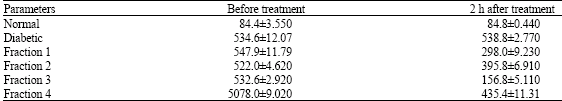Research Article
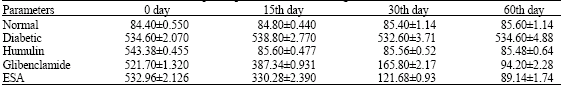
Effect of Crude Extract and Fractions from Elephantopus scaber on Hyperglycemia in Streptozotocin-Diabetic Rats
Department of Biotechnology, Holy Cross College, Tiruchirappalli-620 002, Tamil Nadu, India
P. Daisy
Department of Biotechnology, Holy Cross College, Tiruchirappalli-620 002, Tamil Nadu, India