Research Article
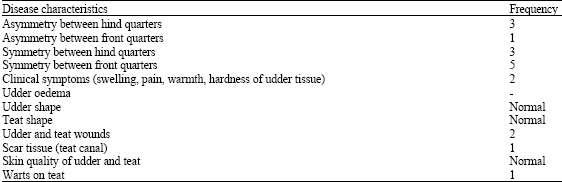
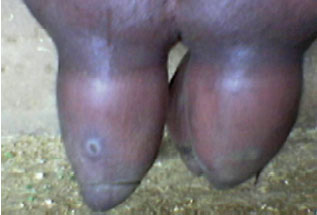
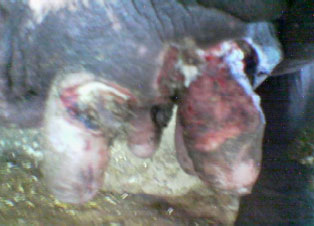
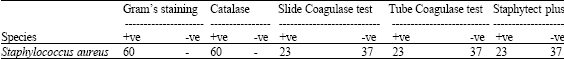
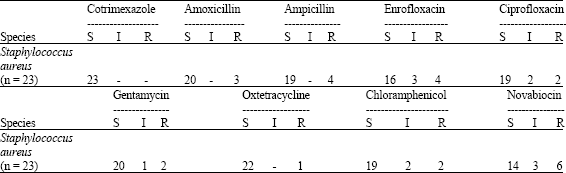
Clinical and Subclinical Staphylococcus aureus Mastitis in Dairy Buffaloes: Disease Characteristics and Antibiotic Susceptibility Profiles of Isolates
Department of Clinical Medicine and Surgery, Faculty of Veterinary Sciences, University of Agriculture, Faisalabad, Pakistan
Abdul Shakoor
Department of Clinical Medicine and Surgery, Faculty of Veterinary Sciences, University of Agriculture, Faisalabad, Pakistan
Muhammad Akbar Shahid
Department of Veterinary Microbiology, Faculty of Veterinary Sciences, University of Agriculture, Faisalabad, Pakistan
Muhammad Numan
Veterinary Research Institute, Ghazi Road, Lahore Cantt-13, Pakistan
Fahad Gulraiz
Department of Veterinary Microbiology, Faculty of Veterinary Sciences, University of Agriculture, Faisalabad, Pakistan