Research Article
Liver of the Snow Trout, Schizothorax curvifrons Heckel: A Histochemical Study
Department of Zoology, University of Kashmir, Srinagar-190006, India
A. Channa
Department of Zoology, University of Kashmir, Srinagar-190006, India
Fish liver is a dense organ ventrally located in the cranial region of the visceral cavity. Its shape size and volume are adapted to the space available between other visceral organs. In many species of teleostei the liver is bi or tri lobed, however no lobulation was recognized in some teleostei (Brusle and Gonzalez, 1996). The teleost liver responds very sensitively to environmental changes like, in temperature, seasons, feeding conditions or presence of various chemicals in the water (Storch and Juario, 1983). Attempts have been made to use this responsiveness as an indicator for aquatic pollution effects (Pierce et al., 1978, 1980; Haensly et al., 1982). Aquatic pollution is recognized globally as a potential threat to both humans and other animal populations which interact with the aquatic environments (Biney et al., 1987; Svensson et al., 1995). According to Jqrgensen et al. (1988), heavy metals are considered as the main cause of pollution in aquatic ecosystem and are expected to be so in the future, having the highest environmental stress index, often in excess of the recommended threshold limit values. The dangers involved from the presence of heavy metals in the environment derive not only from their persistence and toxicity, but also from the remarkable degree of bioaccumulation they undergo, thus becoming serious danger to humans (Hernandez-Hernandez et al., 1990; Bishop, 2000). Not only the heavy metals but also a myriad of other hazardous aquatic pollutants may accumulate in the fish particularly in the liver which is considered as the chief storage site. The accumulation of the pollutants may alter the concentrations of inorganic constituents, organic constituents and various enzymes in the liver of fishes.
The snow trout, Schizothorax curvifrons locally called as Satter Gad is a prized indigenous herbivorous cold freshwater teleost of Kashmir valley whose population is at decline due to multiple factors. The fish belonging to the family cyprinidae proves to be morphometrically, meristically and economically most variable and valuable promising food species of the paradise dale and can always be recognized by the combination of large high scale count, high gill raker number and thin lips (Mir and Channa, 2009, 2010). The liver is of utmost physiological importance as it is the chief site for storage of high energy foods (glycogen, lipids) which utilizes the fish at the time of emergency and it also plays a vital role in detoxification, digestion and other physiological processes. The present investigation has been undertaken for the first time as an effort to localize histochemically inorganic constituents, organic constituents and enzymes in an unaltered liver of a cold freshwater teleost, Schizothorax curvifrons with the intend that this study will provide a strong base line for future studies in relation to pollution, stress conditions in aquaculture, natural and polluted environments and pathobiology of teleost liver.
Living specimens of wild, normal and healthy snow trout, Schizothorax curvifrons (25-30 cm in length, body weight 300-350 g) collected from their natural habitat were dissected, the abdominal cavity was operated and the liver was excised quickly and cut into small pieces. The liver samples were then fixed in suitable fixatives for specified periods of time, depending up on the constituent to be studied. The fixatives used in the present study for the histochemical localization of inorganic and organic constituents along with the time of fixation at the room temperature are given in Table 1. After fixation the liver samples were processed as usual in the recognized method of dehydration, cleared in xylene and finally embedded in paraffin wax. Blocks were prepared by routine method and paraffin sections of 6-8 μ thick were cut from a rotary microtome and processed for the histochemical demonstration of various constituents. The various histochemical techniques employed during the present investigation were taken according to Pearse (1972a, b) and are given in the Table 1.
| Table 1: | Summary of the tissue fixation and techniques employed for localization of inorganic and organic constituents |
 | |
| Table 2: | Summary of incubation techniques for enzyme histochemistry |
 | |
Enzyme histochemistry: For enzymatic study the liver samples were fixed in cold (4°C) 10% neutral buffered formalin for a brief period of time. Frozen sections (10-15 μ thick) of the fixed liver sampler were cut at -25°C from a cryostat (Leica CM 3050S) and processed for histochemical detection of enzymatic activities. The histochemical techniques adopted for the localization of enzymes were taken according to Pearse (1972a, b). The overall summary of the enzymatic histochemistry is given in Table 2.
Photomicrography of the representative preparations was carried out under Olympus OIC microscope by using PM-6 Olympus camera. This study has been conducted from 2005 to 2007 in the Ichthyology laboratory of the Department of Zoology University of Kashmir, Srinagar, India.
The distribution and localization of inorganic constituents, organic constituents and enzymes have been carried out in the liver of Schizothorax curvifrons.
