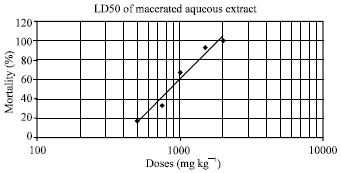
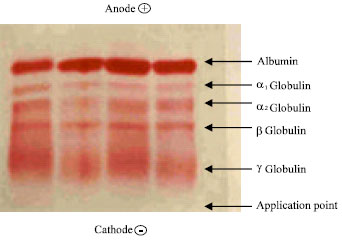
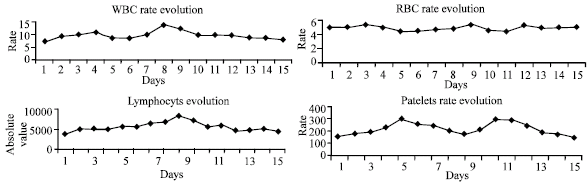
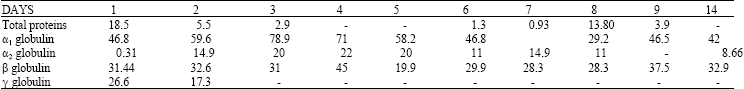Research Article
Biological and Toxicological Study of Aqueous Root Extract from Mitragyna inermis (Willd oktze) Rubiaceae
Xacoulma Institut de recherche en sciences de la Sante (IRSS/CNRS T): 03-BP 7192 Ouagadougou-03, Burkina Faso
Innocent Pierre Guissou
Laboratoire de Biochimie et Chimie Appliquee UFR/SVT Umversite de Ouagadougou (33 BP Ouagadougou 03, Burkina Faso Departement de Sciences Pharmaceutiques Appliquees/Pharmacologie-Toxicologie/Universite de Ouagadougou. BP 7021, Burkina Faso 4C H U Yalgado Ouedraogo -01 BP 7022 Ouagadougou 01, Burkina Faso
Odile Germaine Nacoulma
Laboratoire de Biochimie et Chimie Appliquee UFR/SVT Umversite de Ouagadougou (33 BP Ouagadougou 03, Burkina Faso