Research Article
Evaluation of Catalytic Cracking Reactivity of Zeolites using 1-Dodecene as a Model Feedstock-Classification of Zeolites Based on Hydrogen Transfer Reactivity
Department of Chemical Engineering, Kagoshima University, 1-21-40 Korimoto, Kagoshima, 890-0065, Japan
Y. Ueda
Department of Chemical Engineering, Kagoshima University, 1-21-40 Korimoto, Kagoshima, 890-0065, Japan
K. Ijichi
Department of Chemical Engineering, Kagoshima University, 1-21-40 Korimoto, Kagoshima, 890-0065, Japan
K. Mizuta
Department of Chemical Engineering, Kagoshima University, 1-21-40 Korimoto, Kagoshima, 890-0065, Japan
Y. Uemura
Universiti Teknologi Petronas, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia









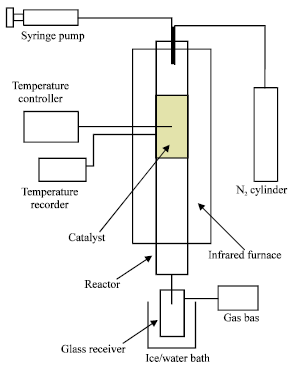
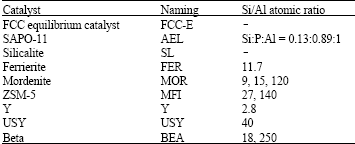
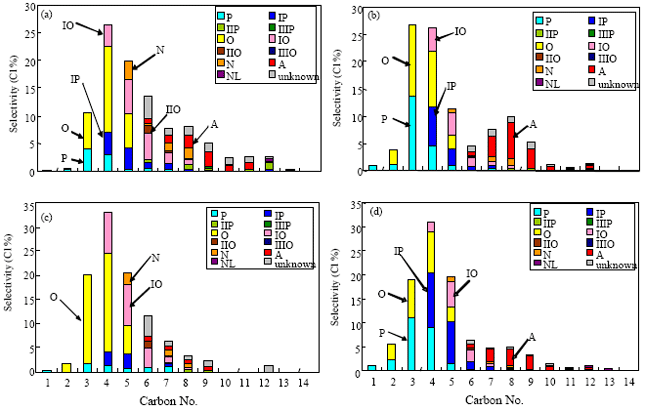
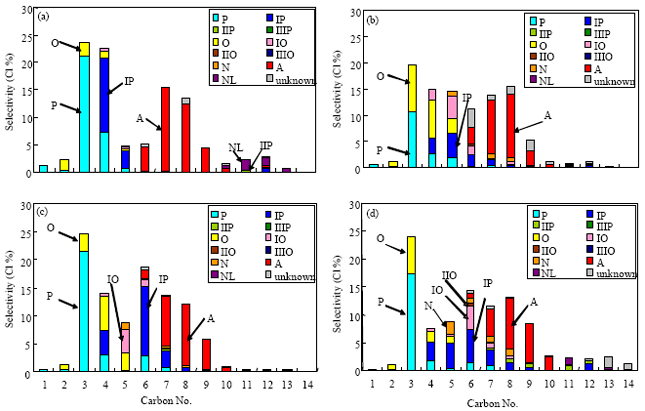
mahmoud abdelaziz Reply
thank you
mahmoud abdelaziz Reply
thanks