Research Article
Application of Multivariate Analysis in Understanding Anions in Soils Close to an Abandoned Manganese Oxide Ore Mine
Geology, Mining and Minerals Programs, University of Limpopo, P/B 1106 Sovenga, 0727 South Africa
Manganese minerals, bixbyte, Mn2O3; braunite, Mn+2 Mn6+3 SiO12; ramsdellite, MnO2; pyrolusite, MnO2 and cryptomelane, K2-xMn8O16 (Vink and Ekosse, 2002) have been mined at Kgwakgwe, Southeastern Botswana from 1957 (Aldiss et al., 1989; Lanzincka, 1992), to the last two decades when the project was abandoned (Ekosse and Fouche, 2005a, b). These minerals identified in the Mn oxides ore were also present in the soils at Kgwakgwe. It is thus suspected that the mining and related anthropogenic activities carried out at Kgwakgwe may have affected the soil chemistry of the environment. Due to pressing demands, such as rapid population growth and increasing call for expansion of agricultural activities, on justifiable land use, there is need to understand the status of soil nutrients as well as cations and anions in the Kgwakgwe soil.
Anions of significance in soils include Cl¯, SO42¯ and CO32¯. These anions influence the bioavailability of soil nutrients and plant growth. Anions induce deleterious effects on the physico-chemical and chemical properties of soils; thereby affecting plant growth. High levels of Cl¯ affect flowering and fruiting of plants and causes shoot and root injuries (Environment Canada, 2006). Chlorine is involved in photosynthesis and its deficiency causes paleness, wilting, bronzing and abscission of leaves (Nutrient Properties, 2006). High concentrations of soil SO42¯ increase soil acidity which could lead to decrease of vegetation cover and possibly development of wasted lands devoid of vegetation. Its deficiency also causes leaves and fruits to remain immature lacking succulence (Environment Canada, 2006; Nutrient Properties, 2006). It is therefore imperative to understand the relationship of concentrations of these anions and their possible environmental implications.
By separate studies, concentration levels and spatial distribution of the soil nutrients (Ekosse and Fouche, 2007) and the spatial distribution of anions (Ekosse, 2007) in the soils have been established. Unfortunately the work on anions did not bear on their statistical relationship. Multivariate analyses have not been used in depicting relationship of concentrations among anions particularly those of Cl¯, SO42¯ and CO32¯ in soils close to an abandoned Mn oxide mine in Botswana. This work therefore investigated the concentrations and the three anions in Kgwakgwe soils and attempted to establish possible relationships based on multivariate analyses. Findings of this study could aid in understanding anionic behaviour of soils associated with similar geologic terrains.
Study area and soil sampling: Kgwakgwe (latitudes 24°59' E and 25°02' E and longitudes 25°17' S and 25°20' S) is 4 km south of Kanye township (Fig. 1), Southeastern Botswana. The Kgwakgwe soils are ferric luvisols (LVf) and calcic luvisols (LVk) (FAO, 1998; World Reference Base, 2006), associated with lowland areas having ephemeral streams.
