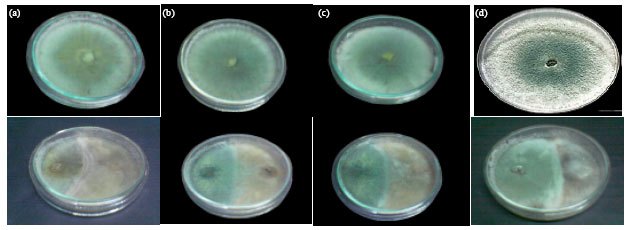
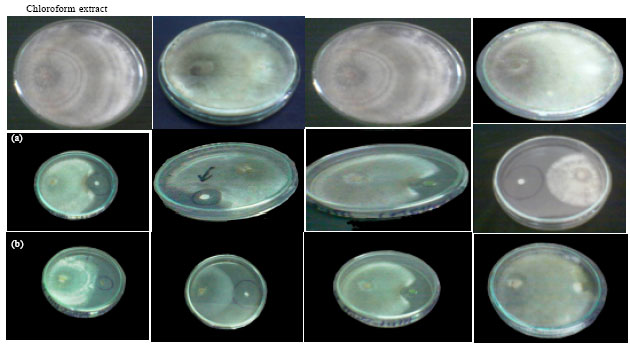
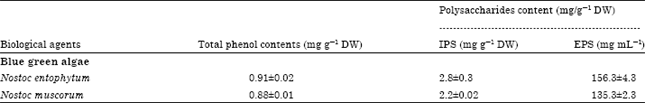
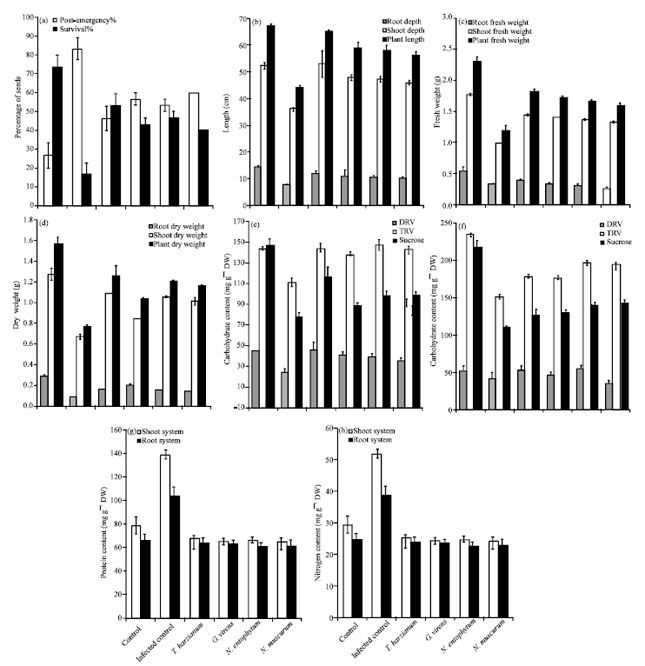
Research Article
Antagonistic Activity of Some Fungi and Cyanobacteria Species against Rhizoctonia solani
Department of Botany, Faculty of Sciences, Tanta University, Tanta 31527, Egypt
Mostafa M. El-Sheekh
Department of Botany, Faculty of Sciences, Tanta University, Tanta 31527, Egypt
Metwally A. Metwally
Department of Botany, Faculty of Sciences, Tanta University, Tanta 31527, Egypt
Abd El-whab A. Ismail
Plant Pathology Institute, Agriculture Research Center, Giza, Egypt
Mona M. Ismail
Department of Botany, Faculty of Sciences, Tanta University, Tanta 31527, Egypt