ABSTRACT
Seeds of onion (Allium cepa L.) cv. Ailsa Craig were irradiated at various doses of gamma radiation (0, 10, 20, 40, 80 and 100 krad) and either tested immediately or stored for 24 h at 25oC before testing. Seed viability, final germination percentage, germination speed and seedling growth decreased, while electrical conductivity (EC) of seed leachates, time to complete 50% germination (T50), and percentage of abnormal seedlings increased with increase in radiation dose. After 24 h of storage, percentage of abnormal seedlings increased significantly, while root and overall seedling lengths were decreased. However, seed viability, EC of seed leachates, seed germination percentage, germination speed, T50 and shoot growth were unaffected by the storage. It was concluded that after storage, the damaging effect of irradiation was increased due to the continued effect of radiation.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2003.702.707
URL: https://scialert.net/abstract/?doi=ajps.2003.702.707
INTRODUCTION
Gamma radiation has been widely used to get mutants in crop plants to use as a source of variability in breeding/improvement programs (e.g. Anjum et al., 1990; Al-Safadi and Simon, 1996; Kumar and Chaudhary, 1996; Munishamanna et al., 1998). The effects of radiation on seed may continue for some time after the period of exposure. Holding seeds for a time after irradiation, and before germinating them, has long been known to increase the amount of damage caused by the action of radiation (Curtis et al., 1958). This extra damage has been called "indirect" radiation damage, and is considered to be due to the continued activity of the free radicals produced by radiolysis of water (Conger, 1961). The amount of damage has been shown to increase with time in post-irradiation storage. However, Mishra and Raghuvanshi (1988) suggested that storage (6 months) of Trigonella foenum-graecum seeds after gamma irradiation might lead to genetic repairing. Latha and Nair (1999) exposed the seeds of barley cv. IB-65 with moisture content (m.c.) of 4 or 10% to gamma radiation. They found that with the increase in radiation dose, there was an almost linear increase in radiation damage (seedling damage and chromosomal aberrations). Storage of seed at 23% m.c. potentiated the radiation-induced damage,while storage at 33% m.c. resulted in recovery from radiation damage.
Hydration-dehydration pre-treatment of stored seeds greatly reduce their physiological deterioration under both accelerated and "natural" ageing conditions (Basu, 1976). These treatments have also shown significant radio protective actions in wheat (Dasgupta et al., 1977), and jute (Basu and Dasgupta, 1978). Punjabi and Basu (1982) stated that hydration-dehydration of seeds, before or soon after irradiation, considerably minimized the adverse effect of irradiation on the fall in germinability, and especially in the large reduction of root growth found in seedlings from irradiated seeds. In the present investigation, no hydration-dehydration treatment was used and short-term storage for only 24-hour was used after irradiation. Therefore, half of the seed samples were tested immediately after irradiation and other were stored at 25°C for 24 h in preparation to observe the continued effects of gamma radiation. The aim of the present study was to observe the direct effect of the absorption of radiation energy by the organic molecular components of the seed and the indirect effects of ions and free radicals produced in the radiolysis of water, by the addition of a short period of post-irradiation holding of the seeds before testing.
MATERIALS AND METHODS
Seed source and irradiation: Onion (Allium cepa L.) cv. Ailsa Craig seeds were obtained from Booker Seeds Ltd., Sleaford, England, divided into smaller batches and stored in closed glass bottles at 5°C. Samples of seeds were taken out from cold store (5°C) and still in their air-tight glass bottles, kept overnight to equilibrate fully with laboratory temperatures before irradiation. Then the seeds were transferred to test tubes and exposed to various doses of gamma radiation using a 60Co source for a time corresponding to the required dose. The doses of gamma radiation employed were 10, 20, 40, 80 and 100 krad.
Tetrazolium viability test: Four replicates of 50 seeds in each treatment were soaked in distilled water for 18-20 h. Each seed was cut longitudinally without completely separating into two halves. The seeds were submerged in 1% 2,3,5-triphenyl tetrazolium chloride solution for at least 8 h at 35°C in darkness, after which the staining patterns were recorded.
Electrical conductivity test: Four replicates of 100 whole seeds (i.e., broken seeds removed) were weighed in each treatment and placed in 100 ml beakers separately, each containing 75 ml distilled water. The seeds were gently stirred to ensure that all seeds were completely immersed and evenly distributed. The beakers were placed in an incubator at 20 ± 2°C. After 24 h the seeds were gently stirred and the conductivity of the each soaking water was measured without filtration by using a digital conductivity meter (JENWAY, Model 4070). The conductivity per gram of seed weight was calculated and recorded.
