Research Article
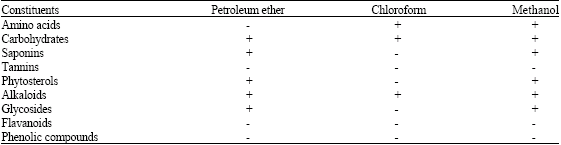
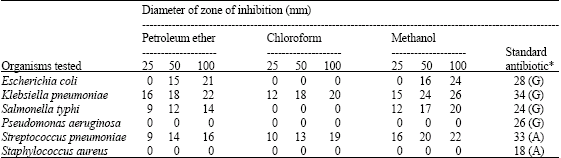
Preliminary Phytochemical Analysis, Elemental Determination and Antibacterial Screening of Codium decorticatum-A Marine Green Algae
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
R. Suraj
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
K. Anandarajagopal
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
G. Rejitha
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
M. Vignesh
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia
P. Promwichit
School of Pharmacy, Masterskill University College of Health Sciences, Taman Kemacahaya 11, Jalan Kemacahaya, Cheras, Selangor, Malaysia










g subramanian Reply
Sir
This is very good publication for futher rference