ABSTRACT
A perfusion reversed phase High Performance Liquid Chromatography (HPLC) method was carried out for the rapid separation and quantitative determination of immunoglobulins (Igs) from ultrafiltration retentate of sweet whey, using Hypersill column. The elution profile of whey Igs showed the presence of three classes of Igs namely, IgG, IgM and IgA. The highest Igs content was found for IgG, while the lowest was for IgA. Considerable differences were found in the classes and total Igs of different samples of UF retentate of sweet whey.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ijds.2007.268.274
URL: https://scialert.net/abstract/?doi=ijds.2007.268.274
INTRODUCTION
Whey contains a wide range of biologically active proteins, i.e., about 60 indigenous enzymes, vitamin-binding proteins, metal-binding proteins, immunoglobulins and various growth factors and hormones. The significance roles of these components have been reviewed (IDF, 1991).
Whey is being touted as a functional food with a number of health benefits. Whey proteins, namely i.e., lactoferrin, β-lactoglobulin, α-lactalbumin, glycomacropeptide and immunoglobulins, demonstrate a range of immune-enhancing properties. In addition, whey exhibited benefits in the arena of exercise performance and enhancement (Marshall, 2004).
Recently, the isolation of individual whey proteins has gained considerable attention with the advances in out of the knowledge to adapt whey proteins to dietetic requirements. Also, much attention has been focused on the minor whey proteins, as they are difficult to recover. Revealing the importance of these minor components has enhanced the demand to prepare there in highly purified form and in large quantity. Immunoglobulins and lactoferrin are the two minor whey protein components with proven antimicrobial activities (Reiter, 1978; Ekstrand, 1989).
Bovine colostrum is often used as the source of Igs as it contains 50 mg mL-1 of Igs whereas milk has only 0.6 mg mL-1. About 80% of Igs in milk or colostrum were of the IgG class (Jenness, 1988). Colostrum is not available in large quantities, which makes it unsuitable source for Igs. Milk or whey may be better sources of Igs since these are available in large volumes.
The separation and determination of biopolymers (peptides, proteins, etc.) are frequently achieved by reversed-phase high performance liquid chromatography, although other HPLC methods such as Ion-Exchange Chromatography (IEC), Hydrophobic Interaction Chromatography (HIC), Size-Exclusion Chromatography (SEC) and different types of Affinity Chromatography (AC) have also been used (Snyder, 1990; Chicz and Regnier, 1990; Strange et al., 1992; De Jong et al., 1993; Chen and Tusak, 1994; Leonil et al., 1995; Recio et al., 1995, 1996; Bobe et al., 1998a, b; Bordin et al., 2001). With the development of recombinant DNA techniques, HPLC has become an important tool in both quality control and process control in the production of recombinant proteins of pharmacological interest. This has contributed significantly to the development of reversed-phase high performance liquid chromatography methods that could be used to carry out very rapid analysis for proteins with minimum complexity in both instrumentation and operating conditions (Kirkland, 1993; Paliwal et al., 1993), non of the previous references gave optimum separation of Igs.
Therefore, in the present study, we present a method allowing the determination of immunoglobulins by using reversed phase high performance liquid chromatography for the separation and quantitative determination of the immunoglobulins from ultrafiltration retentate of sweet whey.
MATERIALS AND METHODS
Materials
Samples
Sweet whey was collected from Edam cheese manufacture from Arab Dairy Company (Kaha-Alexandria agriculture road), Egypt, during the period from October 2005 until January 2006. Whey transferred to the Dairy Laboratory, National Research Centre, Egypt, to prepare whey protein concentrate by using ultrafiltration (UF) technique and to analysis their gross compositions. Fat was removed from the whey by a cream separator and then ultrafiltered.
Preparations of Whey Protein Concentrate (WPC)
Ultrafiltration (UF) of cheese whey was carried out using a carbosep pilot plant (modules 151 UF system, Orelies, France), equipped with zirconium oxide membrane (Mol. cut off 50000 Daltons). UF was carried out in a batch mode at 45-50°C and pH 7, inlet and outlet pressure of 6 and 4 bars, respectively. UF was continued until to the concentration factor 20. WPC samples were packed in plastic bags and kept frozen at -20°C until use. Frozen WPC was thawed at 2-4°C overnight before use.
WPC samples were defatted by centrifugation at 4000 rpm for 30 min and then adjusting the pH to 4.6 using 1 N HCl solution and centrifuging at 6000 rpm for 15 min to remove precipitate cheese particles.
Methods of Analysis
Gross Composition
Whey and WPC samples were analysed for total solids by the drying oven method at 105°C for 3 h, fat and ash contents as described by AOAC (1990). Total nitrogen was determined by the Kejldahl method (Ling, 1963). Lactose content was colorimetrically determined as described by Barnett and Abd El-Tawab (1957). The average of three replicates was taken for each sample.