Germination tests: Germination percentages, using 4 replicates of 50 seeds, were determined by placing the seed samples in 90 mm Petri dishes on filter papers (Whatman No. 1) moistened with 4 ml of distilled water. Seeds were distributed evenly within each dish. Each Petri dish was covered with its lid and placed in an incubator, maintained at 20 ± 2°C and adjusted to give 12 h of fluorescent light illumination alternating with 12 h of darkness. Each Petri dish was watered daily according to its requirement. Germination in terms of radicle emergence (at least 2 mm) was assessed each day from the second day until no further radicle emergence was noted on two successive days. Germination capacity was expressed as a percentage of all seeds with fully emerged radicle in a given batch.
The average germination speeds are derived from the formula of Kotowski (1926).
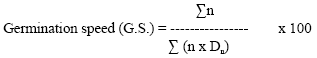 |
Where, n is the number of seedlings germinated on day Dn. Dn is the number of days from sowing; corresponding to n, and highest (G.S.) is the greatest speed.
Time to reach 50% of final germination (T50) was calculated from the formula of Coolbear et al. (1984).
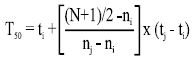 |
Where, ni < (N+1)/2 < nj. N is a number of seeds germinated, while ni and nj are total number of seeds germinated by adjacent counts at times ti and tj, respectively.
Morphology of seedlings: Morphological study was carried out when seedlings were 10 days old. Those seedlings classed as "normal" were recorded, following the guidelines given by the International Rules for Seed Testing (ISTA, 1985). Seedlings are classed as abnormal when one or more of the essential structures fails to develop normally because of previous damage to the embryo, or when development as a whole is weak or out of proportion compared with that of a normal seedling germinated at the same time and in the same conditions. Abnormal seedlings were further examined for the types of abnormalities.
Seedling growth: After 10 days from the initiation of imbibition, average shoot lengths, root lengths, and overall seedling lengths were measured using a ruler. The data are reported in terms of the effects of radiation doses and storage treatment on seedlings, i.e. the percentage change/ reduction in mean seedling height as compared to the un-irradiated and not subjected to storage treatment (control), as calculated from the following formula:
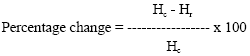 |
Where, Hc is the mean seedling height of the control and Hr is the mean seedling height after irradiation or storage treatment or combination of both.
Statistical analysis: The data were analysed statistically using Fisher’s analysis of variance technique in a completely randomised design with factorial arrangements. The treatment means were compared using Duncan’s multiple range test at 5% probability level (Petersen, 1994).
RESULTS AND DISCUSSION
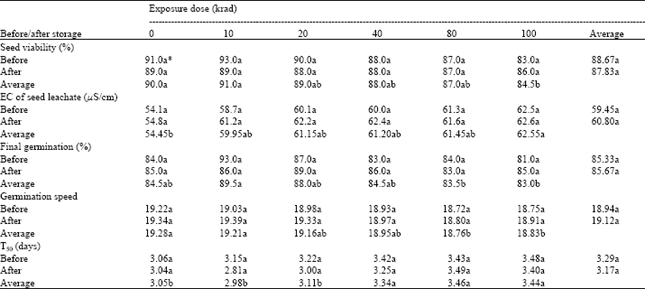
Seed viability: Radiation doses significantly affected the viability of the seeds. However, storage treatment alone and the interaction between radiation doses and storage treatment have no significant influence on seed viability. The higher viabilities were recorded in un-irradiated seeds and in those exposed to lower doses of gamma radiation. The viability was significantly reduced, being minimum at the highest dose of radiation (Table 1).
| Table 1: | Effect of various doses of gamma radiation and 24 h storage at 25°C on viability, electrical conductivity of leachates and germination potential of onion seeds |
 | |
| Table 2: | Effect of various doses of gamma radiation and 24 h storage at 25 °C on percentages of seedling types from onion seeds |
 | |
| Table 3: | Effect of various doses of gamma radiation and 24 h storage at 25°C on seedling lengths and reduction in growth from onion seeds |
 | |
| *Means in a group sharing similar letters are non-significant at P = 0.05 (DMR test). The groups have been separated by normal, normal bold and italic bold fonts. | |
These results suggest that irradiation has some damaging effects on seed resulting in reduced viability.
Conductivity of seed soak water: Conductivity of seed leachates was also affected significantly by the radiation doses. As the radiation dose was increased, EC of seed leachate was also increased. The lowest EC was recorded in the leachate of the seeds, which were not subjected to irradiation treatment, while the highest being in the leachates of those irradiated at the highest dose of gamma radiation (Table 1). This indicates that the radiation is not only causing the genetic damage but also some physical damage to the seed structure resulting more solute loss. These results are also in line with the results of tetrozolium seed viability test. When conductivity after the storage treatment is compared with the results of the conductivity test carried out immediately after irradiation, however, there again appears to have been no increase in damage inflicted by the storage treatment. The interaction between radiation doses and storage treatment was also found non-significant, indicating no impact on solute loss.
