ABSTRACT
The invitro antimicrobial activity of Nerium oleander roots bark and leaf extracts were studied against Bacillus pumilus, Bacillus subtilis, Staphylococcus aureus, Escherichia coli and Aspergillus niger. The chloroform, ethanol and methanol extracts of Nerium oleander showed high activity against all the tested bacteria. None of the crude extracts of the selected plant exhibited activity against Aspergillus niger. These results were compared with the Zones of inhibition produced by commercially available standard antibiotics. The inhibitory effects of extracts are very close and identical in magnitude and are comparable with the standard antibiotics used.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.177.180
URL: https://scialert.net/abstract/?doi=ajps.2004.177.180
INTRODUCTION
Nerium oleander Linn. Belongs to family Apocynaceae commonly known as Gandeera, is a large glabrous evergreen shrub with milky juice. Leaves in threes, shortly stalked, coriaceous, 10-15 cm long, linear-lanceolate, acuminate, tapering into the short, dark green and shining above, midrib stout; nerves numerous, spreading horizontally. Flowers are rose-coloured or white, fragrant. Calyx-lobes lanceolate. Corolla 3.8 cm. diam; fragrant, lobes rounded. Filaments hairy, appendages of anthers twice as long as the cells. Follicles 15-23 cm long, rigid, at length separating. Seeds about 13 cm long, tipped with a coma of light brown hairs, distributed upper gangetic plain, Himalaya from Nepal westward to Kashmir upto 6,500 ft., salt range, Waziristan, central and South India. Extensively cultivated throughout the greater part of India as well as in China and Japan.
The root is bitter; aphrodisiac, tonic good for chronic pain in the abdomen and pain in the joints, very poisonous, but an antidote to snake-venom.
The roots, beaten into a paste with water, is recommended to be applied to chaneres and ulcers on the penis. The juice of the young leaves is poured into eyes in opthalmia with copious lachrymation. The bark of the roots and the sweet-smelling leaves of this shrub are considered by vydians as powerful repellents, applied externally. The root itself, taken internally, acts as poison, and is but too often resorted to for the purpose of self-destruction, by the Hindoo women, when tormented with jealousy[1]. The present study was undertaken to evaluate the leaves and roots bark for their possible anti-microbial activity.
The importance, necessity and potentiality of medicinal plants in the practice of medicine today is well established and cannot be over looked. The Indo. Pak subcontinent is very rich in having resources of medicinal plants. A large number of these plants are used in the form of powder, decoction and infusion for the treatment of various diseases including the infection caused by microbes with fair amount of success by Hakims and Vaids. Several workers throughout the world have carried out antimicrobial studies on some medicinal plants including Betula pendula[2]. Ageratum houstonianum[3]. Literature citations reveal that A. houstonianum has shown wide range of antimicrobial activity, more or less comparable to Melia azadirachta. The other potential source is the genus Sternbergia from the family Amaryllidaceae containing Amaryllidaceae alkaloids, having antimicrobial activities. Amongst such alkaloids which are used as preparation in medicinal treatment are lycorine, an abundantly obtained alkaloid, lycorine has a high antiviral effect on DNA and RNA viruses, this effect occurs soon after lycorine stops protein synthesis at peptide tie step. It has been observed that lycorine affects in this way poliomyelitus, coxsackine and Herpes simplex type I virus[4-6]. It has been reported that lycorine extract has an antibacterial effect, the antimycotic effect of Azadirachta indica on Candida albicans has been carried out in different parts of the world[7-10]. From the above discussion we see that the plants can be a very rich source of potential antimicrobial drugs, that can be used for the treatment and cure the various ailments.
MATERIALS AND METHODS
Plant: The drug plant Nerium oleader was procured from Goi District Kotli (A.K) during the months of May-June (2002) and was indentified at the Department of Botany, University of Azad Jammu and Kashmir, Muzaffarabad. The root bark and leaves were dried under shade, coarsely powdered and subjected to cold chloroformic, ethanolic and methanolic extraction, the extraction was carried out by maceration for 10 days in each solvent at room temperature (25±2°C). The solvent extracted material was filtered. These extracts were preserved until required.
