughput as the grain enters or leaves an establishment |
| • | The system can be readily automated |
| • | If proper safety controls are built into the system equipment operators are not exposed to any danger |
| • | All life stages of insects are affected by the radiation |
| • | No harmful chemical residues are left in the grain |
| • | Nutritional values are unaffected at low doses |
| • | Grain temperature is increased les than 0.1°C |
The disadvantages of irradiation include the following:
| • | Adult insects continue to live for sometime |
| • | There is no residual protection from reinfestation from external sources |
| • | The seed is killed by the irradiation required to control insects |
| • | Beta rays (electron beams) penetrate a layer of grain only about one kernel thick, 0.6 cm, while gamma rays penetrate 30 cm (Zakladnoi et al., 1982). The grain handling system must be designed to provide the required thin layer |
| • | The system has a high capital cost; therefore, to be economic it must have a high throughput for several years |
Light and sound: Light may be of some use in luring flying insects into traps (Banks and Fields, 1995). A 5-min exposure to 1 MHz sound at 14.5 W cm-2 killed all stages of S. granarius at 26°C in wheat but commercial application is unexpected (Banks and Fields, 1995).
Thermal control
Control of insects with low temperatures: The optimal temperature for stored product insects is between 25-33°C (Table 3).
Low temperatures reduce insect development and thus lengthen the time before populations increase to a point where they cause significant damage (Table 4).
Mites in damp grain can continue developing down to 2°C. Although no development occurs at these temperatures, insects and mites remain alive for long periods and will cause damage if the grain temperature rises. Burrell (1967) reported that cooling prevented further development of the heavy infestation but since the insects were not killed, this method of control should be used as a preventative rather than a cure.
| Table 3: | The response of stored-product insects to temperature1 (Banks and Fields, 1995; Fields and Muir, 1996) |
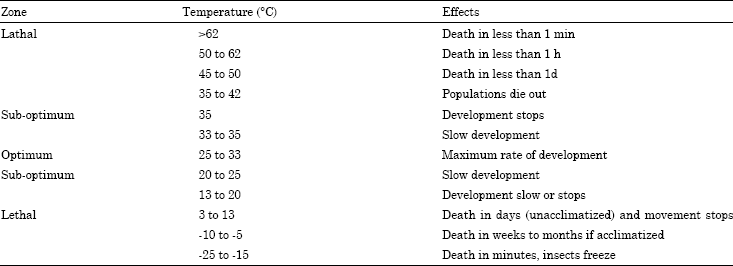 | |
| 1Species, stages of development and moisture content of food will influence the response to temperature | |
| Table 4: | Effect of low temperature on developmental stages of various grain insects |
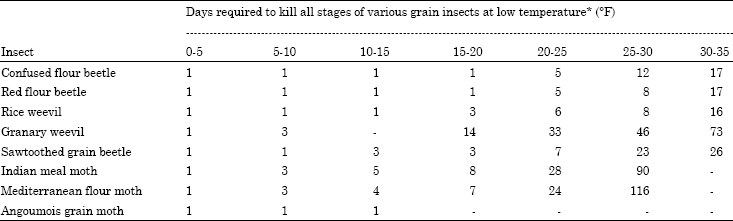 | |
| *Adapted from: Richard T. Cotton. Pests of stored grain and grain products | |
Insects exposed to cool temperatures (20-10°C) become acclimatized and their cold-hardiness can increase by 2 to 10 times. Under normal storage conditions insects in stored grain are acclimatized as a grain bulk slowly cools by conduction and free convection to its periphery.
Lowering the temperature of commodities to prevent spoilage is an ancient technique. Part of the success of the underground storages used in ancient Egypt compared with above ground stores was due to cooler temperatures of the underground stores (Banks and Fields, 1995). The effectiveness of aeration can be increased in tropical climates or in the summer in temperate climates by cooling the air with refrigeration units. Low temperatures reduce the rates of development, feeding, fecundity and survival (Logstaff and Evans, 1983). Temperatures between 13 and 25°C will slow development. This lengthens the time before populations increase to point where, they cause significant damage. S.granarius is the only major stored-product insect that can develop below 20°C and down to 13°C in optimum moisture content grain (Banks and Fields, 1995). Mites in damp grain only stop developing at 2°C. Although no development occurs at these temperatures, insects and mites remain alive for long periods and will cause damage if the temperature of the commodity rises.
