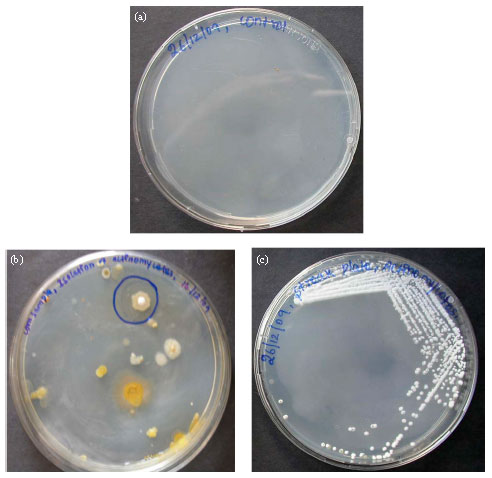
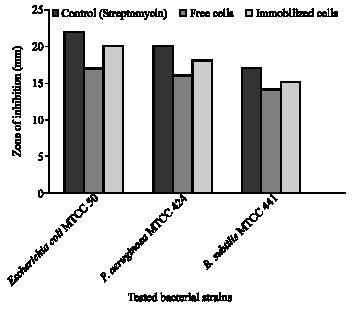Research Article
Isolation, Characterization, Screening and Antibiotic Sensitivity of Actinomycetes from Locally (Near MCAS) Collected Soil Samples
Centre for Biotechnology, Muthayammal College of Arts and Science,Rasipuram-637408, Tamilnadu, India
N. Selvan
Centre for Biotechnology, Muthayammal College of Arts and Science,Rasipuram-637408, Tamilnadu, India
K. Dhanapal
Centre for Biotechnology, Muthayammal College of Arts and Science,Rasipuram-637408, Tamilnadu, India