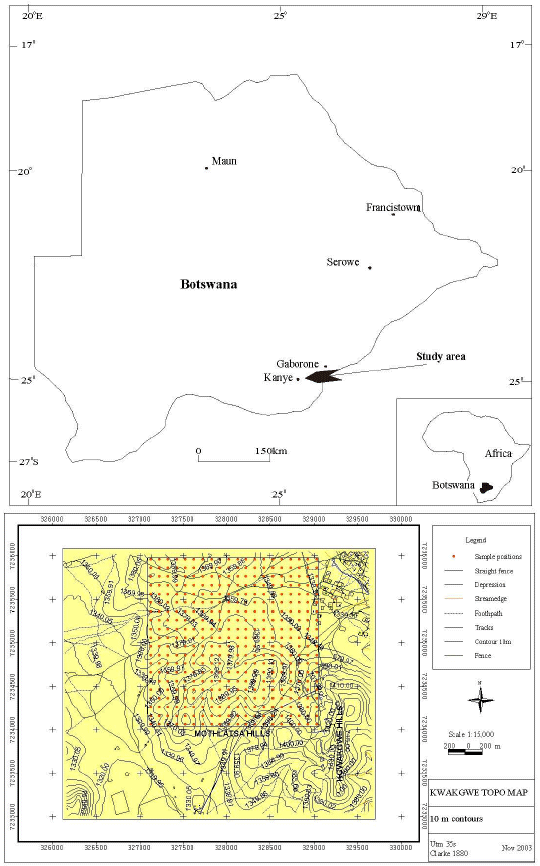 | |
| Fig. 1: | Map of Botswana showing location of the study area (a) and ten metre contour map of the study area with grid revealing the sampling points (b) (Ekosse and Fouche, 2007) |
Soil thickness varies with changes in topographic elevations and the underlying rocks which are granite and rhyolite of the Basement Complex and dolerite from doleritic rocks intrusions. The soils are characterised as moderate, moderate-well to well drained, sandy loam to sandy clay soils (Mitchell, 1976; Reemelzwaal, 1988). Texturally, the soils are dominantly silt loam, but silt and sandy loam textural classes are also present in substantial quantities, as well as trace quantities in the classes of loam, loamy sand and clay (Ekosse and Fouche, 2006). Kgwakgwe soils consist of 11 soil colours classified according to Munsell Soil Colour Book (1992): yellowish red (5YR4/6), red (2.5YR4/6), dark red (2.5YR3/6), brown (7.5YR4/4), dark reddish brown (5YR3/3), strong brown (7.5YR4/6), dark reddish grey (5YR4/2), very dark grey (5YR3/1) and light brown (7.5YR6/4).
Two sampling areas, which included a control site located 4 km south of the study area were chosen. The study site was 4 km2 (2x2 km) and the control site had an area of 900 m2 (30x30 m). This control site was at the other side of a paleotopographic barrier devoid of Mn mineralisation (Ekosse and Vink, 1998); although it shares similar soil lithology and vegetation cover with those of Kgwakgwe area. Grid techniques were used for sampling. The soil samples were taken at 100 m intervals and at a depth of between 0 cm and 20 cm. A total of 400 samples were collected from the study site (Fig. 3) and nine samples from the control site for analyses. The obtained samples were placed in an oven at 60°C overnight for the release of surface soil moisture, prior to analyses.
Laboratory analyses: Laboratory analyses were performed for the measurement of concentrations of Cl, SO4 and CO3 in the Kgwakgwe soil samples. The Metrohm 761 Compact IC Ion Chromatograph was used to determine the Cl and SO4 present in the soil samples. The procedure used has been adapted from Harris (1999), Jackson (2000), Park et al. (2002) and Metrohm News (2004). Peak response was recorded using a mobile phase eluent of Na2CO3/NaHCO3 with the Metrosep A SUPP 5 analytical column and calculation automatically done with integration software IC Net 2.1 against a previously prepared calibration plot.
The Eijkelkamp calcimeter designed to handle five samples at a time was used to determine the CO32¯ content in the soil samples. All the samples were analysed in similar environmental conditions including temperature. The calcimeter method applied was that described in Eijkelkamp (2003), for determination of the amount of carbonate in soil. Calibration was carried out prior to measurements using the anhydrous Na2CO3. A calibration curve was constructed and calculations were done as follows:
| (1) |
| Vs | = | Volume of CO2 produced by soil |
| W | = | Weight of soil |
| Vc | = | Calibration volume of Na2CO3 (calculated as mL CO2 produced by 1 g of Na2CO3) |
Multivariate analyses: Data analyses were carried out using Statistical Package for Social Sciences (SPSS) software. The descriptive statistics gave a summary of Cl¯, SO42¯ and CO32¯ concentrations. Cross tabulation of concentrations of the anions was used to show their relationships of distribution. Regression analysis, analyses of variance (ANOVA) and Pearson’s correlation matrix (Miller and Miller, 2000) were generated to test for the association among Cl¯, SO42¯ and CO32¯. Two-step cluster analysis was used to reveal clusters among the various anions. Plots were created for each variable and t-test of significance was used as a measure of importance at 95% confidence levels. Clustered error bar charts were further generated which summarised the distribution of individual variables within clusters defined by a categorical variable. The error bars represented 95% confidence intervals for mean. The ‘by variable importance’ Bonferroni adjusted charts were generated in order to show the most important variables within each cluster. Hierarchical cluster was used to separate anions into clusters joining all of them in a complete classification tree.
