ABSTRACT
Production and infectivity of Steinernema carpocapsae, S. abbasi, S. karii and S. riobravis (entomopathogenic nematodes) using larvae and pupae of diamondback moth were studied. Nematodes production of all species was determined by the number infective juveniles (IJs) established in DBM larvae and pupae using sand and filter paper bioassy. Maximum number of S. carpocapsae were produced at 25°C, however the production of other isolates was higher at 30°C. Infectivity tests carried out in sand media revealed that maximum IJs of S. carpocapsae were recovered from the DBM larvae and pupae at 25°C whereas other three isolates produced reasonable number of IJs at 30°C. Similarly, DBM pupae with cocoon produced higher number of IJs than without cocoon. The research indicated that application of nematodes with the knowledge of insect pest biology that represents a possible new strategy for controlling DBM larvae and pupae.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.484.488
URL: https://scialert.net/abstract/?doi=ajps.2004.484.488
INTRODUCTION
Diamondback moth (DBM) is a cosmopolitan pest of cruciferous and other crops. Each year, farmers worldwide spend more than one billion dollars to control this pest[1]. DBM has been controlled by various pesticides but in recent years resistance to most of the conventional insecticides has developed[2]. The problems of insecticide resistance as well as the environmental and consumer health hazards associated with insecticide residues in plant material have focused attention on alternative methods for the control of DBM. Alternative control measures investigated for this insect-pest include the use of entomopathogenic nematodes and other biocontrol agents and to incorporate it into IPM programmes against this insect pest.
Entomopathogenic nematodes in the families Steinernematidae and Heterorhabditidae have been shown to be pathogenic to a wide range of agriculturally important pests and are useful alternatives to insecticides for insect control[3]. The steinernematid, S. carpocapsae has been shown to be effective against DBM[4]. A single species of bacterium in the family Enterobacteriaceae was present in the anterior region of the IJs of S. carpocapsae[5]. Once an IJ has penetrated into the host haemocoele, the bacterial symbiont is released from the IJ gut, septicemia becomes established and insect death occurs within 48 h. The bacteria reproduce rapidly; the IJs feed on the bacteria and develop 2-3 generations and then new IJs leave the insect cadaver to search for new hosts. These nematodes carry a specific bacterium of genus Xenorhabdus which has shown a lethal effectiveness.
Temperature also has a profound effect on mobility, reproduction and development of nematodes[6]. Production and infectivity of S. carpocapsae was better achieved at 25°C in both DBM and Galleria[7-9]. However, the efficiency of S. feltiae nematodes against carrot weevil (Listronotus oregonensis) was unaffected by temperatures (15-25°C)[10].
Entomopathogenic nematodes have emerged as excellent candidates for biological control of insect pests. Attributes making the nematodes ideal biological insecticides include their broad host range, high virulence, safety for non target organisms and high efficacy in favourable habitats. Progress achieved in liquid fermentation, formulation stability and application strategy has allowed nematode-based products to become competitive with chemical insecticides in medium and high valued crops on the basis of cost/benefit ratio and ease of application[11]. One of the fundamental steps in development of an entomopathogenic nematode for biocontrol is choosing an appropriate strain. Virulence against the target pest is a basic factor of biological control programme.
Entomopathogenic nematodes have been shown to be pathogenic towards the larvae and pupae of DBM in the laboratory and in the field[4]. These nematodes are capable of seeking out and killing the insect pests in soil and foliar environment. Although these nematodes can penetrate, kill and reproduce in DBM larvae or pupae, a quantitative measure of infection has not been determined fully for isolates such as S. abbasi, S. karii and S. riobravis. The production, establishment and effectiveness of S. abbasi have been studied in the larvae of Galleria mellonella and it was suggested that this isolate could be mass produced and used against this pest[12]. However, in present study a small sized DBM host was used to determine the production of IJs of four steinernematid strains. The aim of this study was therefore to identify the DBM larvae or pupal stage infection and at which stage a virulent tropical nematode isolate is producing the more number of IJs and causing a significant role for infectivity at different temperature.
MATERIALS AND METHODS
DBM was reared on Chinese cabbage cv. Wong Bok in a growth room at 25°C in the Department of Agriculture, University of Reading. S. carpocapsae (All isolates of UK) were obtained from by Biosys, USA while S. karii, S. abbasi and S. riobravis, nematodes IJs suspensions were supplied by CAB Institute of Parasitology, St. Albans, UK. These isolates were cultured in the greater wax moth, G. mellonella at 28°C. S. carpocapsae is a temperate entomophathogenic nematode therefore it was stored at 7°C while all others are tropical isolates hence were stored at 15°C. The fresh IJs were used within one week of harvesting from white traps using the techniques described by Woodring and Kaya[13].
