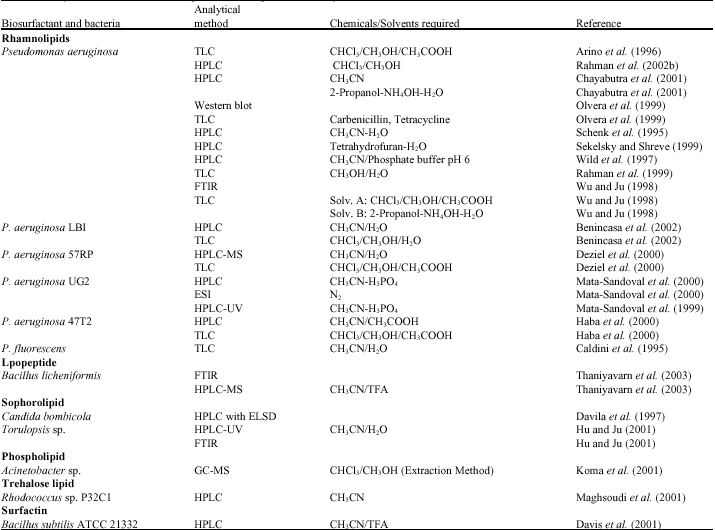
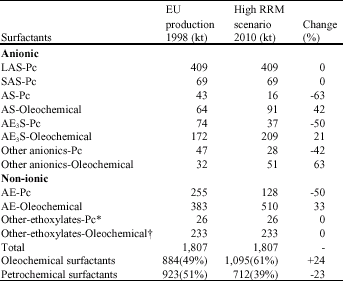
Research Article
Production, Characterisation and Applications of Biosurfactants-Review
School of Science and Technology, University of Teesside, Middlesbrough-TS13BA, UK
Edward Gakpe
School of Science and Technology, University of Teesside, Middlesbrough-TS13BA, UK










YOUNGKOOK CHOI Reply
I have been researching on the biosurfactant. if possible i strongly want to read. please send this file