Inorganic constituents (iron and calcium): Intense reaction for iron is discernible in the cytoplasm of the hepatic cells (Fig. 1). The distribution is observed to be uniform throughout the perilobular and centrolobular regions of the liver. However, the sinusoids and the connective tissue react negatively to Prussian blue stain. The entire liver does not reveal any sign for the presence of calcium.
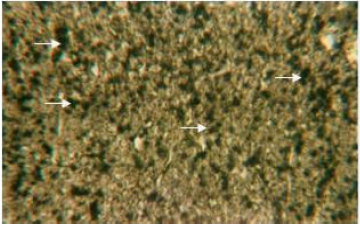
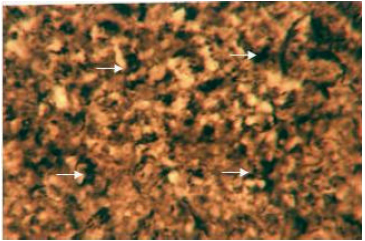
Organic constituents (proteins, DNA, glycogen and neutral lipids): The hepatocytes of the liver of Schizothorax curvifrons are characterized by high concentration of proteins as they stain intensely with mercury bromophenol blue stain (Fig. 2). The reaction of mercury bromophenol blue is mostly in the form of bluish granules which are peripheral or perinuclear in position and is adjacent to the sinusoids. Centrally placed nuclei of the polyhedral hepatocytes reveal well pronounced reaction and uniform distribution of DNA in the perilobular and centrolobular regions of the liver (Fig. 3). Intensely positive Best’s carmine glycogen granules are distributed more or less uniformly throughout the cytoplasm of hepatocytes, but at certain places of liver aggregated clumps of glycogen are noted to be present (Fig. 4), however the hepatic sinusoids are noted to be glycogen negative. Cytoplasm of the hepatocytes of the snow trout, Schizothorax curvifrons depict intense positive reaction for neutral lipids. The lipoid particles are uniformly distributed throughout the perilobular and centrolobular zones of the liver (Fig. 5). However, the nuclei of the hepatocytes and the sinusoids are totally sudanophobic.
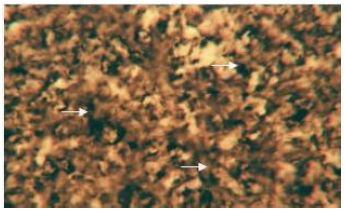
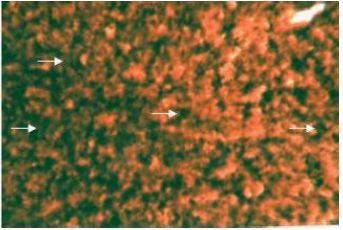
Enzymes (alkaline phosphatase, acid phosphatase, adenosine triphosphatase and lipase): The cytoplasm of the hepatocytes of the snow trout, Schizothorax curvifrons are found to be rich in their alkaline and acid phosphatase activities, giving them a granular appearance (Fig. 6, 7). The alkaline phosphatase activity is observed to be stronger in the perilobular zone than the centrolobular zone, on the other hand activity of acid phosphatase is noticeable along the cell membrane and around the nuclei of the hepatocytes, no enzyme activity is however, found in the sinusoids.
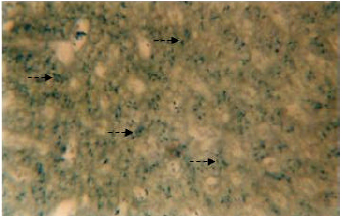 | |
| Fig. 1: | Transverse section of the liver stained with prussian blue stain. Broken arrow showing the presence if iron in the cytoplasm of hepatic cells. X 400 |
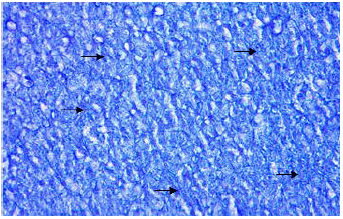 | |
| Fig. 2: | Liver section stained with mercury bromophenol blue showing the localization of proteins. X 400 |
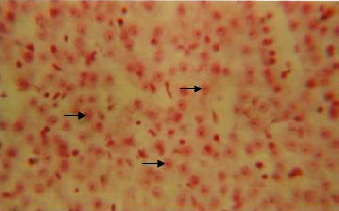 | |
| Fig. 3: | Liver section showing the presence of DNA in the centrally placed nuclei of hepatocytes. X 600 |
Adenosine triphosphatase activity (ATPase) is observed to be intense both in the cytoplasm and nuclei of polyhedral hepatic cells (Fig. 8).
