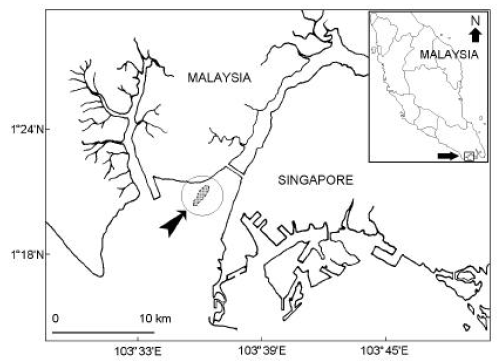
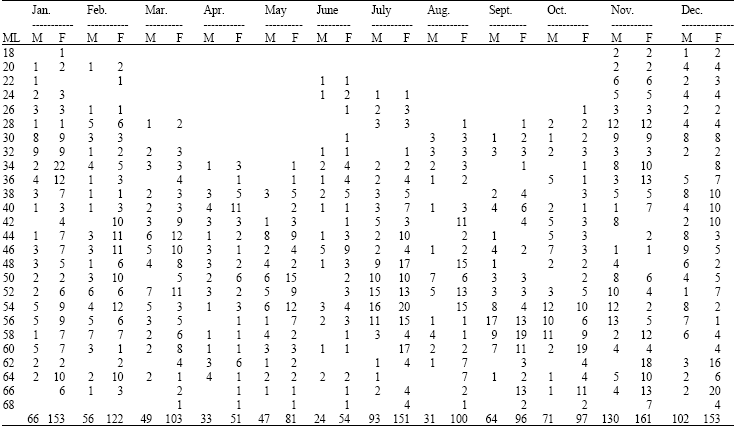
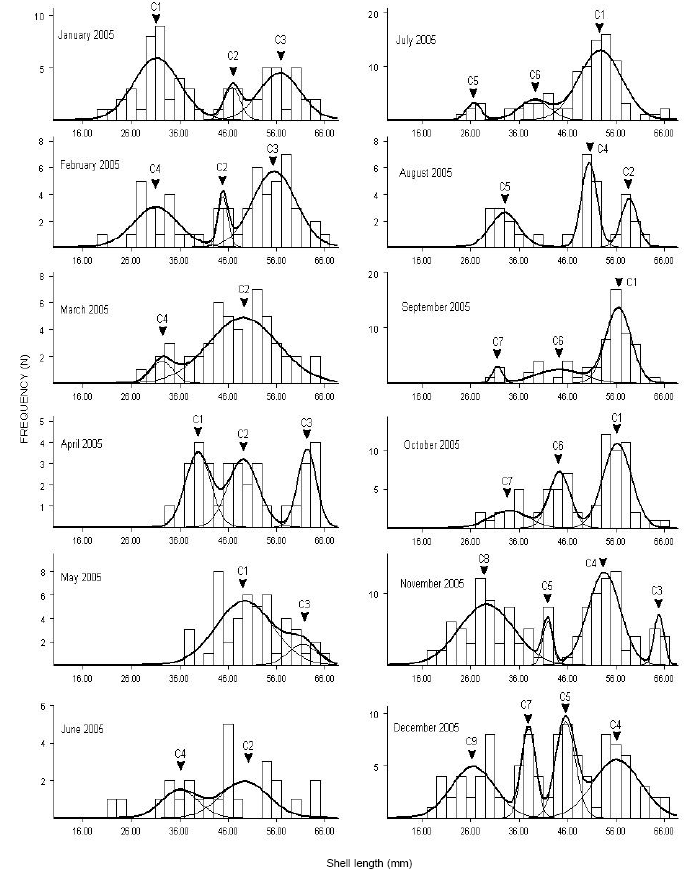
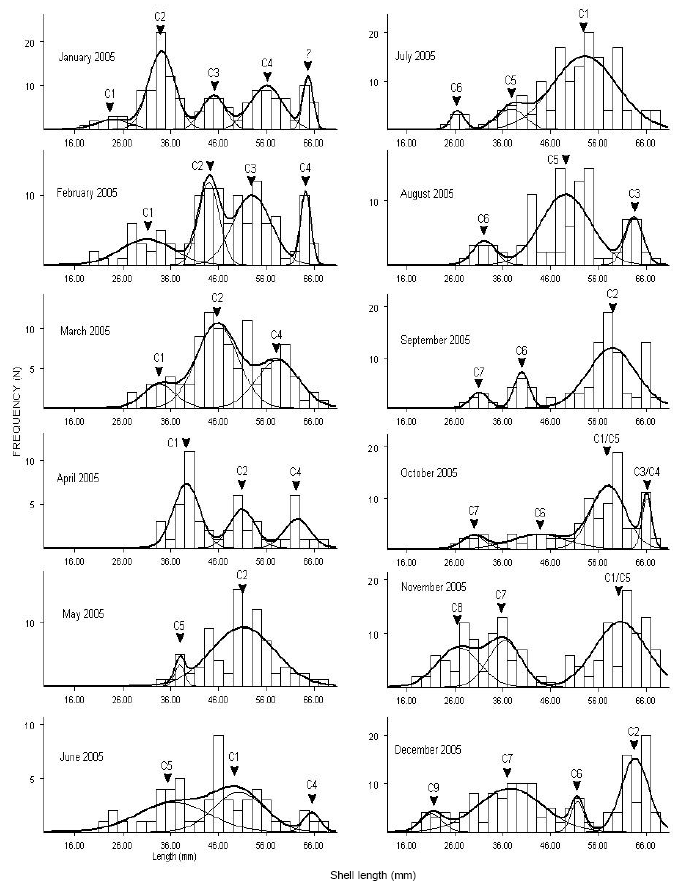
Research Article
Age, Growth, Mortality and Population Structure of Strombus canarium (Gastropoda: Strombidae): Variations in Male and Female Sub-Populations
Marine Ecosystem Research Center, Faculty of Science and Technology, National University of Malaysia, 43600 Bangi, Selangor, Malaysia
A. Arshad
Department of Biology, Faculty of Science, University Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
J.S. Bujang
Department of Biology, Faculty of Science, University Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
M.A. Ghaffar
Marine Ecosystem Research Center, Faculty of Science and Technology, National University of Malaysia, 43600 Bangi, Selangor, Malaysia