Research Article
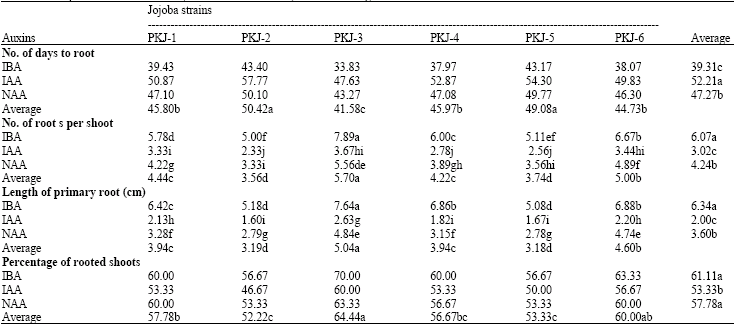
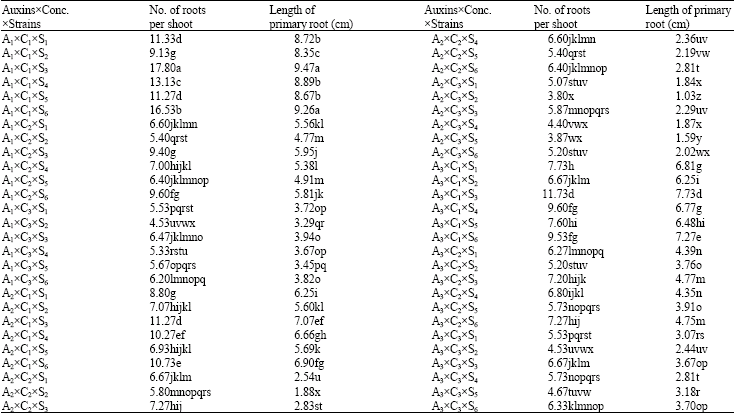
In vitro Root Formation in Micropropagated Shoots of Jojoba (Simmondsia chinensis)
Horticultural Research Station, Bahawalpur-63100, Pakistan
Muhammad Akbar Anjum
University College of Agriculture, Bahauddin Zakariya University, Multan, Pakistan
Hamid Rashid
Agricultural Biotechnology Institute, National Agriculture Research Centre, Islamabad, Pakistan