Research Article
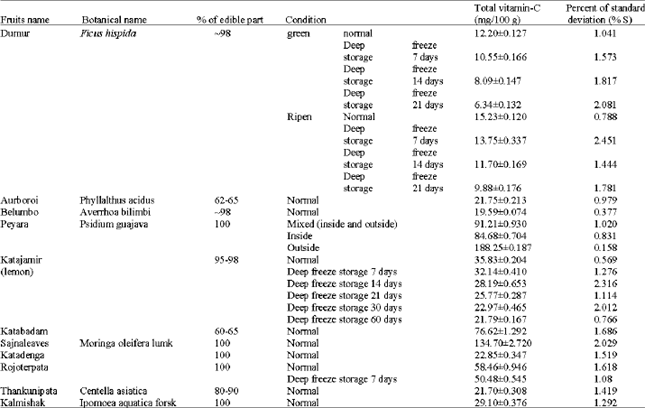
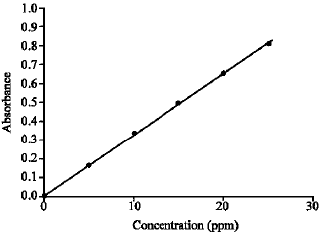
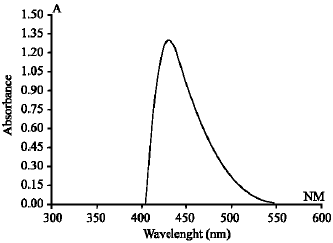
A Simple UV-spectrophotometric Method for the Determination of Vitamin C Content in Various Fruits and Vegetables at Sylhet Area in Bangladesh
Department of Chemistry, Shah Jalal University of Science and Technology, Sylhet-3114, Bangladesh
M.M. Rahman
Department of Chemistry, Shah Jalal University of Science and Technology, Sylhet-3114, Bangladesh
M.S. Islam
Department of Chemistry, Shah Jalal University of Science and Technology, Sylhet-3114, Bangladesh
S.A. Begum
Department of Chemistry, Shah Jalal University of Science and Technology, Sylhet-3114, Bangladesh