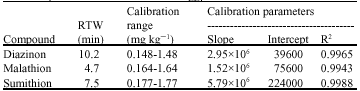
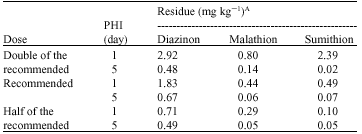
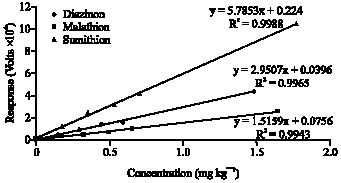
Research Article
Application of High Performance Liquid Chromatography to the Analysis of Pesticide Residues in Eggplants
Laboratory of Biological and Environmental Systems, Faculty of Agriculture, Kochi University, Nankokushi, Monobe B200, Kochi 783-8502, Japan
M.S. Hossain
Department of Chemistry, University of Dhaka, Dhaka-1000, Bangladesh
N. Nahar
Department of Chemistry, University of Dhaka, Dhaka-1000, Bangladesh
M. Mosihuzzaman
Department of Chemistry, University of Dhaka, Dhaka-1000, Bangladesh
M.I.R. Mamun
Department of Chemistry, University of Dhaka, Dhaka-1000, Bangladesh