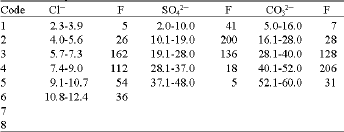
Concentrations of anions: Concentrations of the anions are tabulated to reflect coded frequencies of classes (Table 1) which were used for MVA and the distribution curves for the analysed parameters are given in Fig. 2. Concentration of Cl¯ in the soil samples from the study area ranged from 0.2 to 11.9 mg kg-1, with a mean of 7.63 mg kg-1 and for the soil samples from the control site, they were from 6.3 to 8.3 mg kg-1. Concentration of SO42¯ ranged from 2.1 to 47.5 mg kg-1, with a mean of 19.36 mg kg-1 for soil samples from the study area and 12.3 to 19.4 mg kg¯ for samples from the control site. Concentration of CO32¯ (measured in terms of CaCO3 equivalent) in the soil samples from the study area, were from 5.1 to 59.1 g kg-1, with a mean of 40.98 g kg-1 and for samples from the control site values obtained were from 22.9 to 34.1 g kg-1.
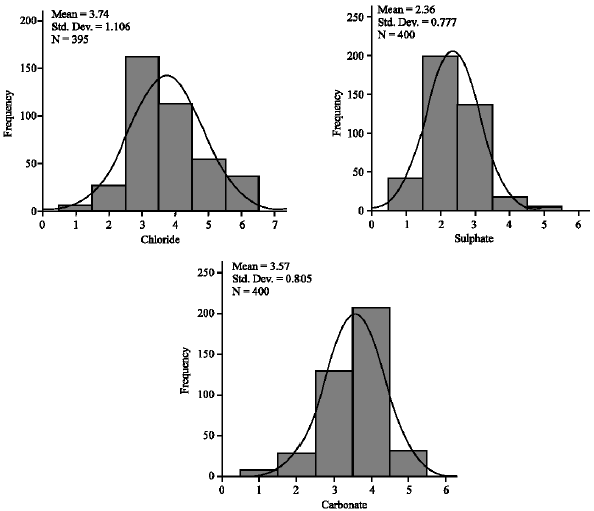 | |
| Fig. 2: | Histogram of (a) Cl¯, (b) SO42¯ and (c) CO32¯ concentrations in soil samples using codes |
| Table 1: | Frequency distribution table of Cl¯, SO42¯ and CO32¯ in soil samples based on coded classes |
 | |
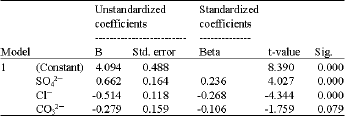
Inferential statistics and multivariate analysis: Figure 3 presents the linear regression trends of the data reflected by the straight line and R Square, the linear coefficient of determination (the squared value of multiple correlation coefficients). In Fig. 3a, 26% and in Fig. 3b 23% of the variation in SO42¯ can be explained by the linear regression model. The two independent variables (Cl¯ and CO32¯) indicated weak relationships with the dependent variable (SO42¯). The regression equations (Cl¯ = 1.971 + 0.751 SO42¯ and CO32¯ = 2.401 + 0.49 SO42¯) derived from the statistics was used to predict SO4 concentration in soil samples based on Mn concentrations from same samples (Ekosse and Fouche, 2005a). The equation derived is Mn in soils = 4.094+0662 SO42¯-0.514 Cl¯-0.279 CO32¯. The t-statistic indicated the relative importance of each variable in the model (Table 2). Sulphate emerged as very good predictor of Mn because its t-statistics was <2. Chloride and CO32¯ had least relative importance in the model, respectively and therefore were not very good predictors.
The sum of squares output for regression displays information about the variation accounted for and that for residual addresses variation not accounted for (Table 3). The value of significance is <0.05 indicating that the independent variables (Cl¯, SO42¯ and CO32¯) are predictors of the Mn concentrations in the soils. Product moment correlation coefficients among concentrations of anions in the soil samples are shown in Table 4. In general, the correlation coefficients depicted positive associations Cl¯/SO42¯ and Cl¯/CO32¯.