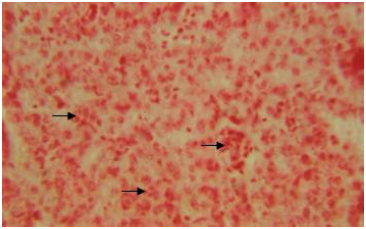 | |
| Fig. 4: | Liver section showing the distribution of Best’s carmine glycogen granules in the cytoplasm of hepatocytes. X 600 |
 | |
| Fig. 5: | Liver section stained with sudan black B. Solid arrow showing the localization of neutral lipids in the cytoplasm of hepatocytes. X 600 |
 | |
| Fig. 6: | Liver showing activity of alkaline phosphatase in the cytoplasm of hepatic cells. X 400 |
However, the enzyme activity is well pronounced along the perilobular region than the centrolobular region, the sinusoids do not show any sign for the presence of adenosine triphosphatase (ATPase). In the liver of the snow trout, Schizothorax curvifrons well pronounced lipase activity is discernible in the cytoplasm around the nuclei of the hepatocytes.
 | |
| Fig. 7: | Acid phosphatase activity in the cytoplasm of hepatocytes of liver. X 400 |
 | |
| Fig. 8: | Liver showing ATPase activity in the cytoplasm and nuclei of hepatic cells. X 400 |
 | |
| Fig. 9: | Liver showing intense lipase activity in the cytoplasm and nuclei of hepatic cells. X 400 |
The enzyme is however, more concentrated in the perilobular zone than the centrolobular zone of the liver (Fig. 9).
Iron in the tissues is mostly present in ferric form. The present study reveal the presence of iron in the cytoplasm of the hepatic cells of the snow trout, Schizothorax curvifrons after following the Perl’s prussian blue technique, as it has been considered as the method of choice for the localization of iron by Pearse (1972a, b). The occurrence of iron in the liver of the investigated fish is indicative of the fact that iron is stored in the liver and latter utilized for various physiological activities as has also been reported by Mitchell (1956). Iron as a component of haemoglobin, myoglobin and cytochromes plays a key role in oxygen transport and cellular oxidation. The absence of calcium from the liver may probably be due to the fact that as soon as the calcium is absorbed through the mucosa of the intestinal tract, it is either immediately transported through the blood to the active sites for immediate use or the amount of stored calcium if present in the liver is too small to be localized histochemically.
The hepatocytes of the snow trout, Schizothorax curvifrons possess high concentration of proteins, being perinuclear or peripheral in position and close to sinusoids. Abdelmeguid et al. (2002), has also reported the similar results in an unaltered liver of the Tilapia zilli. The same author has also reported an increase in the protein content and accumulation of protein granules in the liver of Tilapia zilli caught from the polluted locations. The occurrence of proteins in the liver is suggestive of the fact that it exhibits enormous diversity of biological functions, thereby confirming that the hepatocytes has an ability to produce proteins with strikingly different properties and activities by covalently joining the amino acids in many different combinations and sequences. Utilizing such productive proteins, fish can make such widely diverse products of utmost biological importance as enzymes, hormones, antibodies and a myriad of other substances having distinct biological activities.
DNA is of utmost biological importance as it plays a vital role in cellular metabolism. In the present study, Feulgen reaction has been employed for the histochemical localization of DNA. The Feulgen reaction is generally considered specific for DNA (Lillie, 1954).According to Pearse (1972a, b), the Feulgen reaction can be applied after almost any fixative except Bouin’s fixative with which the excessive hydrolysis occurs during fixation. However, during the present investigation, the tissues were fixed in Carnoy’s fixative which has been considered as an excellent nuclear fixative by Lillie (1954). Kurnic (1955) considered 8-12 min hydrolysis optimum for obtaining the deepest stain. In the present study however, the hydrolysis time was maintained at 10 min in N-HCl at 60°C. Goldshtein et al. (1952) and Deb and Banerjee (1957) observed that vitamin C regulates the viscosity and formation of DNA in the cells. In the present study however, the localization of DNA was carried out without providing any extra diet containing vitamin C. The centrally placed nuclei of the hepatic cells of Schizothorax curvifrons are observed to be sites of intense localization of DNA. The present observations and the earlier (Channa and Bhat, 2007; Channa and Lone, 2007) reveal that intense reaction for DNA is an indication for large synthesis of proteins taking place in the liver, suggesting high metabolic activity.
The presence of glycogen in the cytoplasm of hepatocytes is a characteristic feature of various fishes (Hampton et al., 1985; Moon et al., 1985; Gonzalez et al., 1993). The high content of glycogen in the cytoplasm of liver hepatocytes of Schizothorax curvifrons confirm previous observations in hepatocytes of Salmo gairdneri (Scarpelli et al., 1963), Carassius auratus (Yamamoto, 1965), Micropterus salmoides (Hinton et al., 1972), Ictalurus punctatus (Hinton and Pool, 1976), Oreochromis mossambicus (Chakrabarti et al., 1994) Tilapia zilli (Abdelmeguid et al., 2002) Nemacheilus kashmirensis (Channa and Lone, 2002), Oreochromis niloticus (Vicentini et al., 2005). The presence of glycogen is suggestive of an ability of Schizothorax curvifrons for synthesis (glycogenesis) or breakdown (glycogenolysis) of glycogen in relation to metabolic demand.