Precipitation of Immunoglobulins with Ammonium Sulfate
Saturated ammonium sulfate (SAS) was prepared by dissolving excess in distilled water until some crystals of ammonium sulfate remained undissolved. Working solution of 80% ammonium sulfate solutions was freshly prepared by diluting SAS with necessary amount of water. Equal volumes of the WPC and 40% SAS solution were mixed; the formed precipitate was removed by centrifuging, dissolved in the minimum quantity of distilled water and dialyzed until the complete removal of the ammonium salt (Nawar, 1999). The dialyzed extract was kept frozen at -20°C until analysed.
Separation of Immunoglobulins by High Performance Liquid Chromatography (HPLC)
Samples of the four dialyzed solution (Igs separated from whey) were collected and then fractionated by the complete chromatographic system (GBC HPLC England), equipped with LC 1150 pump and LC 1210 uv/vis detector at 280 nm and linear gradient from 0 to 80% in 20 min (1 mL min-1). The column used was REZ.XP.SAX (50x4.6 mm) anion exchange chromatography (Hyper sill ™). The manufacturer of the column Hypersill™ has recommended protocol used in this method.
The Igs samples were dissolved in mobile phase, filtered through 0.45 μm membrane filters and 10 μL of the diluted Igs were injected in the column and then eluted. The following eluents were used as mobile phase: (A): 0.2 M Triethanolamine pH 7.5 and (B): 0.5 M NaCl pH 7.5.
All peaks of standards and samples were detected by using the chromatographic system software Win Chrome Version 1.3. Immunoglobulins standard used (all supplied by Bethyl, Argentina) were IgG, IgM and IgA.
RESULTS AND DISCUSSION
Table 1 shows the mean chemical compositions of cheese whey, which were 7.70, 0.81 and 4.92% for TS, TP and lactose respectively. While, the mean chemical compositions of WPC were 16.17, 8.53 and 4.76% for TS, TP and lactose respectively. From the Table 1, it was noticed that the total protein content in WPC was higher than whey; in contrast, the lactose content was the lowest.
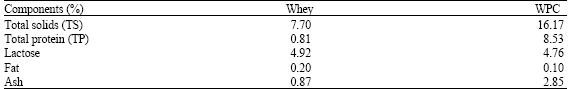
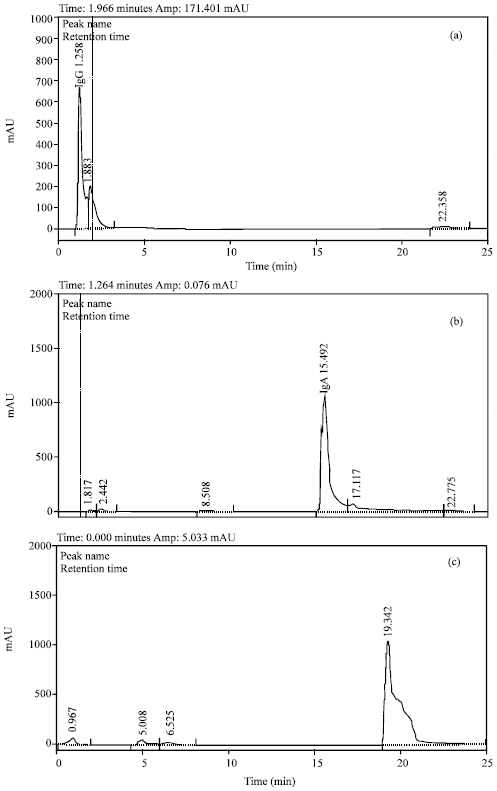
Attempts were made up to separate Igs from whey by reversed phase HPLC chromatography. The elution profiles of pure reference IgG, IgA and IgM are shown in Fig. 1. Peak identification was carried out using standard proteins, it can be deduced that the each of three Igs gave a single peak except IgG, which gave two peaks, the retention times of the different immunoglobulins, IgG (1.258 and 1.883), IgA (15.492) and IgM (19.342) minutes, were close indicating difficulty in getting sharp separation of their mixture. This can be explained on the basis of high molecular weight of these proteins and similarity of their nonnumeric units. The elution profiles of whey Igs are shown in Fig. 2, it is clear that the Igs were separated into four completely resolved peaks. The highest Igs values were observed in IgG content (2.5678 μg 10 μL-1), while the lowest was in IgA content (0.1502 μg μL-1) (Table 2). Considerable differences were found in the classes and total Igs of different samples of UF retentate of sweet whey.
Syvaoga and Korhonen (1993) studied the elution profiles of bovine colostrum, they found that all Igs, namely IgM, IgA and IgG, as well as the other whey proteins bovine serum albumin, β-lactoglobulin and α-lactalbumin were well separated in one run. The highest Igs values were observed in the first milking, after which they declined rapidly.
