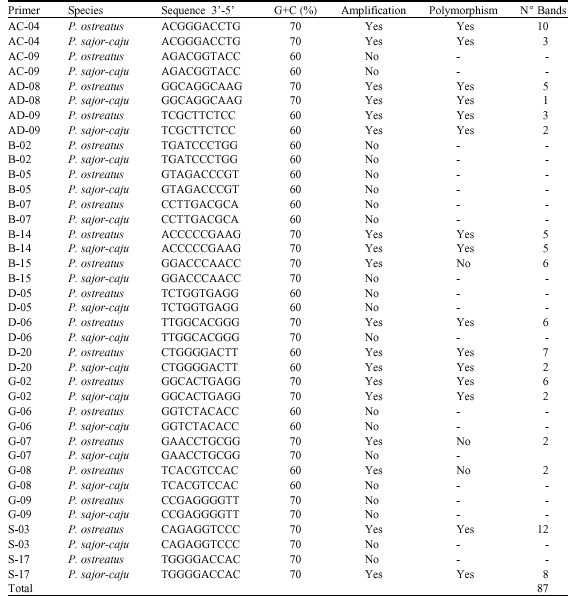
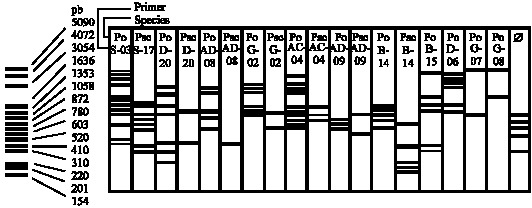
Research Article
Oyster Mushrooms Species Differentiation Through Molecular Markers RAPD
Laboratório de Engenharia BioquÃmica, Laboratório de BioquÃmica Marinha, Departamento de QuÃmica, Fundação Universidade Federal do Rio Grande, CEP 96201-900, Rio Grande, RS, Brasil
Eliezer Avila Gandra
Laboratório de Engenharia BioquÃmica, Laboratório de BioquÃmica Marinha, Departamento de QuÃmica, Fundação Universidade Federal do Rio Grande, CEP 96201-900, Rio Grande, RS, Brasil
Liana Fossati Sclowitz
Laboratório de Engenharia BioquÃmica, Laboratório de BioquÃmica Marinha, Departamento de QuÃmica, Fundação Universidade Federal do Rio Grande, CEP 96201-900, Rio Grande, RS, Brasil
Ana Paula Antunes Correa
Laboratório de Engenharia BioquÃmica, Laboratório de BioquÃmica Marinha, Departamento de QuÃmica, Fundação Universidade Federal do Rio Grande, CEP 96201-900, Rio Grande, RS, Brasil
Jorge Alberto Vieira Costa
Laboratório de Engenharia BioquÃmica, Laboratório de BioquÃmica Marinha, Departamento de QuÃmica, Fundação Universidade Federal do Rio Grande, CEP 96201-900, Rio Grande, RS, Brasil
Jose Alberto Levy
Laboratório de BioquÃmica Marinha, Departamento de QuÃmica, Fundação Universidade Federal do Rio Grande, CEP 96201-900, Rio Grande, RS, Brasil