ABSTRACT
The present investigation was under taken to determine the effect of cumulated Camcolit doses (lithium carbonate) in healthy Swiss albino mice concerning the function of testis, epididymis and vas deferens as well as hormonal function. In this study 40 adult male mice taken (23.25 mg mouse-1 - a cumulated dose for a period of 35 days), this dose is equivalent to the low human therapeutic dose and was injected intraperitoneally. Camcolit has no significant effect on mortality but there is significant decrease in both testes and body weight. Its administration induced histopathological changes included: disappearance of spermatogonia and decreased number of spermatocytes which lead to hypo spermatogenesis, hypertrophied nuclei and vacuolated cytoplasm were recorded in Sertoli cells, Intracellular and intercellular vacuoles were formed between germinal cells and Leydig cells have atrophied nuclei with vacuolated cytoplasm. Widening of the ductus epididymis and their cells became cubical with pyknotic nuclei and destruction or total loss of stereo cilia. Sperms disappeared from the vas deferens which suffered from disruption of muscular layers. Also low sperms number with formation of deformed ones with no head, coiled or bent tail. By using radioimmunoassay, it was found that Camcolit reduced serum testosterone level in lithium carbonate exposed male mice.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijzr.2008.85.95
URL: https://scialert.net/abstract/?doi=ijzr.2008.85.95
INTRODUCTION
It has been suggested that lithium can be considered for the prevention of Alzheimer`s disease in younger people with inherited forms of Alzheimer`s or Down`s syndrome (Phiel et al., 2003). Also Lithium salts are a mainstay and long term maintenance treatment in manic depressive disorders and abundant research supports their protective efficacy against acute recurrences (Kim et al., 2004). In addition to its present use in bipolar patients, lithium could be used to treat acute brain injuries such as stroke and chronic progressive neurodegenerative diseases (Chuang, 2004). There have been an increasing number of new generation antidepressant drugs developed to treat bipolar disorder. However, lithium salt is only the drug that is most efficient in long-term preventive treatment and it also has an anti-suicidal effect (Shastry, 2005).
However, the broad use of the element in treating mood and behavioral disorders has led to the realization of the fact that prolonged treatment with this alkali metal may lead to a wide rang of adverse effects (Bowden, 2000; Shaldubina et al., 2001).
Aizenberg et al. (1996) evaluated the sexual function and behavior in 35 male patients who were taking lithium (31.4%) reported sexual dysfunctions, so he suggested that lithium may have direct access at testicular level to inhibit steroid genesis. The relevant published literature at the past decade exhibits a reduction in testis weight of different animal species treated with lithium (Banerji et al., 1999).
Perez Romera et al. (2000) reported that the exposure of 1 mmol kg-1 body weight day-1 of lithium chloride for 35 days affected sperm motility and viability. This alkali element and its salt, as its higher doses, may lead to various side effects as lithium significantly reduced serum testosterone level in treated animals as compared to control group after prolonged therapeutic use and sub chronic supplementation of lithium carbonate induces reproductive system toxicity in male rat. Lithium might also cause an early onset on the end of the reproductive life of the mice (Thakur et al., 2003).
But less is known about the potential toxic effects of lithium therapy on the reproductive system especially on testicular activity which has not received much attention (Bowden, 2000).
MATERIALS AND METHODS
Zootechnics
Forty mature laboratories bred male Swiss mice of 8-12 weeks and weighing 25-30 g were used for the present investigation last summer. They were divided randomly and equally into two groups, control and experimental maintained under standard laboratory condition in special cages (14 h light: 10 h darkness) at 25±31°C with constant humidity 40-60% standard pellet diet and supplied daily by bottle for drinking water during the course of the entire experimental schedule.
The animals were allowed to acclimatize to this laboratory conditions by keeping them for 7 days prior to use for experimental purpose. The body weight was observed before and after the experiment.
