Research Article
Factors Affecting the Prevalence of Human Schistosomiasis in Aseer Region, Saudi Arabia
Department of Biological Sciences, College of Science, King Khaid University, P. O. Box 10347, Abha 61321, Saudi Arabia
Schistosomiasis is still a major helminth infection at the beginning of the 21st century and an important public health problem in many non-industrialized countries. As the second major parasitic disease in the world after malaria, schistosomiasis affects 200 million people, 800 million being exposed to the risk of infection. It is also estimated that 20 million individuals suffer from severe consequences of this chronic and debilitating disease responsible for at least 500,000 deaths per year (Capron et al., 2002; Gryseels et al., 2006).
Schistosomiasis caused by worms of the genus Schistosoma. The three main species infecting humans are Schistosoma haematobium, S. japonicum and S. mansoni and transmission cycle requires specific freshwater snails as intermediate hosts (Engels et al., 2002; Zhou et al., 2008).
According to the Statistical Year Book of Ministry of Health in Saudi Arabia (2002), the mean prevalence of schistosomiasis in the Kingdom of Saudi Arabia (KSA) from 1990-2000 was 2.2/100,000. The highest schistosomiasis rates were recorded in Aseer (39%), Jazan (27.6%), Bishah (16.3%), and Al-Bahah (9.8%). The least rate was recorded in Riyadh (0.2%) and Tabouk (0.2%). Both Najran and Al-Jouf reported no case. In addition, it is noticed that there is still on going transmission of infection in Jazan, Aseer and Al-Bahah as new cases are discovered among the young and middle-aged individuals.
Both abiotic and biotic factors contribute to the schistosomiasis transmission cycle and these determine the spatial distribution of the disease (Cox, 1993; Satayathum et al., 2006; Zhou et al., 2008). The abiotic factors affecting schistosomiasis transmission include climatic factors such as temperature and rainfall (Michelson, 1961; Appleton, 1978; Martens, 1995; Zhou et al., 2008). These factors, however, can affect the prevalence of schistosomiasis indirectly as they affect the breeding and development of the intermediate snail hosts (Appleton, 1978; Thieltges et al., 2008). In addition, it has been reported that many biotic factors can affect the prevalence of schistosomiasis such as host sex and age as a result of behaviour, hormonal or genetic reasons (Cox, 1993; Brooker, 2007).
As Aseer region showed the highest rate of human schistosomiasis in Saudi Arabia in the past few years, it is necessary to investigate the temporal and spatial variations in the prevalence of infection within this region. It is also important to investigate the role of biotic and abiotic factors which may affect the prevalence of human schistosomiasis in the study area. Therefore, this study aims to study the temporal and spatial variations in the prevalence of human schistosomiasis in two areas in Aseer region: Abha and Tihamah over eight years period (2000-2007). In addition, the contribution of various biotic and abiotic factors (i.e., location, year, season, host sex, host age, proportion of snails infected with schistosomes and the nationality of people examined for schistosomiasis in Aseer region) to the prevalence of the infection will be estimated. This is in order to have a better understanding of the epidemiology of schistosomiasis in Aseer region and hence to develop good and effective schistosome control programmes.
Study areas: The study took place in two areas in Aseer region (Fig. 1):
| • | Abha which is located in the south-western Saudi Arabia. it is highly elevated as it is situated at 2,200 m (7,200 feet) above sea level. The area is subjected to high annual rainfall. It also has cooler weather in winter as temperature reaches 5 Celsius. However, it has a mild weather in summer as temperature do not exceed 34 Celsius |
| • | Tihamah area which is the lowland located on the Red Sea coast. The weather in this area is hot in the summer and mild in the winter. The average temperature throughout the year is about 23-25 Celsius. The average rainfall is about 45-100 mm per year and there are many rivers, dams and open wells in this area |
Data collection and analysis: Data on human schistosomiasis in Aseer region from 2000 to 2007 were obtained from the Vector Department, Saudi Ministry of Health. Variation in the prevalence of human schistosomiasis (proportion of infected people) was analysed using Generalised Linear Models (GLMs) with binomial error and a logit link function (Crawley, 2002) using the R software v 2.9.0 (R Development Core Team, 2009). The Generalised Linear Model (GLM) is a flexible generalisation of ordinary least squares regression. The GLM generalises linear regression by allowing the linear model to be related to the response variable via a link function and by allowing the magnitude of the variance of each measurement to be a function of its predicted value. The GLMs were formulated as a way of unifying various other statistical models, including linear regression, logistic regression and Poisson regression, under one framework. This has allowed the development of a general algorithm for maximum likelihood estimation in all these models (Mccullagh and Nelder, 1989).
