Research Article
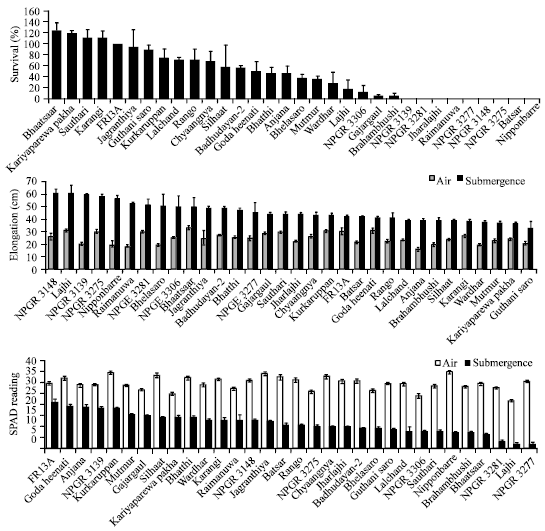
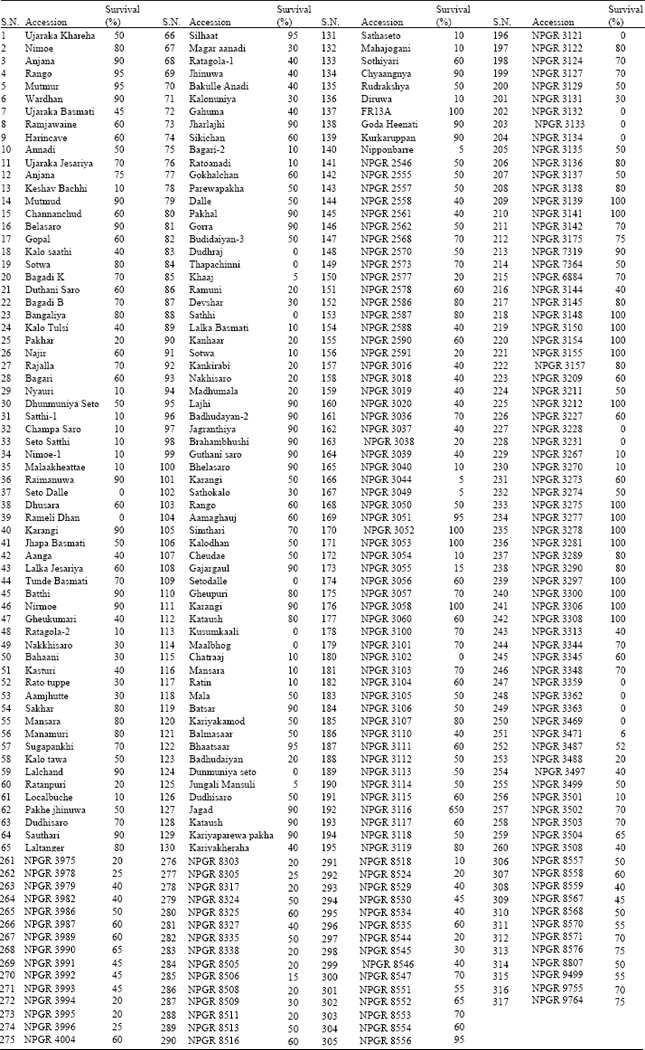
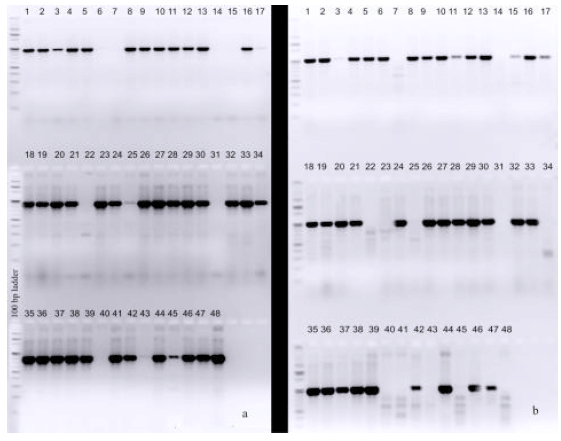
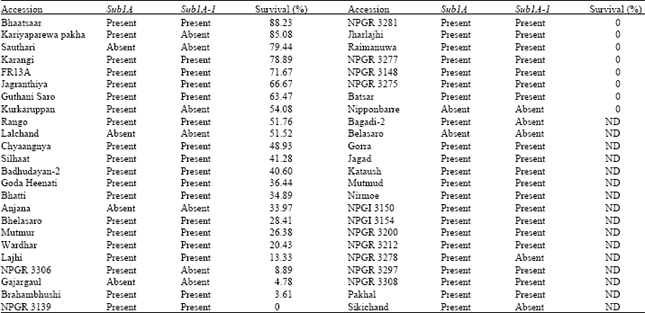
Determination of Adaptive Mechanisms for Flash Flooding Tolerance in Nepalese Cultivated Rice Genepool based on Morpho-physiological and Molecular Analysis
Tribhuvan University, Institute of Agriculture and Animal Science, Rampur, Chitwan, Nepal
Raj Kumar Niroula
Biotechnology Unit, Nepal Agricultural Research Council, Khumaltar, Lalitpur, Nepal
Resham Babu Amgai
Biotechnology Unit, Nepal Agricultural Research Council, Khumaltar, Lalitpur, Nepal
Bindeswar Prasad Sah
Biotechnology Unit, Nepal Agricultural Research Council, Khumaltar, Lalitpur, Nepal
Surya Kant Ghimire
Tribhuvan University, Institute of Agriculture and Animal Science, Rampur, Chitwan, Nepal