Burks et al. (1999) demonstrated that low temperatures prevents emergence of unparsitized larvae of the rice weevil Sitophilus oryzae (L.) while not affecting emergence of parasitoid Anisopteromalus. calandrae.
High temperature disinfestation: Possible methods of heating grain; hot-air convection, infrared and microwave radiation all appear to require similar grain temperatures to achieve the same lethal effect. Most heated-air grain-driers cannot heat the grain uniformly and efficiently to the desired temperature. Fluidized beds, spouted beds, pneumatic conveyors, a counter-flow heat exchanger (Lapp et al., 1986), high frequency waves (Nelson and Kantack, 1966), microwaves (Locatelli and Traversa, 1989) infrared waves and solar radiation (Kitch et al., 1992) have been used to satisfactorily disinfest seeds. Maximum kernel temperatures and kernel residence times must be carefully controlled to obtain disinfestations without thermal damage to the kernels (Evans et al., 1983). Most of the energy cost of the process is in heating the grain, evaporating moisture concurrently, plus the unused heat in the exhaust air. Dielectric heating (but not microwave heating) may heat insect tissue more rapidly than grain, thus reducing energy costs of heat disinfestations.
Grain temperatures of 60 to 65°C for a few seconds or minutes are necessary to kill all stored grain insects. The same minimum temperature is required to kill the insects whichever method is used to heat the grain. Such temperatures can damage the baking quality of wheat, the malting quality of barley and the germination of most seeds (Evans, 1987). Thus the temperature of the grain must be carefully measured and controlled.
Selection of the method to heat the grain will depend on capital costs and energy efficiencies of the various heating systems and the relative costs of various energy sources. The same minimum temperature must be attained to kill the insects when using any of the available methods of heating the grain. The simplest and least costly method is probably heating the grain with hot air in a grain drier or fluidized bed.
Grain can be heated by passing a thin layer of grain under an infrared or microwave radiation source. Separate equipment to cool the grain must also be included in a radiant heating system. Because radiation is produced with electricity, the energy cost of radiant heating is usually higher than using fossil fuels to heat air and grain even though the heated air system is less energy efficient.
Fluidized bed heating is the only method of high temperature disinfestation developed to the stage of a full-scale prototype for treating more than 100 t h-1 of grain. Others have been successfully demonstrated at laboratory or pilot plant stages of development (e.g., Sutherland et al., 1987).
Systems, such as fluidized beds, have the advantage that they can easily be adapted for cooling the treated grain after exposure. Such cooling is necessary as grain held for more than brief periods at temperatures that are rapidly insecticidal (>60°C) loses important qualities (e.g., germination, baking quality). Approaches using radiative heating will require grain cooling after treatment, adding to the cost and complexity of the system.
A technique known as. bake-out., where structures are heated to around 50°C for about one day may hold potential as a structural treatment in grain storage sheds. Dryacide can assist bake-out effectiveness when applied to hard-to-heat surfaces such as external walls and cavities (Neeson and Banks, 2004).
Ozonation: Ozone (O3) is an allotrope of oxygen, which can be generated by UV-light and electrical discharges in air (corona-discharge). Ozone generation by electrical discharge is most common and has several advantages, including greater sustainability of the unit, higher ozone production and higher cost affectivity. Ozone has a half-life of 20-50 min, rapidly decomposing to diatomic oxygen, a natural component in the atmosphere. Ozone has a half-life of 20-50 min, rapidly decomposing to diatomic oxygen, a natural component in the atmosphere. Because ozone can be easily generated at the treatment site using only electricity and air, it offers several safety advantages over conventional post-harvest pesticides. First, there are no stores of toxic chemicals, chemical mixing hazards, or disposal of left over insecticides or containers (Law and Kiss, 1991). Second, with a short half-life, it reverts back to naturally occurring oxygen leaving no residue on the product or to dispose of. Third, if needed it would be possible to neutralize ozone through techniques such as thermal activated charcoal, as well as catalytic and chemical abatement (Law and Kiss, 1991).
Ozone, a known sterilant, can be used as an insect control agent in food commodities at levels less than 45 ppm. Ozone is readily generated from atmospheric oxygen and is safe to the environment when used for fumigation. However it is highly unstable and breaks down to molecular oxygen quickly. A major disadvantage with ozone is its corrosive property towards most of the metals (Mason et al., 1998). Mendez et al. (2002) showed that treatment of grains with 50 ppm ozone for 30 d had no detrimental effect on popping volume of popcorn, fatty acid and amino acid composition of corn and milling characteristics of corn. These data indicate that if repeated ozone treatments are needed, such treatment should not decrease the quality of grain for end-users. These results suggest that ozonation is a potential alternative to conventional pest control treatments.