Proportion of human schistosomiasis infection was entered as a dependent variable. The explanatory variables were: area (two levels: Abha and Tihamah), year (eight levels: 2000-2007), season (four levels: spring, summer, autumn and winter), host age (three levels: 0-14.15-35 and >36), host sex (two levels: male and female), proportion of infected snails and nationality of examined people (two levels: Saudi and non-Saudi).
 | |
| Fig. 1: | Map of the Saudi Arabia showing (a) Aseer region and (b) the two study areas in Aseer |
Backward stepwise model selection based on Akaike Information Criteria (AIC) to identify the models best fitting the data. Models with a difference in AIC<2 compared to the lowest were considered not to differ in their ability to describe the data (Venables and Ripley, 2002).
Analysis of the prevalence of human schistosomiasis in Aseer region using data of 1004953 people (682982 from Abha and 321971 from Tihamah) over eight years (2000-2007) showed that there was a decline in the prevalence of schistosomiasis over the study period and the prevalence of infection in year 2007 was the lowest and significantly differed from the prevalence in all other years except year 2006 (Table 1).
In addition, there was a special variation in the prevalence of schistosomiasis in Aseer region as it was significantly higher in Abha than it was in Tihamah (Table 1).
Prevalence of human schistosomiasis was affected by seasons in Abha but not in Tihamah (Fig. 2). The prevalence of infection peaked in the summer season in Abha and was significantly higher than it was in other seasons (Fig. 2, Table 1). However, the rate of schistosomiasis prevalence was almost similar in autumn, spring and winter in Abha (Fig. 2). In Tihamah, the prevalence of infection in all seasons did not differ from each other (Fig. 2).
| Table 1: | Estimates of parameters contributing to the prevalence of human schistosomiasis in Aseer region from 2000-2007. (Test statistics and associated p values for class variables are directed at the hypothesis that variation between all other levels relative to the last mentioned level equals zero) |
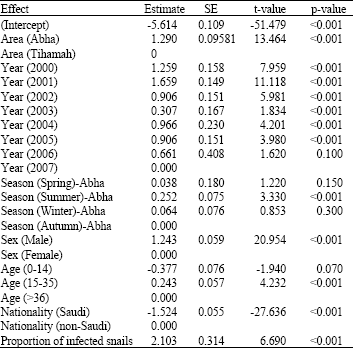 | |
Prevalence of schistosomiasis was found to be affected by host sex as males had significant higher infection rate than females (Table 1). In addition, There was a significant association between the host age and prevalence of schistosomiasis as the age group of 15-35 showed the highest infection rate (Table 1).
Prevalence of schistosomiasis was affected by nationality of the examined people as it was significantly higher in non-Saudi in comparison to Saudi people (Table 1). Prevalence of human schistosomiasis was positively correlated with the intermediate host (snail) infection rate (Table 1).
Although, Aseer region had the highest rate of human schistosomiasis in Saudi Arabia in the past few years, the results of the present study showed that there was a decline in the prevalence of infection in this region during the study period. This may due to the increase of applying schistosome control programmes run by the Saudi Ministry of Health as well as the increase of people awareness about schistosomiasis.
