Research Article
Piperine and Quercetin Enhances Antioxidant and Hepatoprotective effect of Curcumin in Paracetamol Induced Oxidative Stress
Department of Pharmacology, School of Pharmacy and Technology Management, NMIMS, Mumbai-400056, India
Ginpreet Kaur
Department of Pharmacology, School of Pharmacy and Technology Management, NMIMS, Mumbai-400056, India
Meena Chintamaneni
Department of Pharmacology, School of Pharmacy and Technology Management, NMIMS, Mumbai-400056, India









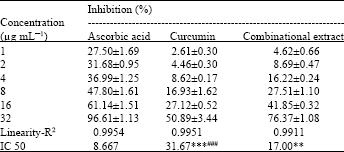

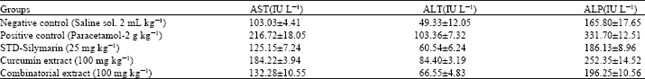
indra gunawan Reply
my research is interest to combine curcumine, piperine, and quercetine to increase curcumine bioavaibility with nanomolecule formulation. any advice from you, what the trick to combine that three compund or any others compund to increase the bioavaiblity of curcumine except piperine and quercetine ???