The Chemical Used
Lithium Carbonate has an umpired formula of (Li2CO3) and molecular weight of 73.89 and it`s produced like tablet 250 mg by Norgin Company and it is available under the international trade (Camcolit). One tablet of the drug (250 mg) was ground down into powder and dissolved in 100 mL mammalian saline (0.9%) using intra-peritoneal injection (i.p.) at 9-11 h morning (0.3 mL animal-1) for 35 days.
In the present study, Paget and Barnes (1964) formula was applied to determine the equivalent low dose of Camcolit. At the termination of the respective experimental periods, all the animals at 10-11 h were sacrificed by decapitation 24 h after the last injection. Testes were excise from fat and connective tissue and weight were recorded in an electronic balance.
These studies comprise the following:
Histological Analysis
Light Macroscopically Examination
The testis with epididymis and vas deferens of both control and treated mice were immediately excised and cut into small pieces (about 5 mm) to allow good penetration of the fixative. Specimens of chosen organs were placed in Bouin`s fluid. The standard method of dehydration, clearing and paraffin embedding was followed according to Bancroft and Stevens (1977).
Sections stained with haematoxylin and eosin (H and E) (Pantin, 1964). All sections were carefully mounted, observed by the light microscope Nikon Edipse E400 ACT-2F.
Measurements of Seminiferous Tubular Diameter
About a total of 25 cross sections of round shape seminiferous tubules randomly selected from each testis of each mouse for measuring their diameter using a calibrated eye piece (x400) ocular micrometer in order to drew their mean value for individual mice.
Spermatogenetic Activity
The numbers of 1 ry spermatocytes were counted in transverse section of 50 randomly somniferous chosen tubules at each testis of a given mouse. The mean value of this stage in every seminiferous tubule was taken as the index of spermatogenetic activity.
Leydig Cell Nuclear Diameter
At least 25 Leydig cells from 5 to 7 section of testis of each mouse were considered for the measurement of nuclear diameter using a calibrated eye piece (x1000) ocular micrometer.
Measurements of Ductless Epididymis Diameter
About a total of 25 cross sections of round shape ductless epididymis randomly selected from each epididymis of each mouse for measuring their diameter using a calibrated eye piece (x400) ocular micrometer in order to drew their mean value for individual mice.
Sperm Morphology
To analyze sperm morphology, the epididymis was dissected out and was rinsed with 1 mL to 0.9% NaCl and sperm suspension was collected. An aliquot of sperm suspension was stained with 2% eosin to assess the percentage of morphological abnormal sperm. Two hundred sperm from at least five different areas on the slide were studied microscopically using an oil-immersed lens and sperm with abnormal heads and/or abnormal tails were scored as reported earlier (Saeed et al., 1996). The percentage of abnormal spermatozoa were calculated using the formula:
![]()
Evaluation of Sex Steroid Level (Testosterone)
Circulatory blood testosterone levels were also measured in each animal at the time of tissue collection. After 24 h of the last injection, animals were slightly anaesthetized and their eyes were bled within two minutes by using a heparinized syringe. Plasma samples were separated by centrifugation, frozen and stored at 851°C until all samples have been collected for the determination of testosterone. Plasma testosterone levels were measured by Radio Immunoassay (RIA) according to the methods of (Chang et al., 1995).
Statistical Analysis
Body weight, testicular weight, tubular diameters, Leydig cell nuclear diameter, sperm morphology, spermatogenic activity, ductless epididymis diameter and testosterone level were analyzed by using the program SAS (Institute Inc., 1988, SAS Cary, NC, USA), where a t-test was used to assess the significance of changes between control and treated mice (Sokal and Rohlf, 1981).
RESULTS AND DISCUSSION
During the experiment, all animals survived and this means that the doses administered, the laboratory conditions of water, food and shelter were appropriate.
Although no significant change in feeding behavior was noted in the Li-treatment mice as compared to that of the control animals, a significant (p<0.001) decrease in the body weight was noted following lithium treatment (30.45±1.43) in compare with control group (31.06±2.23) (Table 1). Chatterjee et al. (1990) showed that significant loss in body weight, coupled with depletion of subcutaneous fat observed in Li fed rats could be due to reduced food intake and/or an increase in metabolic rate.
