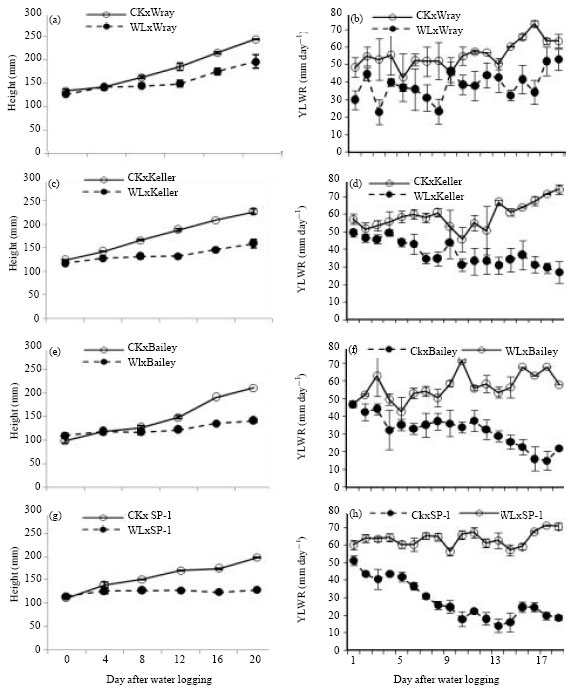
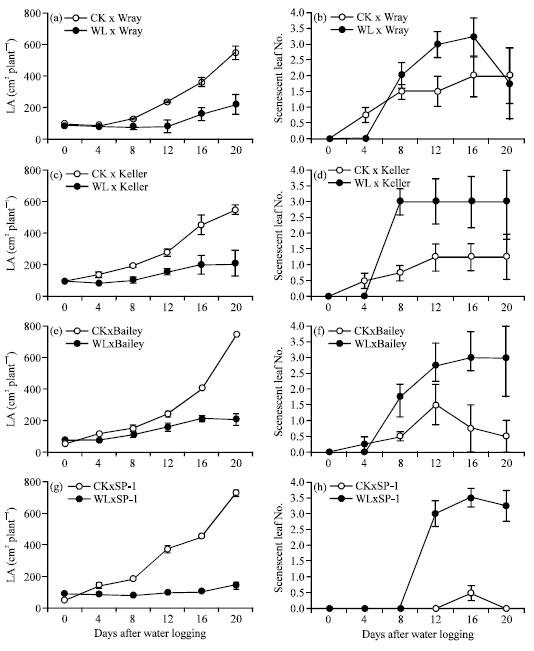
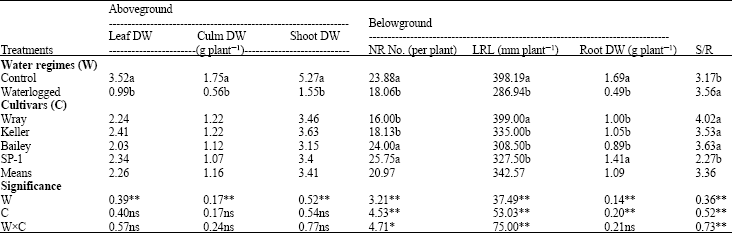
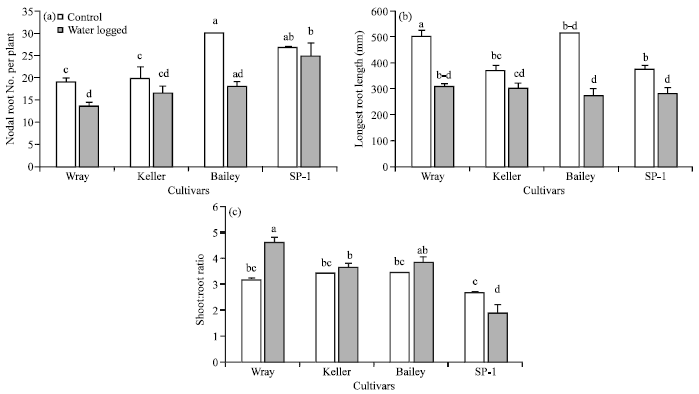
Research Article
Morphological and Physiological Responses of Sorghum (Sorghum bicolor L. Moench) to Waterlogging
Department of Plant Science and Agricultural Resources, Faculty of Agriculture,Khon Kaen University 40002, Thailand
A. Younger
School of Agriculture, Food and Rural Development, Agriculture Building, Newcastle University,Newcastle Upon Tyne, NE1 7RU, United Kingdom
A. Polthanee
Department of Plant Science and Agricultural Resources, Faculty of Agriculture,Khon Kaen University 40002, Thailand
C. Akkasaeng
Department of Plant Science and Agricultural Resources, Faculty of Agriculture,Khon Kaen University 40002, Thailand