Research Article
Characterization of Collagen from Eggshell Membrane
Food College, Northeast Agricultural University, Harbin, People`s Republic of China
Yu-Jie Chi
Food College, Northeast Agricultural University, Harbin, People`s Republic of China
Collagen isolated from the skins and bones of land-based animals is capable of utilization, mainly it is from bovine and swine. Non denatured collagens from these sources are widely and diversely used in food, cosmetics, biomedical and pharmaceutical industries (Ogawa et al., 2004). After the outbreaks of bovine spongiform encephalopathy and foot-and-mouth disease crisis, the restrictions on collagen trade have been established and we need alternative safe sources of collagen (Devore et al., 2004). Another problem of bovine collagen application is the risk of autoimmune and allergic reactions, about 2-3% of the population is allergic in this regard. According to the facts mentioned above, many researchers have done a lot of work to investigate the possibility of new source of collagen from aquatic life. Their researches involve optimal isolation methods from different kinds of fish and the functional properties of marine source collagens (Takeshi and Nobutaka, 2002; Sadowska et al., 2003; Jongjareonrak et al., 2005; Wang et al., 2008; Woo et al., 2008).
As a kind of common industrial waste, eggshell can be readilycollected anywhere in plenty. Eggshell membranes are composed of protein fibers between egg white and inner surface of eggshell. Eggshell membrane protein has high percentages of certain amino acids such as arginine, glutamic acid, methionine, valine, cystine and proline. The presence of hydroxyproline in hydrolysates of membrane layers suggest that membrane layers fiber structure consists of collagen as component (Wong et al., 1984). Biochemical and immunological tests have verified the deduction. Eggshell membrane primarily contains type I collagen, type V collagen and type X collagen (Wong et al., 1984; Arias et al., 1992), all the three kinds of collagen can be used in various fields. It has been determined that about 10% of total proteinaceous constituent in eggshell membrane structure is collagen.
Eggshell membrane will be an alternative potential important source of collagen for application in foods, cosmetics and biomedical materials, if we can verify the similar character of collagen from eggshell membrane and land-based mammalians tissue. But few are known about the chemical properties of eggshell membrane collagen, although eggshell membrane has been shown to contain collagenous protein (Wong et al., 1984). In China, eggshell is consistent available as by-product from food industry, that is about 4 million tons per year. Eggshell membrane collagen has been proved to be of very low autoimmune and allergic reaction (Long et al., 2004). According to the result of research on characteristic of eggshell membrane and biosafety, the possibility of application in functional foods, cosmetic and other industries is great.
The aim of the research is to investigate the characteristics of collagen extracted from eggshell membrane with amino acid analysis, Sodium Dodecyl Sulphate-Polyacrylamide Gel Electrophoresis(SDS-PAGE), Fourier Transforms Infrared (FTIR) spectroscopy and Differential Scanning Calorimetry (DSC) and compare its characteristic with collagen from land-based mammalians.
Materials: Raw eggshell membrane: obtained from commercial eggs, the membrane is peeling off manually and consisted of inner and outer membranes. Pepsin (1:10000): purchased from Sigma Inc. All reagents used in this study were analytical grade.
Extraction of collagen from eggshell membrane: The collagen was extracted with the method of Wong et al. (1984) with a slight modification. The manipulations were performed at 4°C. The eggshell membranes were homogenized with a homogenizer. To remove non-collagenous proteins, fresh eggshell membranes were soaked in 0.1 N NaOH at a sample/alkaline solution ratio of 1:10 (w/v) for 24 h. The alkaline solution was changed every 6 h. Then, alkali-treated eggshell membrane was washed with cold deionized water until neutral or faintly basic pH of wash water was obtained. Then the precipitate was subjected to pepsin digestion at 0.5% for 24 h in 0.5 M acetic acid. The digest was centrifuged at 10,000 g for 45 min and the precipitate was washed with three 50 mL portions of 0.5 M acetic acid and discarded. The supernatant and washes were pooled together and filtered through a fritted disk funnel. Solid NaCl was added to the solution to achieve a final concentration of 0.9 M and stirred for 12 h. The precipitate was collected by centrifugation (10,000 g, 45 min), redissolved in 0.5 M acetic acid and dialyzed exhaustively against the 0.5 M acetic acid, 0.1 M acetic acid and deionized water. The dialysates were then lyophilized.
Amino acid analysis: A 5 mg aliquot of collagen was dissolved in 3 mL of 6 N HCl and hydrolyzed in vacuum-sealed glass tubes at 110°C for 24 h using a dry bath incubator. Hydrolyzed samples were filtered through glass filters and the filtrates dissolved in citric acid buffer (pH 2.2) and injected into an amino acid auto analyzer (Amino acid analyzer 835, Hitachi Co., Japan).
