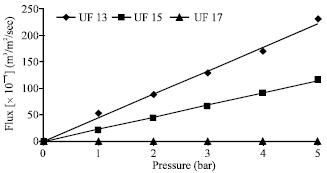
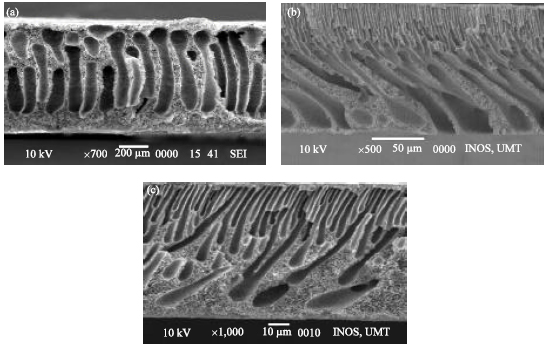
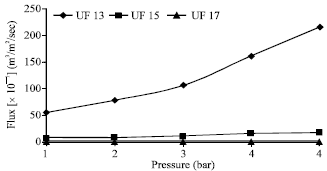
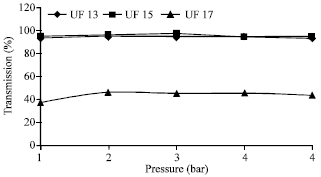
Research Article
The Influence of Polymer Concentration on Performance and Morphology of Asymmetric Ultrafiltration Membrane for Lysozyme Separation
Department of Engineering Science, Faculty of Science and Technology, University Malaysia Terengganu, 21030 Kuala Terengganu, Malaysia
A. Nora`aini
Department of Engineering Science, Faculty of Science and Technology, University Malaysia Terengganu, 21030 Kuala Terengganu, Malaysia
M.A. Marinah
Department of Chemical Sciences, Faculty of Science and Technology, University Malaysia Terengganu, 21030 Kuala Terengganu, Malaysia