Experiment 1. Effects of temperature on production of infective juveniles in DBM larvae and pupae: The production of IJs of all four isolates in DBM larvae and pupae was investigated at 25 and 30°C. Single last instar larvae and single pupae of DBM (same size and weight) were infected with 100 IJs in multi-well dishes with 12 cells (2.5 cm in diameter and 2.0 cm in depth) filled with eight gram of moist autoclaved sand (14%MC). In this experiment IJs were applied to each cell, multi-well dishes were sealed with paraflim to avoid desiccation and placed in incubator at 25 and 30°C. After 5 days, each larvae and pupae were transferred to separate white trap and the number of emerging IJs were counted after every alternative day until there was no further increase in numbers. Replication was 12 fold.
Experiment 2. Effects of temperature on infectivity of DBM larvae and pupae: Infectivity of four entomopathogenic nematodes to DBM larvae and pupae was determined at two different temperatures using sand-based assay[14]. Single fourth instar of DBM larvae and pupae (with and without cocoon) of same age, size and weight were infected with 100 IJs using sand media in the same manner as already described in previous experiment. Multi-well dishes were incubated at 25 and 30°C. After 2 days, larvae and pupae were transferred to separate petri dishes containing Ringer solution [12] and were then dissected to count total number of penetrated IJs. Replication was 12 fold. Data taken from both experiments were statistically analysed using regression and t-test techniques of GENSTAT-5, Release 4.2 (Lawes Agricultural Trust, Rothamsted Experimental Station, UK.).
RESULTS
Production of nematodes (IJs) in DBM larvae and pupae: Temperature has a significant (P<0.05) effect on the establishment of all nematodes in DBM larvae (Fig. 1A and B) and pupae (Fig. 1C and D). However, all isolates studies indicated a much significance difference between the infected DBM larvae and pupae, as the number of IJs of all isolates were significantly higher in DBM larvae. Maximum number of IJs (1558) were produced by S. carpocapsae at 25°C whereas at 30°C, 1460 IJs were counted after 45 days, when DBM larvae was used as host. Other three nematodes (S. abbasi, S. karii and S. riobravis) produced higher number of IJs at 30°C when infected DBM larvae. S. abbasi produced 1248 IJs at 30°C, whereas at 25°C these were 592 after 45 days. However, the difference in number of IJs produced by S. karii and S. riobravis was less but significant between 25 and 30°C, S. karii produced 415 and 532 IJs whereas S. riobravis produced 396 and 442 IJs at 25 and 30°C, respectively (Fig. 1A and B). S. carpocapsae again produced maximum number of IJs (678) after 30 days of infection in DBM pupae at 25°C whereas at 30°C number of IJs were counted as 514. S. abbasi produced maximum IJs (717) as compared to all three isolates at 30°C. The same isolates produced 412 IJs at 25°C. Similarly, S. karii produced 482 and 402 IJs whereas S. riobravis produced 425 and 355 IJs at 30 and 25°C, respectively (Fig. 1C and D).
Infectivity of nematodes (IJs) in DBM larvae and pupae: S. carpocapsae was found significantly (P<0.05) virulent and appeared to be more infective at 25°C in DBM larvae whereas S. abbasi, S. Karii and S. riobravis did best at 30°C (Fig. 2A). About 28% IJs of S. carpocapsae succeeded to penetrate into the body of DBM larvae at 25°C whereas at 30°C only 23% IJs recovered from insect body.
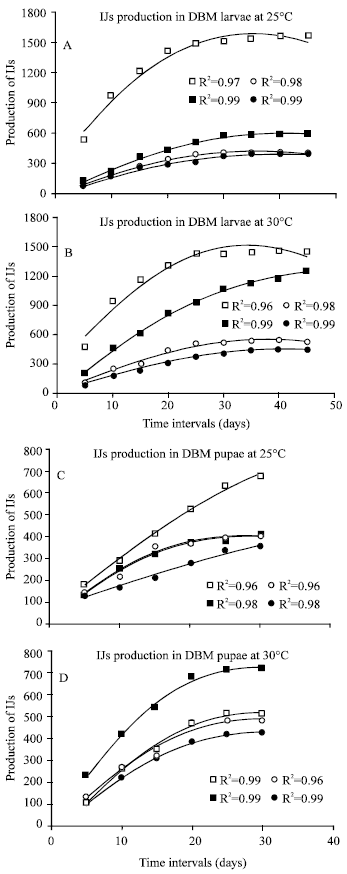 | |
| Fig. 1: | The number of infective juveniles of Steinernema carpocapsae (□), S. abbasi (■), S. karii (ο) and S. riobravis (●) produced in DBM larvae (A and B) and pupae (C and D) at 25 and 30°C temperatures |
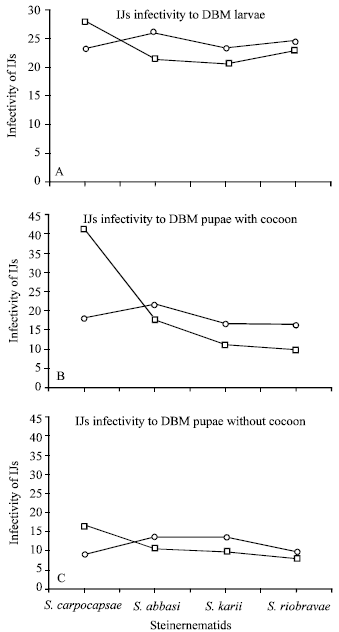 | |
| Fig. 2: | The number of infective juveniles of Steinernematids penetrated into the body of a single DBM larvae A) or pupae, with B) and without C) cocoon at 25 (□) and 30°C (ο) temperatures. Graphs depicted the average number of IJs from 12 DBM larvae or pupae while 100 IJs were introduced in the sand based media |
Similarly, 26% IJs of S. abbasi were penetrated into DBM larvae followed by S. riobravis (25%) and S. karii (23%) at 30°C. However, a non-significant difference (P<0.05) was observed between the two temperatures when last three tropical isolates were used.