SDS-polyacrylamide gel electrophoresis (SDS-PAGE):Electrophoresis patterns were measured with the method of Guo (2005), using Mini-Protein 3 (Bio-Rad Laboratories, Hercules, California). Polyacrylamide gel was prepared with 5% stacking gel and 12% resolving gel.
The collagen samples were dissolved in the sample buffer (0.5 M Tris–HCl, pH 6.8, containing 4% SDS, 20% glycerol) in the presence of 10% β-ME. Samples were loaded onto the gel. After electrophoresis, gel was stained with Coomassie brilliant blue R-250 in 15% v/v) methanol and 5% (v/v) acetic acid). Molecular weight markers were used to estimate the molecular weight of protein bands. Type I collagen of pig bone was used as control.
Fourier transforms infrared spectroscopy (FTIR): FTIR spectra analysis was performed with 1.5 mg collagen in approximately 150 mg potassium bromide (KBr). All spectra were obtained from 4000 to 400 cm-1 at a data acquisition rate of 4 cm-1 by using a FTIR spectrophotometer (FTIR-8400S, Shimadzu, Japan).
Differential scanning calorimetry: The denaturation temperature (Td) of collagen was determined with DSC using a Perkin-Elmer DSC-6. The samples (collagens of eggshell membrane) were prepared with slightly modified method of Rochdi et al. (2000) and Komsa-Penkova et al. (1999).
Temperature calibration was carried out with Indium thermogram. The samples (5-10 mg) were weighed accurately and sealed into aluminium pans. The samples scan speed is at 5°C min-1 over the range of 40-100°C. An empty pan was used as the reference. Total denaturation enthalpy (ΔH) was estimated by measuring the area in the DSC thermogram. The maximum transition temperature (Td) was estimated from the thermogram.
Amino acid composition: The results of amino acid analyses of eggshell membrane collagen were listed in Table 1. Compared with the analysis result of bovine-skin type I collagen (Jiang, 2006), the amino acid composition of eggshell membrane collagen is of similar ratio to the former, i.e., the amino acid profile is similar. Glycine is the most prevalent amino acid in eggshell membrane collagen at 30.27%. The stabilization of conformation requires the occurrence of glycine residues at one of every three position in specific amino acid sequence of peptide chain. The contents of proline and hydroxyproline may be important for structural integrity of collagen. The proportions of hydroxyproline and proline residues are 9.28 and 11.94 residues per 100 amino acids residues, respectively. There are small proportion of cysteine, methionine, histidine, phenylalanine and tyrosine residues with 0.34, 0.76, 0.80, 0.81 and 0.92 residues per 100 amino acids residues, respectively. The amino acid composition of collagen consists of numerous repeating Gly-Y-X residues in a triple helical conformation (Woo et al., 2008). Consequently, we can deduce that the structure of eggshell membrane collagen is a (Gly-Pro-Hyp)n pattern with the consideration of these facts.
| Table 1: | Amino acid compositions of eggshell membrane collagen and bovine-skin collagen |
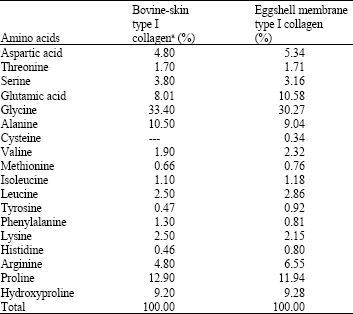 | |
| Amino acid composition of bovine-skin type I collagen, cited from Jiang (2006) | |
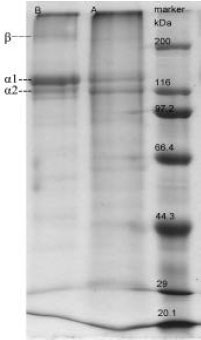 | |
| Fig. 1: | SDS-PAGE pattern of collagen from eggshell membrane. Lane A: Type I collagen of eggshell membrane; Lane B: The pig-bone type I collagen; Lane marker: Protein molecular weight standards |
And eggshell membrane collagen possesses the most common triplet helical configuration in 3 dimension space.
SDS-PAGE profile of eggshell membrane collagen: Electrophoretic profile of eggshell membrane collagen was laid out by SDS-PAGE with pig-bone collagen as reference.