On the other hand Moussa et al. (2001) observed that a daily intrapeitoneal injection of rabbits with lithium, for 4 weeks showed insignificant difference in body weight gain throughout the experimental period.
| Table 1: | Fluctuations of body weight, testes weight, seminiferous tubules diameter, primary spermatocytes count, Leydig cells nuclear diameter, epididymis ductules diameter, sperm count and testosterone level |
 | |
| Each value is given as the mean with standard deviations, *: Significantly different from control group at <0.05, **: Significantly different from control group at <0.0001, Each result corresponding to a mean value of 6 animals | |
Contrary Atmaca et al. (2002) showed that body weight increased mostly within the first two years of treatment and then leveled off so excessive body weight gain might be clarified by patients under long term Li-administration, as weight gain is a frequent effect associated with lithium use, as a significant difference in leptin levels which observed after lithium treatment.
In the present experiment, Li clearly brought about a number of adverse reproductive effects on the testicular morphology; this was evident from the significant reductions in testicular weight (0.16±0.016) in comparison with controls (0.24±0.027) (Table 1) as in toad (Nandi et al., 1994) and in bird (Banerji et al., 1999).
Moussa et al. (2001) reported that Li treated rabbits showed tendency of decreasing testis weight which was apparent after the 7th week of Li treatment.
Thakur et al. (2003) showed that the reduction of spermatozoa and spermatid in the seminiferous tubules most likely account for the effects on genital organ weight.
Histological Analysis of the Testes
The specimens of all animals of the control group were similar and revealed normal histological pattern of chosen specimens (Fig. 1A-C).
Further, the sections of the testes showed that in control animals the sperms remained in aggregated form near the Sertoli cells and also free form scattered in the tubular lumen (Fig. 1A). The Sertoli cells, which were always bordered by the limiting membrane of the seminiferous tubules, had large triangular nuclei (Fig. 1B), after treatment they became vacuolated (Fig. 1*B) as reported before (Thakur et al., 2003).
The number of Sertoli cells per testis is correlated with level of spermatogenesis and testicular weight in rats (Bemdtson, 1989). They have been shown to play several important functions in spermatogenesis among these functions are the support and the nutrition of the developing germ cells, compartmentalization of the seminiferous tubule by tight junctions to provide a protected and specialized environment for the developing germ cells, release of late spermatids into the tubular lumen, secretion of fluid, proteins and several growth factors, phagocytosis of the degenerating germ cells and phagocytosis of excess cytoplasm remaining from released sperm (Russell et al., 1993).
Defects of Sertoli cell result in loss of spermatic cells and may lead to the destruction of this tissue and infertility, according to Monsees et al. (2000) and Yano and Dolder (2002).
A greater loss of cells could lead to the degeneration of tubules, leaving open lacunae, as found in this study. Hence the reduced spermatogenesis potential of adult male rat could be due to Li-induced reduction in Sertoli cell proliferation (Thakur et al., 2003).
For instance, per tubular myoid cells (Fig. 2B) are functionally very important for the development of the spermatic process and together with Sertoli cells are responsible for the formation of the basal lamina (Dym, 1994).
The present histological study revealed marked alterations in the mice testes after the treatment with lithium; this included a general disruption in the organization of the various layers of differentiating germ cells from spermatogonial layer to the mature spermatozoa, with disruption of cellular association and a decreased number of germ cells (Fig. 2*B) with wide lumen as reported before in rabbit (Moussa et al., 2001). There was no constant point of arrest but a generalized decrease in spermatogenetic activity, i.e., hypospermatogenesis in comparison with controls animals.