Table 5 and Fig. 4 give the cluster profile of Cl¯, SO42¯ and CO32¯ concentrations in soil samples and Fig. 6 reflects their 95% simultaneous confidence intervals for their means. For the anions, coded values (1-6) which are occurring in the clusters are based on real concentration values shown in Table 1. In cluster 1, the mean coded value for Cl¯ was 4.67 indicating that 9 mg kg-1 Cl¯ was contained in the samples.
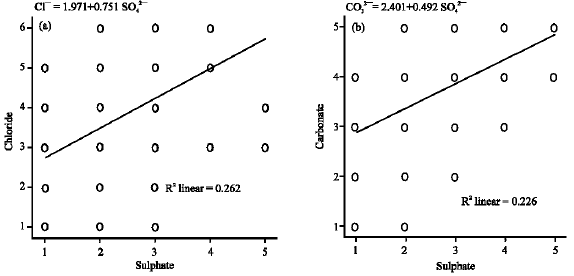 | |
| Fig. 3: | Scatter plots of Cl¯, SO42¯ and CO32¯ in soil samples with a regression fit line |
| Table 2: | Unstandardised coefficients and t-statistics of the linear regression line |
 | |
| a: Dependent variable: Manganese in soils | |
| Table 3: | Analysis of variance (ANOVA) derived from linear regression |
 | |
| a: Predictors: (Constant), Cl¯, SO42¯ and CO32¯; b Dependent Variable: manganese in soils | |
| Table 4: | Pearson correlation test for chloride, sulphate and carbonate concentrations in soil samples |
 | |
| **: Correlation is significant at the 0.01 level (2-tailed) | |
| Table 5: | Centroids of cluster profiles of chloride, sulphate and carbonate concentrations in soil samples |
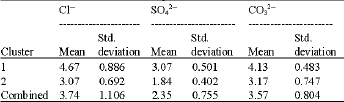 | |
Mean coded value for SO42¯ was 3.07 reflecting 28 mg kg-1 SO42¯ contained in the samples and that for CO32¯ was 4.13 indicating that 52 mg kg-1 CO32¯ was constituted in the samples. Cluster 1 therefore contained all three anions and their concentrations in the cluster were above the average concentrations of these anions in the study area. Based on the mean values obtained for cluster 2, the samples in this cluster on average contained very low concentrations of the three anions.
Bonferroni Adjustment by variable importance charts for two step cluster analysis for Cl¯, SO42¯ and CO32¯ concentrations in the soil samples were generated from the centroid tables (Fig. 5). The variables are lined up on the Y axis, in descending order of importance. Dashed vertical lines mark the critical values for determining the significance of each variable. For a variable to be considered significant, its t-statistic must exceed the dashed line in either a positive or negative direction. A negative t-statistic indicates that the variable generally takes smaller than average values within this cluster, while a positive t-statistic indicates the variable takes larger than average values.
Cluster 1 shows all three anions exceeding the critical value (Fig. 5a). Positive t-statistics for all the anions imply the variables take larger than average values. It is certain that Cl¯, SO42¯ and CO32¯ contribute to the formation of the first cluster and SO42¯ was the most important followed by CO32¯ and to a lesser extent Cl¯. For the formation of cluster 2, the order of importance for measures for all three anions were similar to that of cluster 1 and exceeded the critical value but were all negative t-statistic values (Fig. 5b).
Complementary results of MVA of the soil samples using complete linkage hierarchical clustering method and correlation coefficients as a measure of similarity is given in Fig. 6. The degree of association between groups of variables is represented by the distance axis. The lower the value on the axis, the more significant the association is carbonate and Cl¯ are very well coordinated, followed by Cl¯ and SO42¯. A strong separation exists between CO32¯, Cl¯ and SO42¯.