Active research is going on to exploit ozone as a potential quarantine treatment for controlling stored-product pests (Hollingsworth and Armstrong, 2005).
Fumigation: Vijayanna (2006) mentioned that Fumigation is a method of pest control that completely fills an area with gaseous pesticides to suffocate or poison the pests within. It is utilized for control of pests in buildings, soil, seed and grain produce. It is also used during processing of goods to be imported or exported in order to prevent transfer of exotic organisms. Fumigants are chemical compounds in gaseous form that enter the body of insect through the spiracles spread all over via trachea and tracheoles.
Fumigation plays a major role in insect pest elimination in stored products. Currently, phosphine (from metal phosphide preparations, cylinderized formulations and on-site generators) and methyl bromide (available in cylinders and metal cans) are the two common fumigants used for stored-product protection world over (Rajendran and Sriranjini, 2008). Insect resistance to phosphine is a global issue now and control failures have been reported in field situations in some countries (Taylor, 1989; Collins et al., 2002). Methyl bromide, a broad-spectrum fumigant, has been declared an ozone-depleting substance and therefore, is being phased out completely. In view of the problems with the current fumigants, there is a global interest in alternative strategies including development of chemical substitutes, exploitation of controlled atmospheres and integration of physical methods (MBTOC, 2002). Recently, the use of sulphuryl fluoride, a structural fumigant for termite and woodborer control, has been expanded to food commodities and food handling establishments (e.g., flourmills) in the USA, Canada and Europe (Prabhakaran, 2006). New fumigants such as carbonyl sulphide and ethane dinitrile and the old fumigant ethyl formate (alone and in mixture with CO2) (Damcevski et al., 2003) have also been investigated as alternatives for food and non-food commodities.
Semiochemicals for stored product control: Semiocemicals could be used to control insect pests by behavioural manipulation. Furthermore, interest has been shown in plant products, i.e., essential oils and their components, for fumigant action since it is believed that natural compounds from plant sources may have the advantage over conventional fumigants in terms of low mammalian toxicity (not true in all cases), rapid degradation and local availability (Rajendran and Sriranjini, 2008). Many reviews of the literature have identified many plant genera containing species producing chemicals with potential as repellents or antifeedants for the control of insects including stored grain pests (Prakash and Rao, 1997; Abd-El-Aziz and Ismail, 2000; Abd-El-Aziz, 2001; Dimetry et al., 2002; Sabbour, 2003; Sabbour and Abd El-Aziz, 2007; Haghtalab et al., 2009; Mondal and Khalequzzaman, 2010). Compounds derived from plants continue to be assessed for their potential to control storage pests (Allotey and Azalekor, 2000). By choosing those plant species that are natives or naturalized introductions to a particular region, their exploitation as insect control agents should not pose any of the ethical or quinones problems associated with non-indigenous plants. These would appear to be a good starting point from which to choose a repellent, for example to use in a protective band around grain bulks or as a flushing agent to disinfest store structures. Also, chemicals such as quinines produced by some storage beetles themselves could be assessed for their potential as practical control agents. Ideally any repellent used in these ways should avoid direct contact with the grain unless it can be shown there is no associated health risk or possibility of tainting. In general it is likely that larger quantities of repellents than attractants would be required for practical pest control, so the use of repellents may prove to be more expensive and hold a greater risk of contaminating the grain. On the other hand, production of repellents derived from plants may be easier and less expensive than the synthesis of some more complex attractive semiochemicals.
Evidently, compounds of plant origin can be used only for small-scale applications or for space treatments. They require some carrier gases (e.g., CO2) for even distribution and penetration into the commodities or they can act as adjuvants for conventional fumigants.
The effect of repellents on penetration into packaging by stored-product insects: Although, finished products can be shipped from production facilities uninfested, stored-product insects can enter packaged goods during transportation, storage in the warehouse, or in retail stores. Ultimately, the consumer of the products holds the manufacturer responsible for any insect infestation, even if the cause of the problem is poor storage by a third party. The packaging of products is the last line of defense for processors against insect infestation of their finished products. There are two types of insects that attack packaged products: penetrators, which are insects that can bore holes through packaging materials and invaders, which are insects that enter packages through existing holes, such as folds and seams and air vents (Highland, 1991; Newton, 1988). Sitophilus spp., Rhyzopertha dominica (F.), Plodia interpunctella (Hübner), lasioderma serricorne (F.),and Stegobium paniceum (L.) are some of the stored-product insects that are capable of penetrating food packaging. However, Tribolium spp., Cryptolestes ferrugineus (Stephens),and Oryzaephilus sp. cannot penetrate intact packages and must enter through existing holes in the packae (Highland, 1991).