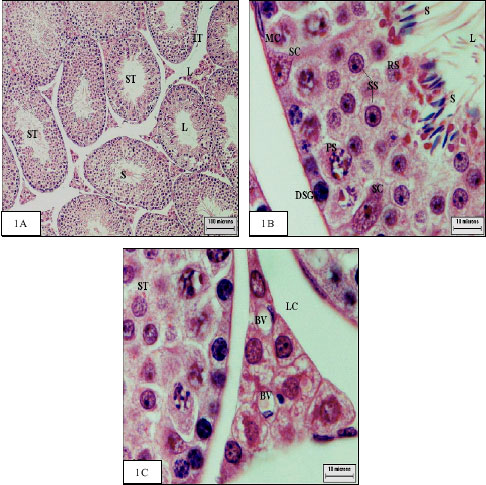 |
| Fig. 1: | Cross section of control testis: |
Other notable changes following lithium administration, there was intraepithelial vacuolization (Fig. 1*B, 1*C). But some tubules still had a relatively normal morphological appearance and large numbers of spermatozoa were found in their lumen.
The effect of lithium carbonate on testicular activity in this experiment provides a number of new observations, like a significant reduction in the numbers of 1 ry spermatocytes in the seminiferous epithelium cycle was observed after 35 days of lithium treatment (p<0.001) (35.40±10.73) when compared with those in controls (51.98±11.73) (Table 1), also they became small in size (Fig. 1*B).
The sperm numbers received from the caudal epididymis also revealed a significant reduction in Li-treated mice (84.20±25.53) in compare with control group (112.8±28.10) (Table 1). The reduction in epididymal sperm count also strongly supports the inhibition of spermatogenesis (Ghosh et al., 1991).
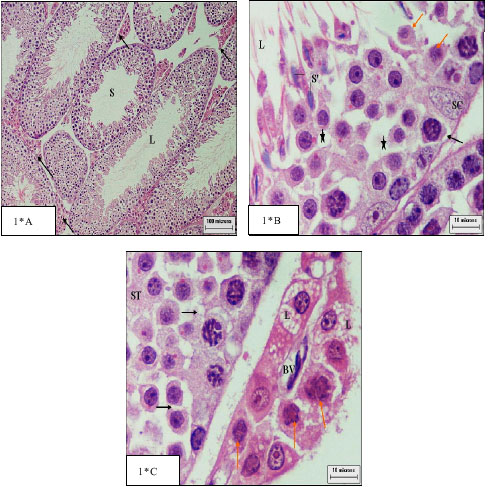 |
| Fig. 1*: | Cross section of treated testis: |
The abnormal-sperm percentage increased in mice after treatment with lithium (24.80±18.7) while in control one (5.20± 2.71) (Table 1) as reported before (US, 1986) who suggested that lithium has crossed the blood-testis barrier and gained access to germinal cells. The alterations observed consisted mainly of abnormal coiled, bend tail or detached head (Fig. 2*C).
Perez Romera et al. (2000) reported that the exposure per day of lithium chloride for 35 days affected sperm motility and viability. Hypogenesis and formation of abnormal spermatozoa may be induced as a consequence of lithium exposure because lithium significantly reduced serum testosterone levels in treated animals (Thakur et al., 2003).
In this study Li-treatment administration for 35 days resulted in a significant (p<0.001) increase in the diameter of seminiferous tubules (235±0.85) when compared to those of the saline-injected control animals (176.25±0.74) (Table 1). The increase in the diameter of seminiferous tubules with regard to the morphology of the germ cells as a majority of the seminiferous tubules showed a wide rang of degenerative changes as in Banerji et al. (1999).
