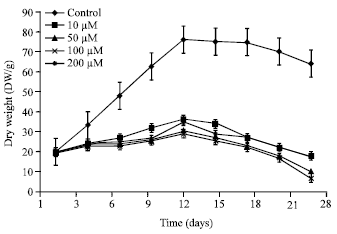
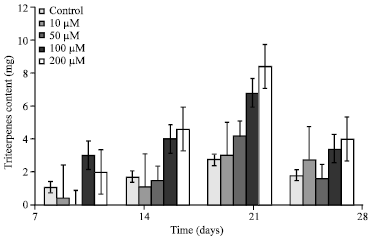
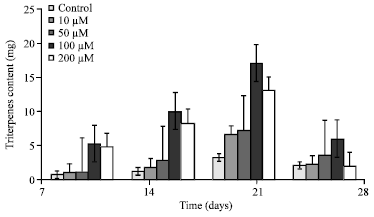
Research Article
Ursolic Acid and Oleanolic Acid Productions in Elicited Cell Suspension Cultures of Hedyotis corymbosa
Faculty of Applied Science, Universiti Teknologi MARA, 40450, Shah Alam, Selangor, Malaysia
M. Yaseer Suhaimi
Faculty of Applied Science, Universiti Teknologi MARA, 40450, Shah Alam, Selangor, Malaysia
A. Rohaya
Faculty of Applied Science, Universiti Teknologi MARA, 40450, Shah Alam, Selangor, Malaysia
N.A.R. Nik Roslan
Faculty of Applied Science, Universiti Teknologi MARA, 40450, Shah Alam, Selangor, Malaysia