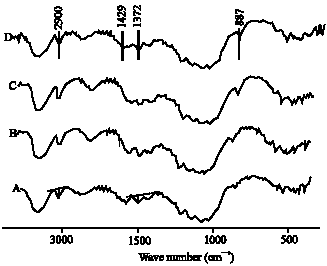
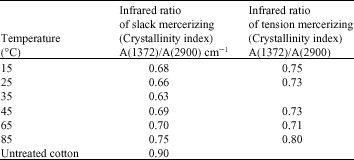
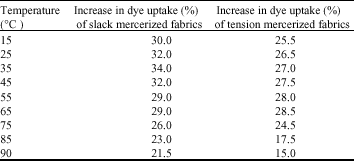
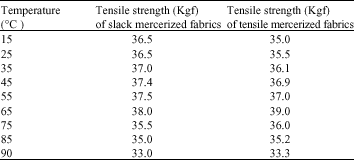
Research Article
An Investigation on the Effect of Hot Mercerization on Cotton Fabrics Made up of Open-End Yarns
Department of Textile Engineering, Faculty of Technical and Engineering, Islamic Azad University, Science and Research Branch, Tehran, Iran
S.M. Mortazavi
Department of Textile, Isfahan University of Technology, Isfahan, Iran
A.S. Rashidi
Department of Textile Engineering, Faculty of Technical and Engineering, Islamic Azad University, Science and Research Branch, Tehran, Iran
S. Sheikhzadah-Najar
Department of Textile Engineering, Faculty of Technical and Engineering, Islamic Azad University, Science and Research Branch, Tehran, Iran