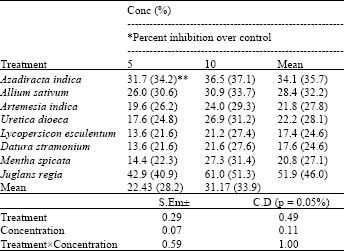
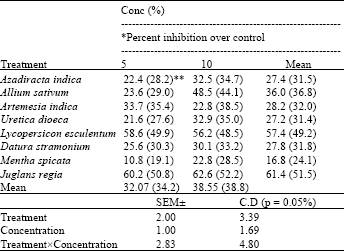
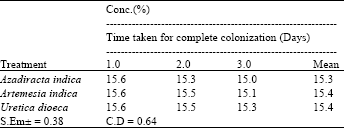
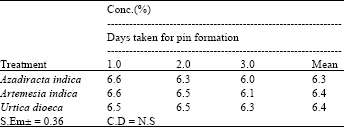
Research Article
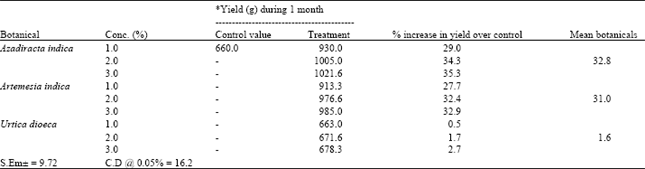
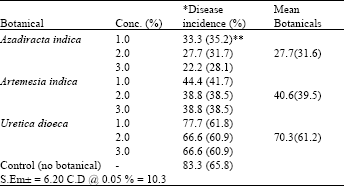
Evaluation of Some Botanicals in Controlling Green Mold (Trichoderma harzianum) Disease in Oyster Mushroom Cultivation
Department of Botany, Government Institute of Science, Babasaheb Ambedkar�s Marathwada, University Aurangabad, 431004 (M.H), India
Sahera Nasreen
Department of Botany, Government Institute of Science, Babasaheb Ambedkar�s Marathwada, University Aurangabad, 431004 (M.H), India
N.A. Munshi
Department of Pathology, SKAUST-K, Shalimar Campus, Srinagar, 191121, Kashmir