In addition to improving the packaging materials and design, insect repellents are used to present insects from entering packages by modifying the behavior of insects (Highland, 1991; Mullen and Mowery, 2000). Pyrethrins synergized with piperonyl butoxide were approved for use as a treatment for insect-resistant packaging on the outer layer of packages or with adhesive in the USA (Highland, 1991). The repellency of pyrethrins was the primary mode of action against insect penetration and invasion (Laudani and Davis, 1955). Methyl salicylate, an insect repellent, has been registered to be used in food packaging to control stored-product insects in the USA (Radwan and Allin, 1997). DEET, neem and protein-enriched pea flour are repellent to many stored-product insects when tested by exposure on filter paper or in preference chambers (Fields et al., 2001).
The number of S. oryzae, T. castaneum, C. ferrugineus and O.surinamensis entering pierced paper envelopes that contained wheat and were treated with DEET was reduced by 99, 86, 97 and 91%, respectively. Neem was less effective than DEET in reducing penetration and invasion of insects. Protein-enriched pea flour did not prevent insects entering pierced envelopes (Hou et al., 2004). Sabbour and Abd El-Aziz (2007) screened the most suitable packaging materials (muslin, paper (multi-wall paper (3 layers)), cheesecloth, wax paper, gunny bags and polypropylene) for prevention of broad bean beetles infestation. Gunny sacks followed by paper bags (multi-wall paper (3 layers) provided more protection against C. maculatus infestation than the other tested packing materials.
Microbial control: Synthetic chemical insecticides provide many benefits to food production and human health, but they also pose some hazards. In many instances, alternative methods of insect management offer adequate levels of pest control and pose fewer hazards. One such alternative is the use of microbial insecticides that contain microorganisms or their by-products. Microbial insecticides are especially valuable because their toxicity to non-target animals and human is extremely low. Compared to other commonly used insecticides, they are safe for both the pesticide user and consumers of treated crops.
In general, Bt is very safe to use. The EPA has found no hazards to human health associated with the use of Bt. It has no known carcinogenic or mutagenic properties at field application rates or in laboratory studies. There are no known poisonings or chronic exposure problems. However, in concentrated form, it may cause sensitization in hypersensitive individuals following long inhalation exposure or accidental injection.
Currently, many entomopathogens are used for the control of invertebrate pests in glasshouses, row crops, orchards, ornamentals, stored products and forestry (Lacey et al., 2001; Khashaveh et al., 2008; Mahdneshin et al., 2009). The effect of two microbial entomopathogens (Bacillus thuringiensis and Beauveria bassiana and three botanical extracts were studied on three stored product insects, Plodia interpunctella, Ephestia cautella and Ephestia kuehniella. Botanical extracts tested when combined with B. thuringiensis caused a significant enhancement to the pathogens and increased the mortality in almost all cases (Sabbour, 2003).
Buda and Peciulyte (2008) tested the effect of four fungi species (Beauveria bassiana, Lecanicillium (Verticillium) lecanii, Metarhizum anisopliae var. anisopliae and Paecilomyces farinosus) isolates on adults of Indian meal moth, Plodia interpunctella and one species tested on mature larvae of the pest. All the fungal isolates tested were pathogenic, however, with a different effectiveness. During the first three day period after spraying, the highest mortality (35-40% versus control) was caused by P. farinosus and M. anisopliae var anisopliae and there was no significant difference in the survival as compared to control when B. bassiana and L. (Verticillium) lecanii were used.
Sabbour and Abd-El-Aziz (2010) mentioned that the persistent effect of formulated mustard oil with either P. fumosoroseus or Nomuraea rileyi fungi on foam covering gunny bags displayed several different modes of action, by reducing oviposition and adult emergence (F1) of Bruchidius incarnatus. The oviposition was completely inhibited when stored broad bean seeds were treated with mustard oil + P. fumosoroseus during 20, 40 and 60 days of storage. Application of mustard oil combined with P. fumosoroseus on foam covering gunny bags provided promising oviposition deterrency, toxicity and suppressing B. incarnates infestation, persistence and protecting broad bean seeds from beetles’ infestation for 120 days during storage.
