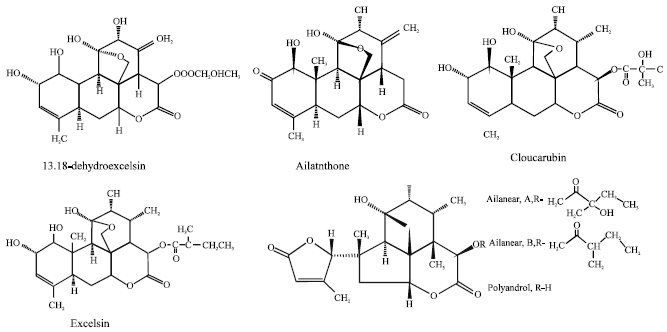
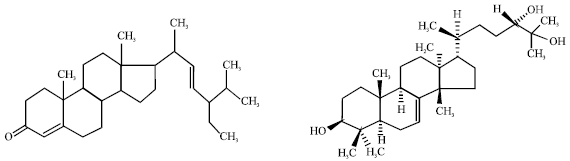
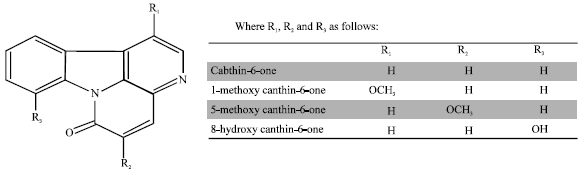
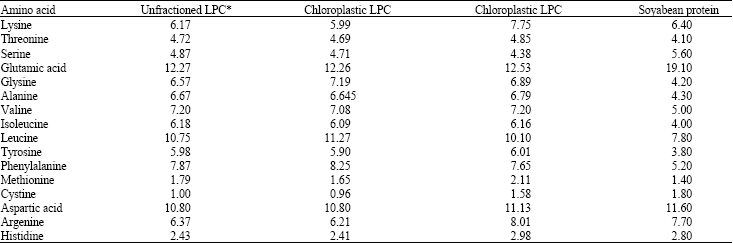
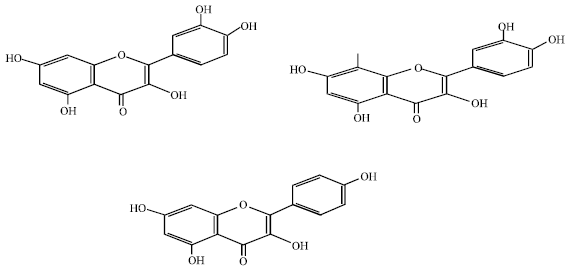
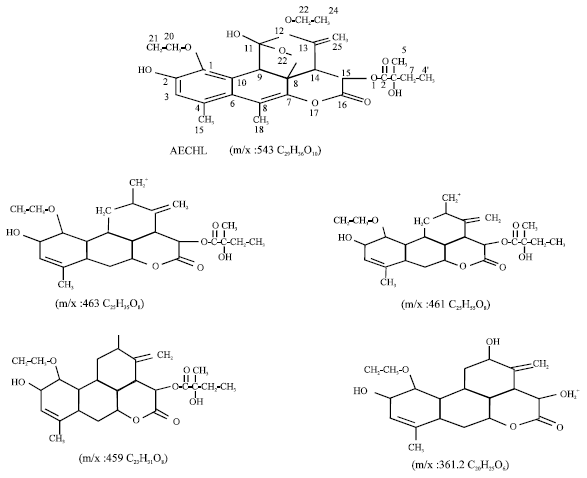
Review Article
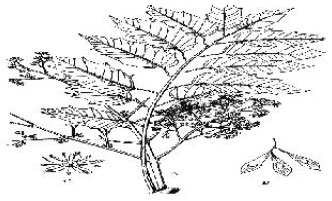
Ailanthus excelsa Roxb. is Really a Plant of Heaven
Department of Pharmaceutical Sciences, University of Kashmir, Jamu and Kashmir, 190006, India
Z. A. Bhat
Department of Pharmaceutical Sciences, University of Kashmir, Jamu and Kashmir, 190006, India
P. Singh
Department of Pharmaceutical Sciences, University of Kashmir, Jamu and Kashmir, 190006, India
M. Y. Shah
Department of Pharmaceutical Sciences, University of Kashmir, Jamu and Kashmir, 190006, India
S. S. Bhujbal
Pad. Shree Dr. D.Y. Patil Institute of Pharmaceutical Sciences and Research,Department of Pharmacognosy, Pimpri, Pune-411018, India