El-Loly and Farrag (2006) studied the concentrations of IgG, IgA and IgM measured by Single Radial Immunodiffusion (SRID) and Enzyme Linked Immunosorbent Assay (ELISA). Immunoglobulin contents of whey from the two techniques showed that no clear differences in IgG and IgM except for IgA, which the ELISA technique was very sensitivity and more accurate than SRID technique. These results attributed to low concentration of Igs in whey than milk or colostrum.
| Table 1: | Gross chemical composition of cheese whey and whey protein concentrate (WPC) |
 | |
| Mean of three replicates for each sample | |
| Table 2: | Concentration of immunoglobulins in extracted samples by HPLC |
 | |
 | |
| Fig. 1: | HPLC separation of Igs standards (IgG, IgA and IgM) on the Hypersill REZ.XP.SAX column |
While in Buffalo blood and milk, the results were different apparent in separated peaks compared to that of blood serum and colostrums, this can explained on the basis that during colostrums period Igs were largely originated from blood by selective filtration (El Loly, 1996).
 | |
| Fig. 2: | HPLC separation of four whey on the Hypersill REZ.XP.SAX column |
CONCLUSION
Reversed phase high performance liquid chromatography (HPLC) was carried out for detection and quantitative determination of the immunoglobulins (Igs) in whey. The method is rapid, easy, accurate analysis and able to separate and quantify the immunoglobulis from whey. The obtained data are important towards the further establishment of a reference method for immunoglobulins analysis.
REFERENCES
- Barnett, A.J.G. and G.A. Tawab, 1957. A rapid method for the determination of lactose in milk and cheese. J. Sci. Food Agric., 8: 437-441.
CrossRefDirect Link - Bobe, G., D.C. Beitz, A.E. Freeman and G.L. Lindberg, 1998. Separation and quantification of bovine milk proteins by reversed-phase high-performance liquid chromatography. J. Agric. Food Chem., 46: 458-463.
CrossRefDirect Link - Bobe, G., D.C. Beitz, A.E. Freeman and G.L. Lindberg, 1998. Sample preparation affects separation of whey proteins by reversed-phase high-performance liquid chromatography. J. Agric. Food Chem., 46: 1321-1325.
CrossRefDirect Link - Bordin, G., F.C. Raposo, B. de la Calle and A.R. Rodriguez, 2001. Identification and quantification of major bovine milk proteins by liquid chromatography. J. Chromatogr., A928: 63-76.
Direct Link - Chicz, R.M. and F.E. Regnier, 1990. High-performance liquid chromatography: Effective protein purification by various chromatographic modes. Methods Enzy., 182: 392-421.
CrossRef - De Jong, N., S. Visser and C. Olieman, 1993. Determination of milk proteins by capillary electrophoresis. J. Chromatogr., 652: 207-213.
PubMedDirect Link - Ekstrand, B., 1989. Antimicrobial factors in milk: A review. Food Biotechnol., 3: 105-126.
CrossRefDirect Link - El-Loly, M.M. and A.F. Farrag, 2006. Isolation of immunoglobulin-rich fractions from whey. Egypt. J. Dairy Sci., 34: 33-40.
Direct Link - Leonil, J., D. Molle, F. Gaucheron, P. Arpino, P. Guenot and J.L. Maubois, 1995. Heterogeneity of the bovine kappa-casein. Lait, 75: 193-210.
Direct Link - Marshall, K., 2004. Therapeutic applications of whey protein. Altern. Med. Rev., 9: 136-156.
PubMedDirect Link - Nawar, M.A., 1999. The optimal ammonium sulphate concentration for high recovery of immunoglobulins prepared from buffaloes-blood serum and colostrum. Alex. J. Agric. Res., 44: 151-159.
Direct Link - Paliwal, S.K., T.K. Nadler and F.E. Regnier, 1993. Rapid process monitoring in biotechnology. Trends Biotechnol., 11: 95-101.
PubMedDirect Link - Recio, I., E. Molina, M. Ramos and M. de Frutos, 1995. Proteins by capillary electrophoresis using uncoated capillaries. Electrophoresis, 16: 654-658.
CrossRefDirect Link - Recio, I., M. de Frutos, A. Olano and M. Ramos, 1996. Protein changes in stored ultra-high-temperature-treated milks studied by capillary electrophoresis and high-performance liquid chromatography. J. Agric. Food Chem., 44: 3955-3959.
CrossRefDirect Link









PRASHANTH S J Reply
Its very good article and I am using this method for analysis of Total Immunoglobulins from colostrum. please send me the some more HPLC Methods for Total Immunoglobulins analysis if you have for my research purpose.
Thanks & Regards
Prashanth S J
Research Scientist - Health Food Division
The Himalaya Drug Company
Bangalore, Karnataka