Regarding the distribution pattern and stain intensity of lipids in the liver, the present observations are in accordance with the findings of Abdelmeguid et al. (2002), who have reported intense stain intensity throughout the cytoplasm of the hepatocytes of Tilapia zilii. Adequate energy (lipids) reserves are required by organisms to mediate the effects of stress (Lee et al., 1983) and to serve as energy buffers during periods of harsh environmental conditions and food shortages (Adams and Mclean, 1985). Though the liver is not normally the chief accumulator of lipids, yet tends to keep its fat content relatively uniform even when large excesses of fat are being piled up in the adipose tissue thereby maintaining a nearly steady state of concentration of various lipids circulating in the blood. The excess fats in the liver leads to the production of lipoproteins. It could be concluded that fatty liver changes are mostly due to decrease in the production of lipoproteins. However, the phenomenon of fatty liver due to the storage of large quantities of fats is quite normal at certain times, including periods of sexual maturation (Kranz and Peters, 1985). Fatty liver must undoubtedly be considered as pathological when the diet is normal and fishes are beyond the spawning season (Roberts, 1978). According to Brauffaldi and Cucchi (1989), the presence of high lipid deposits may exert protective effects by removing and inactivating organic chemicals from the metabolism, thus improving toxicant tolerance and resistance.
Enzymes are the most remarkable and highly specialized proteins having extraordinary catalytic power, often far greater than that of synthetic or inorganic catalysts. They have a high degree of specificity for their substrates. A better knowledge of digestive enzyme activities is essential for a deeper understanding of the physiology of fish nutrition. As in other vertebrates, the ability of fish to utilize ingested nutrients depends on the presence of appropriate enzymes in appropriate locations. In the present study the intense alkaline and acid phosphatase activities observed in the cytoplasm of hepatic cells of Schizothorax curvifrons are in good agreement with the findings of Goel and Sastry (1973), Sastry (1975a) and Chakravorty and Sinha (1982). According to Loyda et al. (1979), alkaline phosphatase is found primary in the cell membranes where active transport takes place, however, the alkaline phosphatase activity in the hepatocytes is mainly concerned with the formation of glycogen through dephosphorylation process (Sinha et al., 1988). Srivastava (1966) has reported that alkaline phosphatase is firstly involved in the dephosphorylation process and so be concerned in the deposition of glycogen and secondly it possibly absorbs glucose. Acid phosphatase is one of the marker enzymes for lysosomes but its activity has also been detected outside the lysosomes (LIN and Fishmann, 1972). The occurrence of acid phosphatase activity in the liver of investigated fish may probably aid in the process of detoxification and other digestive and physiological processes.
The present observations on the intensity and distribution pattern of ATPase are in accordance with the findings of Sastry (1975b, 1976) who demonstrated strong adenosine triphosphatase activity in the cytoplasm and nuclei of the hepatocytes of Ophiocephalus punctatus and Hetreopneustes fossilis. Gawlicka et al. (1995) and Baglole et al. (1998) are of the opinion that ATpase work in collaboration of alkaline phosphatase. The ATPase plays an important role in maintaining the functional integrity of the cell membrane and other cellular activities. Lipases are the enzymes of low specificity requiring only an ester linkage for their reaction. There is no conclusive evidence about the secretion of lipase by liver, though its presence has been reported in the combined liver and pancreas (Ishida, 1936; Sarbahi, 1951). However, the present observations showing intense lipase activity in the cytoplasm around the nucleus of the hepatic cells of the fish studied are not only consistent with the findings of Sastry (1974a, b) but also extend support to the view of Barrington (1957) who states that the secretion of lipase might be a property of hepatic tissue itself.
This study was realized with financial support from University of Kashmir, INDIA. The authors sincerely offer their thanks to Prof. R.C. Bhagat, former head Department of Zoology and Prof. G. Mustafa Shah, head Department of Zoology, University of Kashmir, Srinagar, INDIA, for providing the necessary lab facilities. The corresponding author, Dr. I.H. Mir personally wishes to thank Miss. Sumaira Nabi, research scholar Department of Zoology, University of Kashmir, not only for her untiring efforts to make this work successful but also for her unforgettable support at every step. Her concern for me has been ever more inspiring and encouraging, hence she deserve my thanks unreservedly.