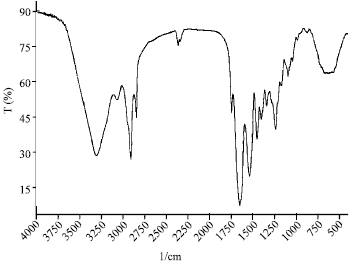 | |
| Fig. 2: | Fourier transform infrared spectrum of eggshell membrane collagen |
Type I collagen extracted from mammalian is mostly consisted of two α chains (2:1 ratio of α1 and α2) and β-component (Lee et al., 2002). Similar to type I collagen of pig-bone, the eggshell membrane collagen comprised at least two different α-chains (α1 and α2) with similar mobilities to pig-bone collagen. The result indicates that eggshell membrane collagen might be type I collagen primarily. Figure 1 shows the SDS-PAGE profile of eggshell membrane collagen (type I) and eggshell membrane type I collagen consists of α1 chain (MW is about 130 kDa) and α2 chain (MW is about 116 kDa). The ratio of α1 (I) and α2 (I) chains is approximate 1:1.
Fourier transform infrared spectroscopy: FTIR spectroscopy has been used to study changes in the secondary structure of collagen (Friess and Lee, 1996).
Amide I band is associated with stretching vibrations of carbonyl groups (C=O bond), within 1600-1700 cm-1, along the polypeptide backbone (Payne and Veis, 1988) and it is the most useful for infrared spectroscopic analysis of the secondary structure of proteins (Surewicz and Mantsch, 1988). The FTIR spectrum of eggshell membrane collagen is shown in Fig. 2. The main absorption bands are amide A (3325 cm-1), amide B (2926 cm-1), amide I (1653 cm-1), amide II (1550 cm-1) and amide III (1240 cm-1). Amide A band is related to NH stretch coupled with hydrogen bond, amide B is related to CH2 asymmetrical stretch. Amide II is associated with NH bending and CN stretching. Amide III is related to CN stretching and NH and is involved with the triple helical structure of collagen. All of these are similar to those results of research about collagen from deep-sea redfish, yellowfin tuna dorsal skin and Nile perch (Muyonga et al., 2004; Wang et al., 2008; Woo et al., 2008).
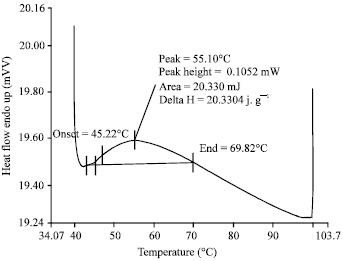 | |
| Fig. 3: | The denaturation temperature (Td) and total denaturation enthalpy (ΔH) of eggshell membrane collagen |
Denaturation temperature (Td): The Td of eggshell membrane collagen can be used as an effective index of assessing the stability of eggshell membrane type I collagen. With increase in temperature (thermal depolymerization process), the hydrogen bonds within collagen are broken progressively and finally, the triple helix structure of collagen maintained by hydrogen bonds is converted into the random coil conformation of gelatin (Wang et al., 2008). The collagen Td of different animal species seems to be correlated with the content of amino acids (proline and hydroxyproline). The higher the amino acid content, the higher the stability of helices (Wong, 1989).
DSC thermogram of collagen from eggshell membrane is shown in Fig. 3. The Td is determined to be about 55.10°C for eggshell membrane type I collagen. The value of ΔH of eggshell membrane collagen is 20.3304 J g-1. The enthalpy changes associated with collagen denaturation processes depend on positional preferences of ionized residues of Gly-X-Y group and formation of hydrogen bonds of inner coil-coiled α-chains (Usha and Ramasami, 2004; Cao and Xu, 2008). The Td of eggshell membrane collagen is lower than pig skin collagen (60°C) and bovine skin (63-65°C), higher than aquatic life collagen. Body collagen stability is correlated with environmental and body temperature.
This study investigated the characteristics of collagen extracted from eggshell membrane with the methods of amino acid analysis, SDS-PAGE, FTIR and DSC. The collagen can be classified as type I collagen according to the results and analysis of amino acid composition and SDS-PAGE tests. The results of FTIR spectra and denaturation temperature show that eggshell membrane collagen maintain their triple helical structures well, be of higher thermal stability. Eggshell membrane collagen should be thought to be of similar characteristics as other sources. Therefore, the results suggest that collagen of eggshell membrane should be a potential resource alternative to mammalian collagen for commercial applications. Eggshell membrane collagen may be applicable to variety of application, such as functional food, cosmetic, biomedical and pharmaceutical industries.
This research was supported by the National Natural Science Foundation of China (Grant No. 30871954).