Heat, desiccation (drying out), or exposure to ultraviolet radiation reduces the effectiveness or several types of microbial insecticides. Consequently, proper timing and application procedures are especially important for some products. Special formulation and storage procedures are necessary for some microbial pesticides.
Biological control of stored product pests: Biological control is an over-looked component of integrated pest management of stored product pest (Flinn et al., 1998). Many species of insect natural enemies occurs in stored product ecosystem (Brower et al., 1996) and these species represent potential biological control agents for the desired pests. The anthocorid bug, Xylocoris flavipes (Reuter) is a cosmopolitan predator of different prey (pests) of stored commodities namely Tribolium castaneum, T. confusum, Crytolestes pusillus, Rhizopertha dominica and Trogoderma granarium (Ahmed et al., 1991; Rahman et al., 2009).
Important natural enemies include parasitoid wasps in the families Braconidae, Ichneumonidae, pteromalidae and Bethylidae and predatory pirate bugs. A collection of other Predators can be found in some situations and these include assassin bugs, hister beetles, pseudoscorpions and predatory mites.
Research during the 1970’s and 1980’s demonstrated the efficacy of parasitoids and predators in controlling storage pests. Releases of the warehouse pirate bug, Xylocoris flavipes, resulted in a 79-100% suppression of moth populations in small storages of peanuts, up to 99% reduction of sawtoothed grain beetle populations in 35-quart lots of corn and a 90-98% suppression of red flour beetles in a simulated peanut warehouse. Rice weevils in wheat spillage in small rooms were suppressed 96% by the parasitic wasp Anisopteromalus calandrae. When the egg parasitoid Trichogramma pretiosum and the larval parasitoid Bracon hebetor were released together in simulated peanut warehouses, they suppressed Indianmeal moth populations by 84% and almond moth (Cadra cautella) populations by 98%, B. hebetor alone supressed almond moth populations by 97.3%. Several other studies have yielded similar results that suggest these natural enemies should provide effective biological control of storage pests. However, these experiments were conducted under laboratory conditions in which the commodities, storage containers or structures and numbers of pest insects were under strict control. Biological controls did not prevent economic damage, partly due to the migration of pest species into the experimental bins. Recently the predatory beetle Teretriosoma nigrescens (Lewis) has been introduced into west Africa to control Prostephanus truncatus (Horn), with moderate success (Markham et al., 1994). Validation studies will be necessary to fully determine the potential for biological controls as replacements for insecticidal protectants and currently such data are lacking for most grain storage system throughout the world.
The negative effects of agricultural pesticides on natural control agents is often cited to support the development of alternatives to chemical controls. The impact of pesticides on natural enemies and the resulting outbreaks of secondary arthropod pests have been documented in many field agricultural systems (Croft, 1990).
Biological controls are not normally considered to be compatible with protestants. However, there is potential to select for insecticide resistance in certain parasitic and predatory species (Baker, 1995; Baker and Arbogast, 1995). Integrating the use of predatory/parasitic mites and insecticides for stored-grain pest management requires knowledge of the impact of the insecticides on the natural enemies or, in other words, their selectivity to natural enemies (Baker and Arbogast, 1995; Goncalves et al., 2002). Goncalves et al. (2004) mentioned that the effect of insecticides on the mite species Acarophenax lacunatus (Cross and Krantz), an egg parasite of the stored grain pest Rhyzopertha dominica (Fabr.). Deltamethrin was less selective in favor of the mite species. Nonetheless the parasitic mite was able to parasitize eggs of R. dominica on wheat treated with all the insecticides evaluated.
There is great potential for using biological control to control pests in stored products. Research is continuing to determine the proper prescriptions for use of natural enemies in stored grain. Behavioral, ecological and physiological data are being collected that will facilitate effective deployment of parasitoids and predators. Storage situations other than grain bins, such as feed mills, food warehouses and food factories may be targeted areas for biological control in the future. As part of an IMP system for stored product management, biological control should help reduce the use of pesticides on food and provide for high quality food products.
Insect growth regulators: Insect Growth Regulators (IGRs) are chemicals which mimic hormones that control molting in insects and thereby disrupt development. Oberlander et al. (1997) reviewed many studies involving IGRs and stored product-insects. Most of the published research involved exposure of eggs, 1st instars, or adults on treated grain, in diet or in glass vials, with subsequent measurements of progeny production. Morphological effects and reduced fecundity in adults have been reported from some of these studies with stored-product insects (El- Sayed, 1988).
