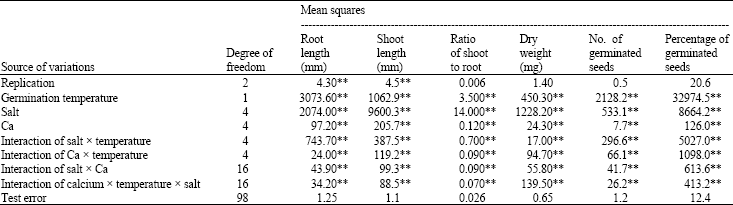
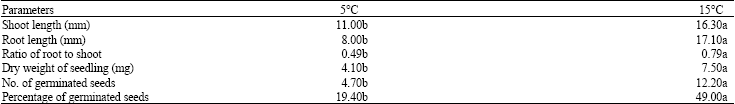
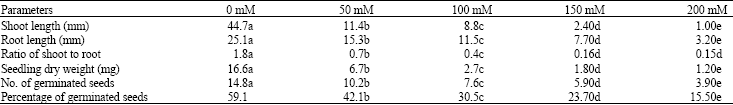
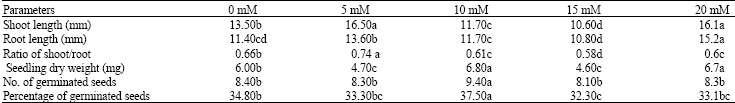
Research Article
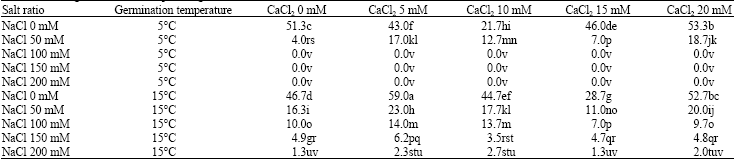
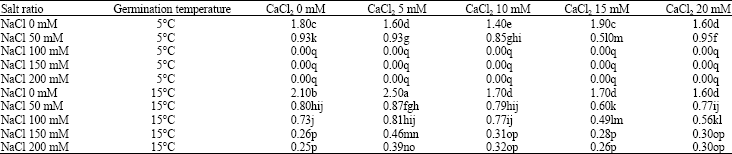
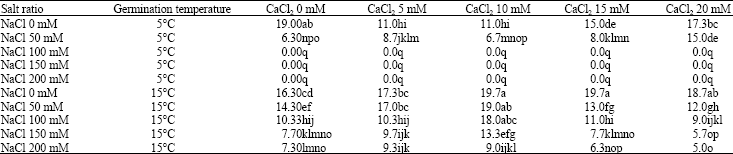
Effect of Exogenous Calcium Chloride on Sodium Chloride Salt and Cold Resistances During Seed Germination Stage of Medicago polymorpha
Department of Agronomy, Faculty of Agriculture, Shahed University, Iran
Shahram Jafari
Department of Computer, Faculty of Engineering, Shiraz University, Shiraz Iran
Reza Tabatabaie
Department of Agronomy, Faculty of Agriculture, Shahed University, Iran