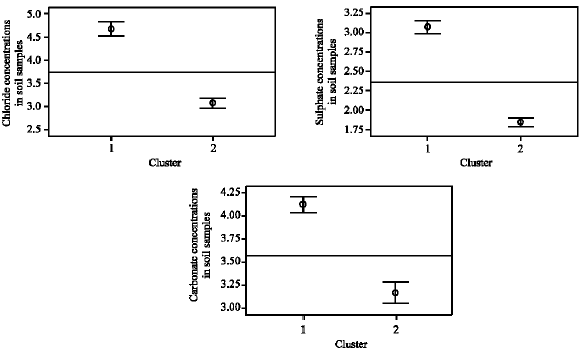 | |
| Fig. 4: | Simultaneous 95% confidence intervals for means and simple scatter plots of (a) Cl¯, (b) SO42¯ and (c) CO32¯ concentrations in soil samples |
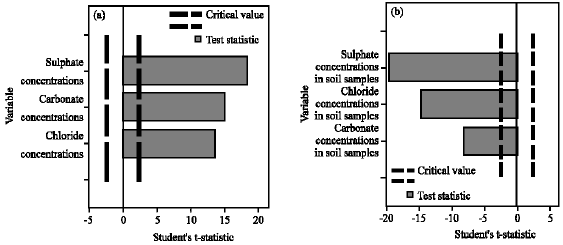 | |
| Fig. 5: | Bonferroni adjusted two step cluster for Cl¯, SO42¯ and CO32¯ concentrations in soil samples |
Environmental implications: The concentration of Cl¯ in soils in general is 0.10 g kg-1 (Flowers, 1988), its principal sources include the most abundant halite and sylvite and karnallite. Its concentration in soils could be increased through atmospheric deposition (Goos, 1987), although its concentration in the air decreases exponentially from coastal environments to those inland (Reynolds et al., 1997; Yaalon, 1963). These values are higher than those obtained for Kgwakgwe soils. Because of exposure of Kgwakgwe soils to anthropogenic activities, Cl¯ has easily been mobilized to different environments, ending up in nearby stream and groundwater.
The main sources of sulphate in Kgwakgwe soils are plant and animal remains and cattle litter and waterbodies within the surroundings. In agricultural soils, sulphur in SO42¯ form is predominant. Sulphate accumulation and its adsorption occurs more in sesquioxides than kaolinite rich soils (Johnson et al., 1979). Kgwakgwe soils are richer in kaolinite than sesquioxides (Ekosse, 2001; Ekosse and Fouche, 2006) and therefore due to its high mobility, sulphate accumulation is low.
Carbonate is the most abundant of the three anions, widely occurring in nature. Principal source of carbonate in the Kgwakgwe soils is the Taupone Dolomite (Ekosse, 2007). Another source of CO32¯ is soil CO2.
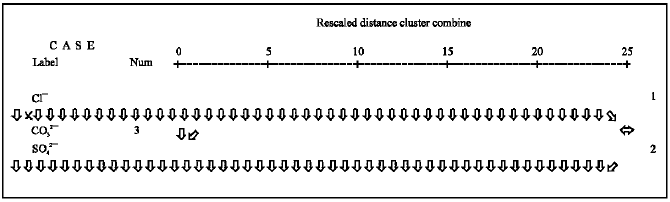 | |
| Fig. 6: | Dendrogram using complete linkage of Cl¯, SO42¯ and CO32¯ concentrations in soil samples |
The soil CO2 from plant root respiration and associated soil microorganisms could also react with soil H2O to produce CO32¯ (Berthelin, 1988; Karberg et al., 2005).
| (2) |
For cations to leach there must be associated anions of high mobility (Cole and Johnson, 1977; Johnson and Cole, 1977) such as Cl¯, SO42¯ and CO32¯ as well as HCO3¯. Cations of Mn Na, K, Ca, Mg and H are all involved at different stages and conditions with corresponding anions in different types of geochemical reactions such as hydrolysis and displacement/dislodgement in Kgwakgwe soils.
Investigations on concentrations of ClG, SO42G and CO32G in soils located close to an abandoned Mn oxide mine in Botswana were carried out in order to establish their statistical relationship and ascertain their spatial distribution using MVA, as well as associated geoenvironmental implications. Soils close to Mn mine workings and exposed outcrops had higher concentrations of the three anions compared to other soils. Strong correlations exist among the anions and SO42G proved to be very good predictor of Mn concentrations in these soils. The spatial distribution of SO42G is very similar to that of ClG and CO32G. The study further infers that MVA is an excellent tool in interpreting concentrations of anions in manganese rich soils.