Many stored-product insects occur not only in raw grains, but also in mills, processing plants, food warehouses and other indoor areas and there is comparatively little research with IGRs that is applicable to insect control programs in those areas. In a recent test late-instar larvae of Tribolium castaneum (Herbst), the red flour beetle and Triboltium confusum (du Val) were directly exposed on concrete treated with hydroprene (Arthur, 2001). Larvae often failed to molt to the pupal stage and adults that did emerge were usually morphologically deformed and quickly died. In addition, T. castaneum appeared to be more morphologically deformed and quickly died. In addition, I. castaneum appeared to be more susceptible to hydroprene than T. confusum.
Currently there is renewed interest in developing reduced-risk low toxicity chemicals, including new formulations of IGRs, to replace older conventional products used in many agricultural systems, including post-harvest protection in areas where food is stored. Expanded use of these IGRs could also be facilitated by identifying physical and biological factors that can compromise efficacy when IGRs are used as a surface treatment. The new hydroprene formulation were equivalent to the registered product Gentrol. Tribolium confusion was less susceptible than T. castareum and residual control of T.confusum, lasted 6-12 weeks (Arthur and Hoeremann, 2004).
IGRs have been developed commercially and are being use to control insect pests in agriculture, forestry, public health and stored products. IGRs affect the biology of treated insects, for example, both embryonic and post-embryonic development, reproduction, behaviour and mortality. Abnormal morphogenesis is the observed effect of IGR action on the insects. Many of them are more potent than current insecticides, even against the eggs.
Compared with the conventional insecticides, IGRs do not exhibit quick knock-down in insects or cause mortality, but the long-term exposure to these compounds largely stops the population growth, as a result of the effects mentioned in both the parents and progeny (Mondal and Parween, 2000).
Hygiene: Well managed aeration and effective hygiene can overcome 85% of storage pest problems facing grain growers.
By following a few relatively simple strategies, growers can ensure their on-farm storage systems maintain grain quality for future sales. Once a grain store has been emptied and physically cleaned, it is normal to reduce the residual insect populations by use of structural treatments. Until recently, the only chemicals registered for such usage were azamethiphos or a mixture of fenitrothion and carbaryl. Another type of treatment has been developed that leaves no synthetic chemical residues that could contaminate food. The treatment involved spraying a slurry of Dryacide®, a diatomaceous earth coated with silica aerogels, on to the structure. This technique has undergone extensive trails in the field and has been shown to give at least 12 months protection (Anonymous,1991b).
There is an obvious need to assess the efficacy of hygiene procedures and structural treatments prior to in-loading of grain. Since, inspection and sampling procedures are labour-intensive, food-baited traps, developed elsewhere (De Coursey, 1931; Pinniger et al., 1984), have been used recently in Australia with considerable success and have demonstrated the long-term benefits of using Dryacide® to suppress residual populations in empty storage structures (Wright, 1990).
Good hygiene in the grain store or storage depot is important in maintaining grain and seed quality. Guidelines for hygiene in the grain store include:
| • | Keep storage areas clean. This means sweeping the floor, removing cobwebs and dust and collecting and removing any grain spills |
| • | Clean storage rooms after they are emptied and this may include spraying walls, crevices and wooden pallets with an insecticide before using them again |
| • | Placing rat-traps and barriers in drying and storage areas. Cats deter and help control rats and mice |
| • | Inspect storage room regularly to keep it vermin proof |
| • | Inspect the stored seeds once a week for signs of insect infestation. When necessary and only under the direction of a trained pest control technician, the storage room or the seed stock may be sealed with tarpaulin and treated with fumigants |
Building maintenance is an important aspect of hygiene and exclusion in a feed mill or grain storage facility. Where possible, seal unloading auger, openings and pipes. Holes should be covered with a screen of fine mesh and any leaky roofs, seals, joints and holes promptly repaired.
Development and marketing of expert systems: Good control strategies are based on reliable methods for estimating insect population level to determine if control measures are warranted. Probe traps and pitfall traps are more efficient than trier samples for detecting insects in stored grain (White et al., 1991). Computer-based decision support systems that use biological and environmental data to predict population trends and evaluate the need for insecticidal inputs have been developed for stored-product storage systems in several countries (Jones et al., 1993; Longstaff, 1994). In certain situations protectants may be eliminated and insect control may be achieved through temperature manipulation or supplemental fumigation should infestations occur during storage. As more of these expert systems are developed management decisions may shift to computer-based pest management.