Research Article
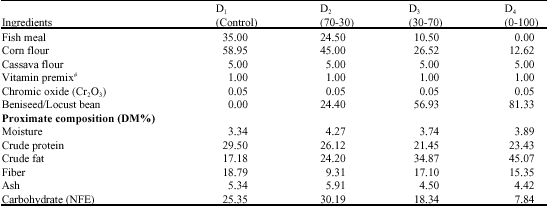
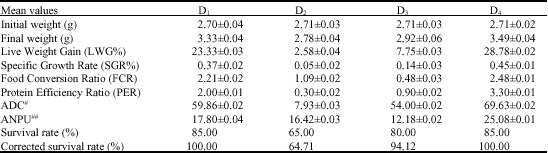
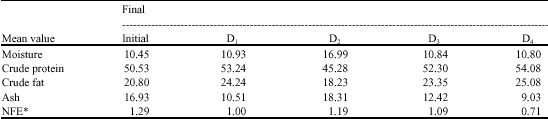
The Effect of Substituting Fishmeal Diets with Varying Quantities of Ensiled Parboiled Beniseed (Sesamum indicum) and Raw African Locust Bean (Parkia biglobosa) on the Growth Responses and Food Utilization of the Nile Tilapia Oreochromis niloticus
Department of Zoology, University of Jos, P.M.B. 2084, Jos, Nigeria
K.M. Adamu
Department of Zoology, Delta State University, P.M.B. 1, Abraka, Delta State, Nigeria
S.A. Binga
Department of Zoology, University of Jos, P.M.B. 2084, Jos, Nigeria