Culture media: The media used for Bacteria was Nutrient agar and for fungus was sabourands dextrose agar. The test organisms used included Bacillus pumilus, Bacilllus subtilis, Staphyloccus aureus, Escherichia coli and a fungus Aspergillus niger.
Anti microbial activity: The extracts obtained from leaves and roots were studied for antimicrobial activity. The antimicrobial study of chloroform, ethanol and methanol extracts were assayed by using Disc Diffusion method[11]. Filter paper discs of 16 mm diameter were soaked with 0.1ml of extracts. The petri dishes were sterilized in an oven at 20°C for 2 h. The sterilized petri dishes were labeled with the bacterial and fungal names and were also labeled with the root, leaves and the solvents used. Each of the petri dish was marked and inner content was divided into four equal parts. The sterilized test tubes were used for the preparation of dilution which were labeled with the names of the micro-organisms used. 10 ml of distilled water was taken in each test tube. Then a loop of bacterium or fungus was inoculated in distilled water in test tube under aseptic conditions. 1ml of inoculum was transferred from the test tube into the corresponding previously labeled petri dishes. The sterilized Nutrient agar medium was poured into the perti dishes containing bacterial suspension. The sabourands dextrose agar medium was poured in the petri dishes containing fungal suspension. All the petri dishes were gently rotated and media were allowed to solidify at room temperature. The filter paper discs which were soaked with extracts were placed in petri dishes at their labed positions. Another set of petri dishes were prepared in the same way in which three different commercially available antibiotic discs i.e. Amoxicilline 10 μg, cephalexim 30 μg and tetracycline 30 μg were placed on the top of the medium.
Incubation of plates: The plates containing the bacterial culture were incubated 37°C for 24 h. On the other hand, the plates with fungal suspension were incubated at 25°C for 72 h. After the incubation time, all the plates were examined for the presence of zones of inhibition as a property of antimicrobial activity.
RESULTS
The extracts from 2 parts (root and leaves) of the plant were used in the present study to investigate their antimicrobial potential. Both gram-negative and Gram-postive Bacteria and a fungus were used. The results and screening of antimicrobial activity of Nerium oleander roots bark and leaf extracts are summarized in (Table 1-6).
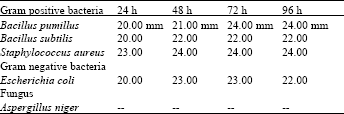
The ethanolic extract of leaves of Nerium oleander showed high antimicrobial activity against all the tested micro-organisms except Aspergillus niger. The results obtained show that the ethanolic extract of the roots of Nerium oleander exhibited moderate activity against Bacillus pumillus and staphylococcus aureus while with Excherichia coli it was high whereas against Bacillus subtilis low activity was observed (Table 5).
| Table 1: | Chloroformic extract of leaves zones of inhibition (mm) after micro-organisms |
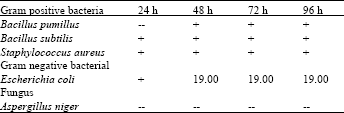 | |
| Table 2: | Ethanolic extract of leaves |
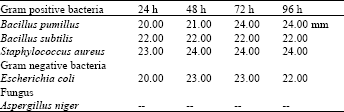 | |
| Table 3: | Methanolic extract of leaves |
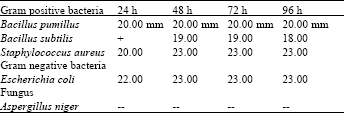 | |
| Table 4: | Chloroform extract of roots zones of inhibition (mm) after micro-organisms |
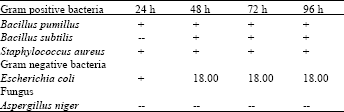 | |
| Table 5: | Ethanolic extract of roots |
 | |
| Table 6: | Methanolic extract of roots |
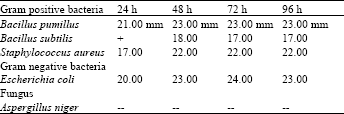 | |
In case of the methanolic extracts of Nerium oleander leaves also showed high activity against staphylococus aureus and E. coli, while it showed moderate activity against Bacillus pumilus and Bacillus subtilis (Table 3). The methanolic extracts of Nerium oleander roots revealed marked activity against all the bacteria used. None of the crude extracts showed activity against Aspertillus niger (Table 6).
