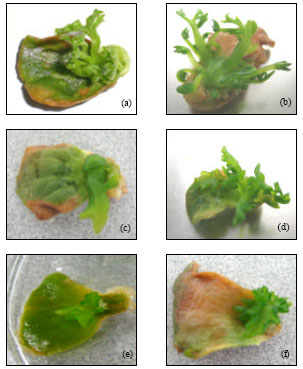
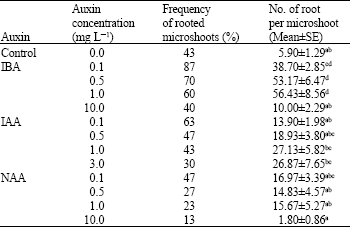
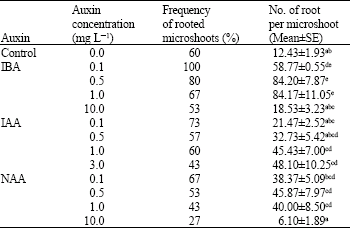
Research Article
In vitro Regeneration of Citrullus lanatus cv. Round Dragon
Department of Industrial Biotechnology, Faculty of Biosciences and Bioengineering, Universiti Teknologi Malaysia, 81310 Skudai, Johor, Malaysia
Fahrul Huyop
Department of Industrial Biotechnology, Faculty of Biosciences and Bioengineering, Universiti Teknologi Malaysia, 81310 Skudai, Johor, Malaysia