There was a spatial variation in the rate of schistosomiasis prevalence in Abha in comparison to that in Tihamah. This result is opposite to what could be expected as in Tihamah there are more rivers, dams and open shallow wells as well as good environmental conditions (e.g., warm weather and availability of water all around the year) for the intermediate hosts (snails) to breed and for schistosomes to be transmitted as indicated in other studies (Gryseels et al., 2006; Brooker, 2007). The conditions in Abha, however, are not suitable as in Tihamah for schistosome transmission especially in winter as the temperature drops to about less than ten degrees. Such low temperature has been found in other studies to affect schistosome transmission (Appleton, 1978; Martens, 1995; Gryseels et al., 2006; Brooker, 2007).
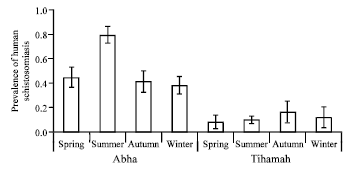 | |
| Fig. 2: | Seasonal prevalence of human schistosomiasis in the two study areas: Abha and Tihamah. Error bars give standard errors |
However, the difference found between infection rate in the two areas may due to the fact that more effort of schistosome control programmes has been put to Tihamah than in Abha. Another reason could be that Abha is a tourist area as the summer time has a good weather and the rate of rainfalls becomes higher making people contact with water increase. Thus, people coming from different areas and countries during the summer time may have brought infection with them to the area at that season.
This study showed that many abiotic and biotic factors have contributed to the high prevalence of schistosomiasis recorded in Aseer region. Seasonality was one of the abiotic factor found to affect the prevalence of human schistosomiasis as it was found to peak in the summer in Abha but not in Tihamah. This may due to the fact that the rate of rainfalls becomes higher in this season and people contact with water also increase. Another potential explanation is that Abha is a tourist area and people coming from different areas and countries during the summer time may have brought infection with them to the area at that season. This is in line with other studies which have reported that prevalence of schistosomiasis usually peak in wet seasons where the contact with water increase (Spear et al., 2004; Scott et al., 2003).
Sex was one of the biotic factors found to affect the prevalence of schistosomiasis in Aseer region as the rate of infection in males was higher than it was in females. The male-biased infection found in this study is in line with many other studies (Talaat et al., 1999; Khoby et al., 2000; Hammam et al., 2000; Brooker, 2007). Sex differences in parasite infection rates are usually attributed to one of two causes: (1) ecological (sociological in humans) and (2) physiological, usually hormonal in origin. Examples of the first cause include differential exposure to parasites because of sex-specific behaviour or morphology (e.g., water contact in the case of schistosomiasis). The second cause may stem from the well-documented association between testosterone and the immune system; sexually mature male vertebrates are often more susceptible to infection and carry higher parasite burdens (Zuk and McKean, 1996; Klein, 2004). However, the obtained results in this study may due to cultural and behavioural practices, as males being associated more with working in farms, dealing with animals at the water ranks and do outdoor swimming where they can contact the intermediate hosts of schistosomes. On the other hand, contact of females with water is less as they mainly concern meal preparation and other duties in houses.
Another biotic factor found to affect schistosomiasis prevalence in Aseer region was host age as it was highest in 15-35 age-group. This may due to behavioural and professional factors (Gryseels et al., 2006) such as being most of people in this age usually are farmers and have more contact with water in their daily practices.
The rate of human schistosomiasis was higher among non-Saudi people in comparison to Saudi people. This may due to the fact that most of people are workers in farming and other activities require contact with water are non-Saudis.
Rate of intermediate host infection with schistosomes was one of the biotic factors affecting the prevalence of human schistosomiasis in this study. The observed positive correlation between the infection of the intermediate snail host and human infection with schistosomiasis is in line with other studies (Scott et al., 1982; Clennon et al., 2004; Okpala et al., 2004; Brooker, 2007). Such correlation reflects the fact that the transmission of schistosomiasis takes place only in the places where snail vector is present and where there is contact between people and infested water (Okpala et al., 2004).
This study showed that many abiotic and biotic factors have contributed to human schistosomiasis in Aseer region. There also was a spatial variation in the prevalence of schistosomiasis in this region as it was higher Abha than it was in Tihamah. Therefore, more attention should be paid to Abha area in the future schistosome control programmes beside the intensive control programmes being applied to Tihamah.