Research Article
Antimicrobial Activity of Essential Oil from Nelumbo nucifera Gaertn. Pollen
Biomedical Research Unit, Department of Chemistry, Faculty of Science, Mahasarakham University, Khamreung, Kantharawichai, Mahasarakham 44150, Thailand
The Nelumbo nucifera Gaertn. is an aquatic plant belonging to the family Nymphaceae (Gaertn.) which almost all parts are edible (La-ongsri et al., 2008). The leaves, root and the embryonic stage of the plant have reported to contain alkaloids such as such as roemerine, nuciferine, nornuciferine, nelumboside, anonaine, 5-methoxy-6-hydroxyaporphine, liensinine and asimilobine (Kulkarni and Juvekar, 2008). It has been reported to have anti-stress (Kulkarni and Juvekar, 2008), anti-obesity (Ono et al., 2006) and anti-oxidant (Rai et al., 2006), hepatoprotective (Sohn et al., 2003), anti-diabetic activity (Mukherjee et al., 1997a), anti-inflammatory (Mukherjee et al., 1997b), antipyretic (Mukherjee et al., 1996) activities.
The anti-malaria and antifungal activity of N. nucifera extract have also been reported (Agnihotri et al., 2008). The leaves have been indicated as part of the plant responsible for its anti-malarial activity (Li and Xu, 2008). In many Asian countries N. nucifera part was found in the recipe of food and folklore such as root and seed (La-ongsri et al., 2008). However, the N. nucifera pollen essential oil anti-microbial activity has never been reported. The present study deals with the antimicrobial activity of the essential oil extracted from N. nucifera.
Sample collection identification and extraction: The N. nucifera was collected on May 2008 from Ayuthaya Province, Thailand. The plant was identified by Department of Biology, Faculty of Science, Mahasarakham University, Thailand. Ten gram of dried flowers were hydro-distilled in 500 mL water and repeat 3 times. The yield of vapor distillation was 0.5-1% of dried weight of dried plant’s flowers. In this study used pooled of batches of the same condition of distillation throughout the studies.
Microbial cultures: Laboratory isolates of the pure culture of gram positive bacteria (Staphylococcus aureus ATCC 25923, Staphylococcus epidermidis ATCC 12228, Micrococcus luteus ATCC 9341, Bacillus subtillis ATCC 6633, Lactobacillus plantarum ATCC 14917) and gram negative bacteria (Escherichia coli ATCC25922, Salmonella typhimurium ATCC 14028, Klebsiella pneumonia ATCC 10031, Pseudomonas aeruginosa ATCC9721, Proteus vulgaris ATCC 13315) were obtained from the Department of Microbiology, Faculty of Pharmaceutical Sciences, Chulalongkorn University, Thailand.
Antimicrobial sensitivity test Agar disc diffusion test: The disc diffusion method used as described in the standard guideline technique (Lorian, 1996). All test bacteria were cultured overnight on Tryptic Soy Agar (TSA) slant at 37°C. Bacteria were washed from surface agar slant with sterile normal saline solution (0.9% NaCl) then adjusted to match turbidity of standard Mcfarland No. 0.5 before used as starter solution. Twenty milliliter of Mueller Hinton agar (MHA) was put in cultivation plates and leave until the agar become rigid. The starter solution was swabbed on agar surface by using swab cotton.
N. nucifera essential oil was dissolved in olive oil (1.25, 2.5 and 5% v/v) and put in sterile stainless steel cylinders (6 mm internal diameter and 10 mm height) were placed on the inoculated agar surface. The various concentrations of plant extract solution were filled in the cylinders (300 μL cylinder-1). After pre-diffusion at room temperature for 1 hour, the plates were incubated at 37 °C for 19 h. The NSS (normal saline solution) filled in the cylinder was used as control and 10 mg L-1 gentamicin sulphate (Sigma Chemical Co., St. Louis, USA) solution was used as standard in same cultivation plate.
MICs and MBCs determination using agar dilution and broth macro dilution methods: MICs of crude water extract of N. nucifera essential oil were determined by agar dilution method (Merck) (Lorian, 1996) while MBCs were determined by broth macro-dilution method were (Lorian, 1996). Gentamicin sulphate (Sigma Chemical Co., St. Louis, USA) was used as a reference antibiotic. Inoculates were prepared in the same medium at density adjusted to 0.5 McFarland turbidity standard (108 colony-forming units (cfu mL-1) and two fold dilution for the broth macro-dilution procedure. The inoculated tube were incubated at 37°C and the MICs were recorded after 24 h of incubation. The MIC was defined as the lowest concentration of ME or gentamicin sulphate at which the microorganism tested did not showed visible growth while MBC was defined as the minimum bactericidal concentration with negative subcultures on agar medium. Values were means of triplicate.
The control growth of food borne pathogen bacteria such as S. typhimurium, E. coli, S. aureus, Ps. aeruginosa and B. subtillis are common in the tropical countries especially in country where poultry products are produced (Van Dijk et al., 2007). The present study has investigated anti-microbial activity of N. nucifera which used in food and folklore. Furthermore, N. nucifera has almost every part is edible which also mean less toxicity. Therefore incorporation of N. nucifera in to food as food born protection consider less toxic. In this study anti-microbial activity of N. nucifera pollen essential oil has been test against various bacteria include food born pathogen bacteria.
The anti-malarial and antifungal activity of N. nucifera extract has been reported (Agnihotri et al., 2008). In earlier study indicated that the leaves of N. nucifera may responsible for antibacterial activity (Li and Xu, 2008). However, the pollen of N. nucifera has never been investigated even it used as the ingredient of food and some folkloric medicine.
In this study the essential oil of N. nucifera were tested against various bacteria to screen the possibility to use it as food preservation additive. The N. nucifera pollen essential oil showed anti-microbial activity against gram negative S. typhimurium ATCC14028 and E. coli ATCC 25922 (Table 1). The MICs are 10 and 40 and MBCs are 20 and 80 ml L-1 (Table 2). In conclusion N. nucifera pollen essential oil show inhibitory effect on growth of food born pathogen bacteria in low concentration which indicated the possibility of used as food preservation additive.
| Table 1: | Diameter of zone of inhibition of bacterial organisms of N. nucifera pollen essential oil against various bacteria |
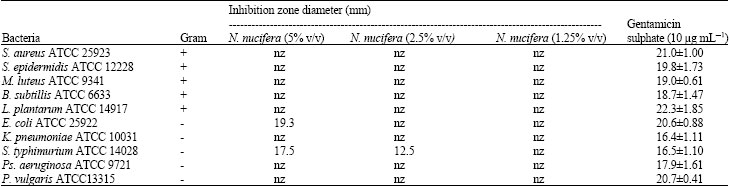 | |
| Data are mean±SD (n = 3); nz: No inhibition zone | |
| Table 2: | The MICs and MBCs of N. nucifera pollen essential oil against various bacteria |
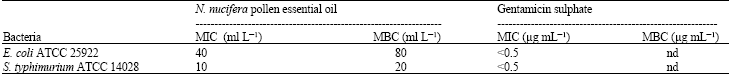 | |
| nd: Not Determine | |
Author would like to express the appreciation to partially support from faculty of Science, Mahasarakham University, Thailand.
Anu Kiruthika. K Reply
Sir goodevening
I am a research scholar in Kamaraj college, Thoothukudi,TN.
My project work is on flower extraction and its antimicrobial activity
so if you send me this full paper it will help me a lot in my project
Thank you sir
By
Anu