Research Article
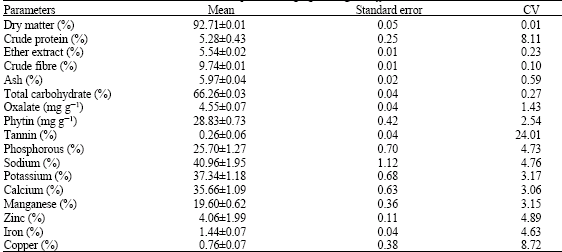
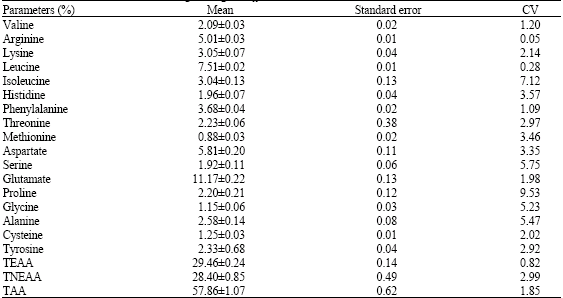
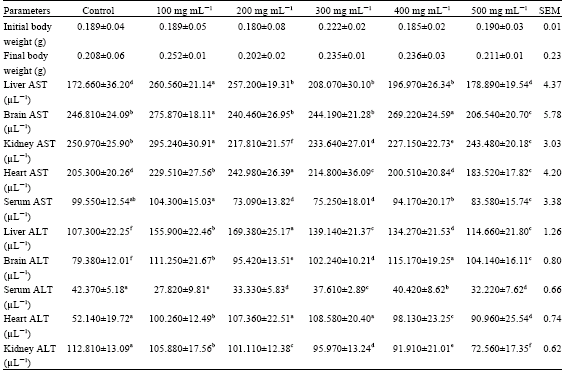
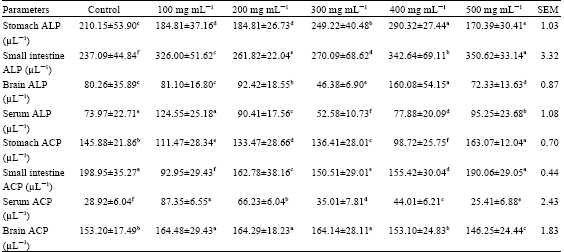
Nutrient and Anti-nutrient Constituents of Ginger (Zingiber officinale, Roscoe) and the Influence of its Ethanolic Extract on Some Serum Enzymes in Albino Rats
Department of Biochemistry, Nutritional Biochemistry Unit, Department of Animal Production and Health Sciences, University of Ado-Ekiti, P.M.B. 5363, Ado-Ekiti, Nigeria
F.A.S. Dairo
Department of Biochemistry, Nutritional Biochemistry Unit, Department of Animal Production and Health Sciences, University of Ado-Ekiti, P.M.B. 5363, Ado-Ekiti, Nigeria