ABSTRACT
The seed proteins of sunflower (Helianthus annuus) were qualitatively and quantitatively investigated. Qualitative studies were carried out using different electrophoretic techniques (2-DE). Analysis of the water extracted flour on SDS-PAGE gave eight major polypeptides clusters with MWs of lower than 14.4, 14.4-18.4, 18.4-25.0, 25-35, 35-45, 45-66.2, 66.2-116 and higher than 116 kDa. Second dimension gel showed that the polypeptides with MWs of 45-66.2 kDa are legumin-like proteins. Isoelectric points of the majority of the sunflower seed proteins were between 3 and 10. Mapping gels, however, showed that sunflower seed proteins were highly heterogeneous, especially the major bands. A quantative study indicated that the albumin, globulin, prolamin and glutelin fractions amounted 38.32, 39.04, 5.53 and 17.09%, respectively of the extracted proteins.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/biotech.2014.245.247
URL: https://scialert.net/abstract/?doi=biotech.2014.245.247
INTRODUCTION
Oil crops have a world importance. Some of them are directly used as food, while most of them are utilized to obtain fats or oils and cake or flour (Lennerts, 1983; Hatje, 1989). Sunflower (Helianthus annuus) is an annual oilcrop (belonging to the family Compositae) (Lennerts, 1983). The defatted flour of sunflower is used as a source of highly digestible and nutritive protein for poultry (Smith, 1968). It has been reported that the major proteins (legumin-like proteins) of sunflower have sedimentation constant of about 11 S and that their Molecular Weights (MWs) range from 300-350 kDa (Joubert, 1955; Gheyasuddin et al., 1970; Sabir et al., 1973; Youle and Huang, 1981; Hatje, 1989). Heterogeneity and quaternary structure of the major storage proteins have been investigated by Baudet and Mosse (1977), Rahma and Rao (1981), Plietz et al. (1983), Dalgalarrondo et al. (1984) and Abasery (1992). As far as is known, all the studies carried out on sunflower seed proteins were done with the major seed proteins. The present study, therefore, is made to investigate new major and minor components of the sunflower seed proteins.
MATERIALS AND METHODS
Sunflower seeds were obtained from the National Institute of Genetic Engineering and Biotechnology (NIGEB), Tehran, Iran. The seeds were dehulled and ground well in a mortar. The flour was defatted by three hexane extractions (10 mL hexane/g flour), each for 2 h with slow stirring at 4°C. After the n-hexane layer was discarded, the flour was air-dried, brushed through a sieve of 125 urn (115 mesh) and then stored at -20°C until used.
Total protein extraction and quantization: Total protein was isolated from seed tissues using Invitrogen’s TRIZOL® Reagent according to the manufacturer’s instruction. Protein concentration was determined by Bradford assay.
2-DE and image analysis: The sunflower proteins in the dried powder were solubilized in 7 M urea, 2 M thiourea, 2% CHAPS (powder to solution/v), 0.5% pH 3-10 IPG buffer v/v (GE Healthcare, USA) and 36 mM 1,4-dithio-DL-threitol (DTT) (5.6 mg mL-1) via incubationat room temperature for 1h, vortexing every 10 min, followed the mixture was centrifuged (15000 rpm) for 15 min and the supernatant collected. Total protein extract (66 μL) was loaded onto GE Healthcare 18 cm IPG gel strips (pH 3-10 L) during strip rehydration overnight, after which, IEF was performed for a total of 65 kV.h using IPG Phor II (GE Healthcare) at 20°C. The IEF buffer contained 7 M urea, 2 M thiourea, 2% CHAPS (powder to solution, w/v), 0.5% pH 3-10 IPG buffer v/v (GE Healthcare) and 36 mM DTT (5.6 mg mL-1). The IPG strips were equilibrated according to manufacturer (GE Healthcare). PAGE gels (12% linear gradient) were run on an Ettan Dalt six (GE Healthcare), 0.5 h at 2.5 W per gel, then at 15 W per gel until the dye front reached the gel bottom. Upon electrophoresis, the protein spots were stained with silver nitrate according to the instruction of protein PlusOne™ Silver Staining Kit (GE Healthcare) which offered improved compatibility with subsequent mass spectrometric analysis.
