Research Article
In vitro Antibacterial Properties of Total Alkaloids Extract from Mitragyna Inermis (Willd.) O. Kuntze, a West African Traditional Medicinal Plant
Centre de Recherche en Sciences Biologiques, Alimentaires et Nutritionnelles, UFR/SVT, Universite de Ouagadougou, 03 BP 7131, Ouagadougou 03, Burkina Faso
Etienne-Francois O. Akomo
Centre de Recherche en Sciences Biologiques, Alimentaires et Nutritionnelles, UFR/SVT, Universite de Ouagadougou, 03 BP 7131, Ouagadougou 03, Burkina Faso
A. Savadogo
Centre de Recherche en Sciences Biologiques, Alimentaires et Nutritionnelles, UFR/SVT, Universite de Ouagadougou, 03 BP 7131, Ouagadougou 03, Burkina Faso
Louis Clement Obame
Centre de Recherche en Sciences Biologiques, Alimentaires et Nutritionnelles, UFR/SVT, Universite de Ouagadougou, 03 BP 7131, Ouagadougou 03, Burkina Faso
Jean Koudou
Centre de Recherche en Pharmacopee et Medecine Traditionnelle, Universite de Bangui, BP 1450, Bangui Republique Centrafricaine
Alfred S. Traore
Centre de Recherche en Sciences Biologiques, Alimentaires et Nutritionnelles, UFR/SVT, Universite de Ouagadougou, 03 BP 7131, Ouagadougou 03, Burkina Faso









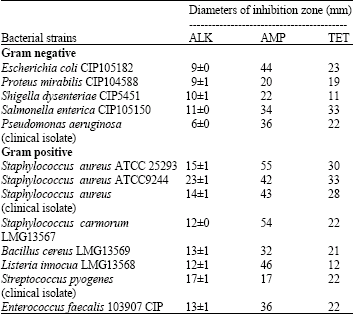
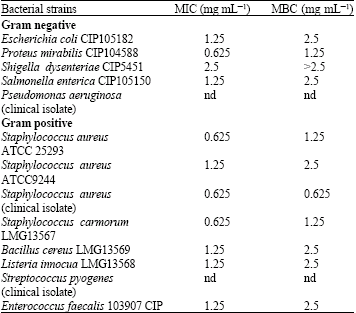
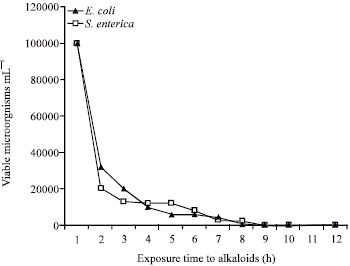
ahmad shafiei darabi Reply
Dear
I am ahmad shafiei
I graduated from shahid beheshti university, Iran
I work on galbanum and have a paper about it.
The title of paper is "The comparison of galbanum (Ferula gummosa Boiss) from five regions of Iran"
Pleas send your address or email
Thanks
zongo cheikna
Thank you for you interest for my work. You can make contact with me for scientic exchanges and over.
Best regards!
saeed aria Reply
hello i am horticulture engineerig student in tehran!
i"d look like
your aricle was good