A significant (P<0.05) difference between DBM pupae with and without cocoon was observed in respect of number of IJs recovered at 25 and 30°C (Fig. 2B and C). About 41% IJs of S. carpocapsae were entered into the insect pupae with cocoon at 25°C whereas the IJs of same species were counted as 18% in the same insect pupae at 30°C (Fig. 2B). A dramatic and significant (P<0.05) decrease was observed in S. carpocapsae IJs when they infected DBM pupae without cocoon at both temperatures i.e. 17 and 10% at 25 and 30°C, respectively. Other species such as S. abbasi, S. Karii and S. riobravis also recovered reasonable percentage of respective IJs (18, 12 and 10%, respectively) when they were introduced to DBM pupae with cocoon at 30°C. A significant (P<0.05) decrease in the number of IJs of all four nematodes was observed when they were recovered from DBM pupae without cocoon at both temperatures (Fig. 2C).
DISCUSSION
Production and infectivity is a useful guide to know the biological study of any entomopathogenic nematode when they are produced commercially for the control of insect pest. For a good biocontrol agent it is necessary to investigate their physiological, ecological and behavioral response to different insect pest. In this research it was observed that all four strains of tropical entomopathogenic nematodes were infective in DBM larvae and pupae but respond differently to temperature as it affects their mobility, reproduction and development[6].
In the present study, 25°C temperature was found effective for the production and infectivity of S. carpocapsae in DBM larvae and pupae however 30°C was found appropriate for all others (S. abbasi, S. karii and S. riobravis). Similar results have been observed when G. mellonella was used as host for the production and infectivity of S. carpocapsae[7,8], S. abbasi and S. riobravis[12]. However, when the production rate of S. carpocapsae juveniles in DBM larvae was compared to G. mellonella, it was about 1/50 of the number produced in former one at 25°C. This difference is because of the smaller size of DBM as compared to Galleria. The juveniles of S. carpocapsae established in DBM larvae were also found most virulent for this pest and killed its larvae within 6 h[4]. It has been reported previously that DBM has got resistant to B. thuringiensis therefore stains of S. carpocapsae can be used in integrated pest management programme to control this pest[15].
The results of present experiments suggested that DBM larvae and pupae can be infested by all these tropical nematodes at temperatures between 25 and 30°C, which is the temperature range of most tropical cropping pattern. Moist conditions of foliage can increase the effectiveness of these nematodes against DBM larvae and pupae when applied in tropical environment. It is possible that these tropical entomopathogenic nematode species could be established against the DBM larval or pupal stage to reduce the pest damage in field conditions. The nematode S. carpocapsae is exempt from pesticide regulation but it seems that all other new tropical isolates of entomopathogenic nematodes will have to be tested thoroughly in other environmental conditions before they are used as a biopesticide.
REFERENCES
- Talekar, N.S. and A.M. Shelton, 1993. Biology, ecology and management of the diamondback moth. Annu. Rev. Entomol., 38: 275-301.
CrossRefDirect Link - Kaya, H.K. and R. Gaugler, 1993. Entomopathogenic nematodes. Annu. Rev. Entomol., 38: 181-206.
CrossRefDirect Link - Baur, M.E., H.K. Kaya, B.E. Tabashnik and C.F. Chilcutt, 1998. Suppression of diamondback moth (Lepidoptera: Plutellidae) with an entomopathogenic nematode (Rhabditida: Steinernematidae) and Bacillus thuringiensis Berliner. J. Econ. Entomol., 91: 1089-1095.
Direct Link