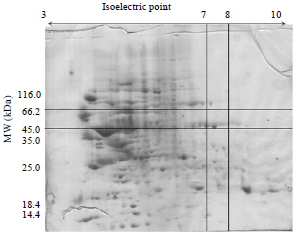 | |
| Fig. 1: | 2-DE of total storage protein in sunflower (Helianthus annuus) proteins between 45.0 and 66.2 kDa |
Briefly, gels were fixed in 40% ethanol and 10% acetic acid for 30 min and then sensitized with 30% ethanol, 0.2% sodium thiosulfate w/v and 6.8% sodium acetate w/v for 30 min. Then gels were rinsed with distilled water three times, 5 min for each time, then incubated in silver nitrate (2.5 g L-1) for 20 min. Incubated gels were rinsed with distilled water and developed in a solution of sodium carbonate (25 g L-1) with formaldehyde (37%, w/v) added (300 mL L-1) before use. Development was stopped with 1.46% EDTANa2.2. H2O w/v and gels were stored in distilled water until they could be processed and the reproducible spots removed from them. Gel images were acquired using Lab scan (GE Healthcare). Image analysis was carried out with Image-master 2D Platinum Software Version 5.0 (GE Healthcare). For the determination of the molecular weight, a mixture of the following marker proteins were used: β-galactosidase (116 kDa), Bovin Serum Albumin (66.2 kDa), Ovaalbumin (45 kDa), Lactatedehydrogenase (35 kDa), Endonuclease bsp981 (25 kDa) and β-lactoglobulin (18.4 kDa). The polypeptides which dissociated under 2-DE conditions were claustered in eight groups: Lower than 14.4, 14.4-18.4, 18.4-25.0, 25-35, 35-45, 45-66.2, 66.2-116 and higher than 116 kDa. The polypeptides with MWs of 45-66.2 kDa are legumin-like proteins (Fig. 1). These polypeptides are clustered into 3 groups including acidic groups consist of 5 sposts, neutral groups consist of 3 spots and basic groups consist of 4 spots (Table 1).
RESULTS AND DISCUSSION
Sunflower seed flour was successively extracted with distilled water (albumin), salt (NaCl) (globulin), alcohol (prolamin) and alkaline solution (glutelin) using the method of Bradford (1976).
| Table 1: | Legumine like protein clusters in sunflower (Helianthus annuus) profile with number of spots in three clusters, acidic, neutral and basic isoelectric points with molecular weight (45-66.2 kDa) |
 | |
| Table 2: | Protein contents of the albumin, globulin, prolamin and glutelin fractions of sunflower seed flour |
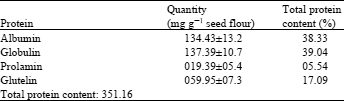 | |
Protein contents of 134.43±13.62 mg g-1 seed flour and 137.39±6.34 mg g-1 seed flour were found respectively in the water and salt extract (Table 1). The protein extracted with the alkaline solution represented nearly half of the protein content found after salt extraction. Protein analysis cannot be carried out in extracts containing ampholyte, since the ampholytes complex with copper ions (Shah and Stegemann, 1983). A protein content in sunflower of 16.6-20.3% has been reported by Hatje (1989) which contradicts our findings (35%). Youle and Huang (1981) reported that 33% of sunflower seed protein was albumin. However, the variation between their data and the data reported in the present study could be due to methodological or varietal differences (Table 2).
Isoelectric focusing was carried out between pH 3-10 in cylindrical tubes containing 8 M urea. The polypeptide distribution of the buffer extracts consists of three clusters of polypeptides reported in Table 1. It is very interesting to notice that the acidic clusters have pIs between 3-7, the neutral clusters have pIs between 7-8 and the basic clusters have pIs between 8-10. The gel also show that the basic and acidic clusters are highly heterogeneous. However, the acidic clusters are distributed in a wide pH range, while the basic clusters are distributed over a narrow pH range. The heterogeneity can be explained as follows:
| • | Clusters could be composed of several nearly identical polypeptides |
| • | The proteolytic modifications could produce charge variants and in this event, the differences between molecules in each preparations would be evident only at the ends of the molecules |
| • | Glutamine and asparagine could be diamidated in some peptides. In conclusion, sunflower seed proteins contain approximatly equal amounts of albumin and globulin proteins. These proteins represent about 77% of the total seed proteins. The total seed proteins, in turn, represent about 35% of the seed meal. This percentage makes sunflower seed proteins a good resource for feeding animals. The richness of the legumin-like proteins in sulphur amino acids makes them highly nutritional. with 2-ME, the legumin-like proteins were cleavaged into acidic and basic subunits. The pI-values of sunflower seed proteins range between 3-10. |
REFERENCES
- Baudet, J. and J. Mosse, 1977. Fractionation of sunflower seed proteins. J. Am. Oil Chem. Soc., 54: A82-A86.
CrossRefDirect Link - Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248-254.
CrossRefPubMedDirect Link - Dalgalarrondo, M., J. Raymond and J.L. Azanza, 1984. Sunflower seed proteins: Characterization and subunit composition of the globulin fraction. J. Exp. Bot., 35: 1618-1628.
CrossRef - Joubert, F.J., 1955. Sunflower seed proteins. Biochemica et Biophysica Acta, 16: 520-523.
CrossRefDirect Link - Rahma, E.H. and M.S.N. Rao, 1981. Isolation and characterization of the major protein fraction of sunflower seeds. J. Agric. Foods Chem., 29: 518-521.
CrossRefDirect Link - Sabir, M.A., F.W. Sosulski and S.L. MacKenzie, 1973. Gel chromatography of sunflower proteins. J. Agric. Food Chem., 21: 988-993.
CrossRefDirect Link - Youle, R.J. and A.H.C. Huang, 1981. Occurrence of low molecular weight and high cysteine containing albumin storage proteins in oilseeds of diverse species. Am. J. Bot., 68: 44-48.
Direct Link