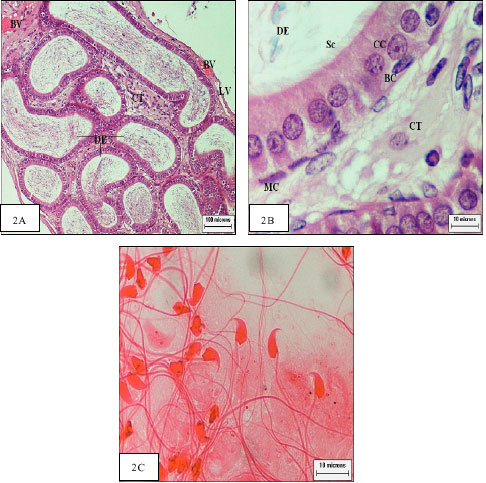 |
| Fig. 2: | Cross section of control epididymis: |
The control Leydig cells are small, inconspicuous cells with strongly basophilic cytoplasm near blood capillary (Fig. 1C).
The present data clearly established that Camcolit in the dose and for the time used in this experiment produced profound degenerative change in the Leydig cells. The over all morphology of Leydig cell and their nuclear diameters in the Li-treated animals (0.60±0.22) affected when compared to those of the control animals (0.82±0.11) (Table 1) and many of them revealed vacuolated cytoplasm near dilated blood capillary (Fig. 1*C).
Histological Analysis of the Epididymis
Jonson (1998) reported that the epididymis is essential for normal reproduction of mammals because sperm leaving the testis are not capable of fertilizing an oocyte. Together, the epididymal cells exert several important functions that are necessary for epididymal function: protein secretion and absorption (principal cells); endocytosis (clear cells); secretary activities responsible for acidification of the luminal fluid (clear cells); immune defense and phagocytosis and production of antioxidants (basal cells) (Hayatpour, 2000).
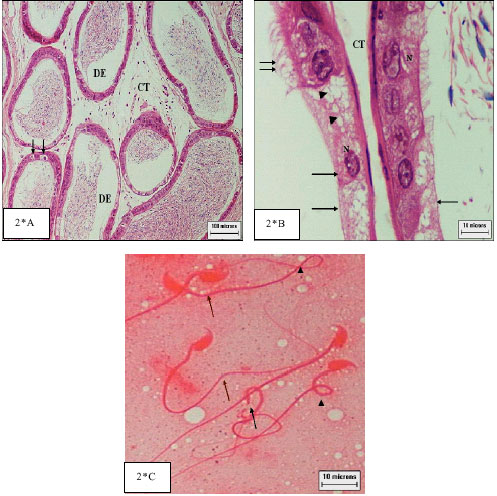 |
| Fig. 2*: | Cross section of treated epididymis: |
In this study, the atrophic in the epididymis may result from insufficient androgen which are remarkably similar regardless of species, as reported by Smithwick and Young (2001) who suggested that androgen is essential for maintenance of the normal epithelial architecture and cellular structure, which in turn are necessary for normal physiological functioning of the epididymis, including the processes of molecular synthesis and secretion that contribute regionally to successful sperm maturation.
The degree of cellular disorganization, cytoplasm atrophy and consequent reduction of epithelial height, loss of stereo cilia and alterations in size, shape and arrangement of the nuclei (Fig. 2*B) which observed in this study were essentially identical to those reported for other species (Korah et al., 2003; Goyal et al., 1994). Also interductular connective tissue was less dense (Fig. 2*A) in contrast to control (Fig. 2A). So, lithium has exerted its direct effects towered epididymis as recorded before in (Thakur et al., 2003).
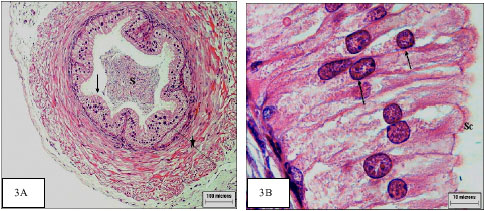 |
| Fig. 3: | Cross section of control vas deferens: |
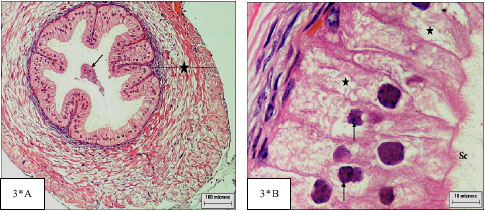 |
| Fig. 3*: | Cross section of treated vas deferens: |
Also Li- administration for 35 days resulted in a significant (p<0.001) increase in the diameter of ductus epididymis (168.7±0.28) when compared to those of the saline-injected control animals (93.75±0.28) (Table 1). Contrary Kim et al. (2004) explained the marked shrinkage in size of the epididymis by the reduction of testosterone concentration.