On the other hand the chloroformic extracts of leaves (Table 1) and roots (Table 4) of Nerium oleander did not show any appreciable activity against any of the microbes used in the present investigation.
Similar findings also have been made by Mehta et al.[12] against S. typhii, S. dysenteriae, S. aureus, K. aerogenus, V. cholera etc.
DISCUSSION
The presence of antifungal and antibacterial substances in the higher plants is well established[13-18]. Plants have provided a source of inspiration for novel drug compounds as plants derived medicines have made significant contribution towards human health. Phytomedicines can be used for the treatment of diseases as is done in case of Unani and Ayurvedic system of medicines or it can be the base for the development of a medicine, a natural blueprint for the development of new drugs[19]. Much of the exploration and utilization of natural products as antimicrobial arise from microbial sources. Through soil micro-organisms or fungi produce most of the clinically used antibiotics, higher plants can be a very good source of antibiotics Firdous et al.[20] studied antibacterial activity of Adhatoda vasica, Calotropis procera, Nerium odorum and Ocimum sanctum leaf on certain gram positive and gram negative bacteria. Present study was conducted to investigate the antimicrobial potential of Nerium oleander leaves and roots. The results obtained are encouraging as the methanolic, ethanolic and chloroformic extracts have shown considerable antimicrobial activity. The antibacterial activity of the plant is appreciable, considering the importance of the microorganisms in infections. Further work is needed to isolate the active principle from the plant extracts and to carry out Pharmaceutical studies.
The potential for developing antimicrobials from higher plants appears rewarding as it will lead to the development of a phytomedicine to act against microbes. Plant based antimicrobials represents a vast untapped source for medicine. Plant based antimicrobials have enormous therapeutical potential as they can serve the purpose without any side effects that are often associated with synthetic antimicrobials. Continued further exploration of plant derived antimicrobials is needed today.
REFERENCES
- Didry, N., L. Dubreuil and M. Pinkas, 1994. Activity of anthraquinonic and naphthoquinonic compounds on oral bacteria. Pharmazei, 49: 681-683.
PubMed - Mehta, B., K. Shitut and H. Wankhade, 1993. In vitro antimicrobial efficacy of Triphala. Fitoterapia, 64: 371-372.
Direct Link - Nickell, L.G., 1959. Antimicrobial activity of vascular plants. Econ. Bot., 13: 281-318.
CrossRefDirect Link - Bhakuni, D.S., M.L. Dhar, M.M. Dhar, B.N. Dhawan and B.N. Mehrotra, 1969. Screening of Indian plants for biological activity: Part II. Indian J. Exp. Biol., 7: 250-262.
Direct Link - Fridous, A.J., S.N.L.M. Islam and A.B.M. Faruque, 1990. Antimicrobial activity of the leaves of Adhatoda vasica, Calotropis gigantea, Nerium odorum and Ocimum sanctum. Bang. J. Bot., 19: 227-229.
Direct Link - Dhar, M.L., M.M. Dhar, B.N. Dhawan, B.N. Mehrotra and C. Ray, 1968. Screening of Indian plants for biological activity: I. Indian J. Exp. Biol., 6: 232-247.
PubMedDirect Link