Decrease number of spermatozoa in epididymis in his study (Fig. 2*A) was agreeing with the quantitative studies of (Nandi et al., 1994) on toads and (Banerji et al., 1999) on bird concerning spermatogenesis.
Histological Analysis of the Vas Deferens
The lumen of control vas deferens contain sperm mass and lined with pseudo stratified columnar epithelium, surrounded by inner and outer longitudinal muscle layers in-between circular muscle layer (Fig. 3A).The pseudo stratified epithelium have oval nuclei and ciliary tuft (Fig. 3B). After treatment with Camcolit the vas deferens had cellular debris in the tubular lumen and disorganization of the muscular layers (Fig. 3*A). This observation may be a result of low level of testosterone and agrees with Fawcett (1986) who reported that testosterone influences the function of smooth muscle in the vas deferens. Also less height epithelial cells with intercytoplasmic vacuoles and hyperchromic nuclei were observed (Fig. 3*B).
Absence of spermatozoa in vas deferens in this study was agree with, the quantitative studies of Nandi et al. (1994) on toads and Banerji et al. (1999) on bird.
Evaluation of Sex Steroid Level (Testosterone)
In this study serum testosterone level measured to determine the possible adverse effect of lithium carbonate on steroidogenesis. Treatment with lithium carbonate resulted in a significant decrease of testosterone (0.13±0.010) in the interstitial cells compared to control values (0.90±0.010) (Table 1), which may be associated with significant decrease in the testicular weight.
With regard to laboratory animals, Li-administration has been shown to be associated with significant alterations in the pituitary testicular axis in the male, characterized by a significant suppression of circulatory testosterone levels and changes in pituitary gonadotropin levels (Russell et al., 1993).
The inhibition of spermatogenesis in our study and in Li-treated toad may be due to low levels of androgen in tissues (Nandi et al., 1994), where the growth of male germ cells is regulated by the level of androgen (Warner, 2003). And there was a decreased production of testosterone by Li-treatment (Kim et al., 2004).
Since Leydig cells are the only source of testosterone production in testis, it is possible that Li-treatment might have modulated Leydig cell function to reduce steroid production or promoted Leydig cell degeneration in treated animal (Thakur et al., 2003).
REFERENCES
- Aizenberg, D., M. Sigler, Z. Zemishlny and A. Weizman, 1996. Lithium and male sexual function in affective patients. Clin. Neuropharmacol., 19: 515-519.
Direct Link - Atmaca, M., M. Kuloglu, E. Tezcan and B. Ustundag, 2002. Weight gain and serum leptin levels in patients on lithium treatment. Neuropsychobiology, 46: 67-69.
Direct Link - Banerji, T., S.K. Maitra, A. Basu and H.K. Hawkins, 1999. Lithium-induced alterations in the testis of the male roseringed parakeet (Psittacula krameri): Evidence for significant structural changes and disruption in the spermatogenetic activity. Endocrine Res., 25: 35-49.
CrossRefDirect Link - Bowden, C.L., 2000. Efficacy of lithium in mania and maintenance therapy of bipolar disorder. Clin. Psychiatry, 61: 35-40.
Direct Link - Chang, C.F., E.L. Lau and B.Y. Lin, 1995. Estradiol-17beta suppresses testicular development and stimulates sex reversal in protandrous black porgy, Acanthopagrus schlegeli. Fish Physiol. Biochem., 14: 481-488.
CrossRef - Chatterjee, S., K. Roden and T.K. Banerji, 1990. Morphological changes in some endocrine organs in rats following chronic lithium treatment. Anat. Anz., 170: 31-37.
Direct Link - Chuang, D.M., 2004. Neuroprotective and neurotrophic actions of the mood stabilizer lithium: Can it be used to treat neurodegenerative diseases. Crit. Rev. Neurobiol., 16: 83-90.
Direct Link - Ghosh, D., N.H. Biswas and P.K. Ghosh, 1991. Studies on the effect of prolactin treatment on testicular steroidogenesis and gametogenesis in lithium-treated rats. Acta Endocrinol. (Copenh), 125: 313-318.
CrossRefDirect Link - Goyal, H.O., V. Hutto and M.A. Maloney, 1994. Effects of androgen deprivation in the goat epididymis. Acta Anatomica, 150: 127-135.
PubMedDirect Link - Johnson, G., 1998. Lithium-early development, toxicity and renal function. Neuropsychpharmacology, 19: 200-205.
CrossRefDirect Link - Kim, S.K., H.J. Lee, H. Yang, H. Kim and Y. Yoon, 2004. Prepubertal exposure to 4-tert-octylphenol induces apoptosis of testicular germ cells in adult rat. Arch. Androl., 50: 427-441.
Direct Link - Korah, N., C.E. Smith, A.D. Azzo, M. El-Alfy and L. Hermo, 2003. Increase in macrophages in the testis of cathepsin a deficient mice suggests an important role for these cells in the interstitial space of this tissue. Mol. Reprod. Dev., 64: 302-320.
Direct Link - Monsees, T.K., M. Franz, S. Gebhardt, U. Winterstein, W.B. Schill and J. Hayatpour, 2000. Sertoli cells as a target for reproductive hazards. Andrologia, 32: 239-246.
CrossRefDirect Link - Nandi, D.K., D. Ghosh, S. Parua and J. Debnath, 1994. Effect of lithium chloride on testicular delta 5-3 beta, 17 beta-hydroxy steroid dehydrogenase, acid phosphatase and gametogenesis in Bufo melanostictus. Ind. J. Exp. Biol., 32: 337-339.
Direct Link - Romera, E.P., E. Munoz, F. Mohamed, S. Dominguez and L. Scardapane et al., 2000. Lithium effect on testicular tissue and spermatozoa of viscacha (Lagostomus maximus). A comparative study with rats. J. Trace Elem. Med. Biol., 14: 81-83.
PubMedDirect Link - Phiel, C.J., C.A. Wilson and P.S. Klein, 2003. GSK-3α regulates the production of Alzheimer's disease amyloidal-B Peptides. Nature, 423: 435-439.
Direct Link - Russell, L.D., T.J. Corbin, K.E. Borg, L.R. Franca, P. Grasso and A. Bartke, 1993. Recombinant human follicle-stimulating hormone is capable of exerting a biological effect in the adult hypophysectomized rat by reducing the numbers of degenerating germ cells. Endocrinology, 133: 2062-2070.
CrossRefDirect Link - Shaldubina, A., G. Agam and R.H. Belmaker, 2001. The mechanism of lithium action: State of the art, ten year later. Prog Neuropsychopharmacol. Biol. Psychiatry, 25: 855-866.
Direct Link - Smithwick, E.B. and L.G. Young, 2001. Histological effects of androgen deprivation on the adult chimpanzee epididymis. Tissue Cell., 33: 450-461.
CrossRefPubMedDirect Link - Thakur, S.C., S.S. Thakur, S.K. Chaube and S.P. Singh, 2003. Subchronic supplementation of lithium carbonate induces reproductive system toxicity in male rat. Reprod. Toxicol., 17: 683-690.
Direct Link - Yano, C.L. and H. Dolder, 2002. Rat testicular structure and ultrastructure after paracetamol treatment. Contraception, 66: 463-467.
Direct Link









ramya Reply
sir, i want the dosage given by u per kg body weight .
u hav given 0.3ml/animal per day . i want the amount given to the animal / kg body weight /day