Germination potential: Gamma irradiation had a significant influence on germination potential of the seeds. Final seed germination (%) was significantly decreased at higher doses (Table 1). This was confirmed by the tetrazolium viability and conductivity tests, which show a similar response. These results indicate that the higher doses of gamma radiation have adverse effect on seed germination. The inhibition of seed germination at higher doses of radiation may have resulted from damage to chromosomes and subsequent mitotic retardation (Al-Safadi and Simon, 1996). These results are also in accordance with the findings of previous workers (Ripa and Audrina, 1993; Kumar and Chaudhary, 1996; Agarwal and Kaul, 1998; Munishimanna et al., 1998), who reported that seed germination percentage decreased in different crops with increase in radiation dose. Further, in the present study, when the final germination percentage after post-irradiation storage is compared with germination percentage of those seeds tested immediately after irradiation, it can be seen that the amount of damage was not increased by the addition of the 24-hour storage period after irradiation.
Germination speed also decreased with an increase in radiation dose (Table 1). However, time to complete 50 % germination was increased with increase in radiation dose. Un-irradiated seed took the minimum time to complete 50 % germination and showed the highest speed of germination. Minimum speed of germination with maximum time to complete 50 % germination was recorded in seeds irradiated at higher doses (80 and 100 krad). This indicates that gamma irradiation interferes with germination process. Higher doses of radiation have inhibitory effect by reducing the rate of cell division. Similar findings have been reported by several workers (Ripa and Audrina, 1993; Al-Safadi and Simon, 1996), who found that higher doses of radiation delayed seed germination.
Seed storage treatment had no pronounced effect on germination speed and time to complete 50 % germination. Further, there are also no significant differences either in speed of germination, or in the time taken to reach 50% of final germination when the interaction between radiation doses and storage treatment was examined (Table 1). Several reports e.g. Kumar (1978), Kawamura et al. (1992) have found that germination potential is affected to a lesser extent by irradiation than other measurements of damage, such as seedling height, but no mention has been found about the effect of post-irradiation storage.
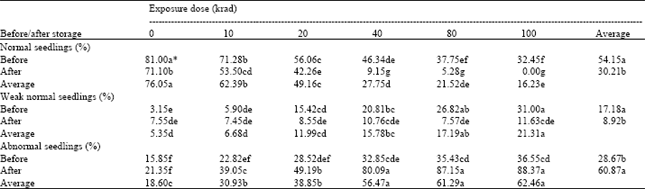
Seedling abnormality: Numbers of normal, week normal and abnormal seedlings were affected significantly by all the factors, i.e. radiation doses, storage treatment and interaction of the both factors (Table 2). Number of normal seedlings decreased while numbers of week normal and abnormal seedlings increased with an increased in radiation dose. This suggests that the genetic material has been affected by the radiation doses resulting in altered morphology. Similar findings have been reported in maize (Viccini and Carvalho, 2001) and buckwheat (Alekseeva and Rarok, 1995).
Seed storage resulted in decreased numbers of normal and weak normal seedlings and increased number of abnormal seedlings (Table 2). This is interesting to record that the radiation treatment increased the number of weak normal seedlings while storage treatment decreased the number. The possible reason for decrease of weak normal seedlings could that more seedlings became abnormal after storage treatment.
A perusal of interaction means indicates that the maximum number of normal and minimum numbers of weak normal and abnormal seedlings were recorded in control (un-irradiated and not subjected to storage) followed by those seeds which were un-irradiated and only subjected to storage and those irradiated at the lowest dose (10 krad) and not subjected to storage. Seed irradiation at 10 krad and storage treatment alone were almost at par. But it does not mean that both the treatments are similar in action. The minimum number of normal seedlings (0 %) and maximum number of abnormal seedlings (88.37 %) were recorded when seeds were irradiated at the highest dose of 100 krad and subjected to storage treatment (Table 2). If the numbers of abnormal seedlings are compared between irradiation alone and irradiation followed by a storage period, the un-irradiated controls produced roughly the same number of abnormal seedlings in each case (15 - 22%). When irradiated, increases in the numbers of abnormal seedlings occurred with increase in radiation dose whether the seeds were held for a post-irradiation storage period or not. However, it can also be seen that very large apparent increases in damage were observed when seeds had been held for a storage period following irradiation. These increases were especially large in the higher range of doses. However, where such large percentages of abnormal seedlings are recorded it must be pointed out that these figures are obtained from a very small surviving fraction of the population, and therefore, represent a very great deal of damage done to the original seed sample from the point of view of their nuclear organization. The results show that the number of abnormalities produced depends on the level of radiation dose and the seed storage enhanced the abnormalities due to the continued effect of radiation.
The abnormal seedlings were further carefully examined for their types and the following recognizable categories of abnormal seedlings were found;
| a) | without primary root |
| b) | primary root short and stunted |
| c) | primary root short and weak or spindly |
| d) | poorly developed leaf-like cotyledon without a definite bend or "knee" and |
| e) | short and weak, or spindly, or watery seedling |
These results indicate that the radiation damage is random and any genome can be affected, therefore, resulting in various types of abnormalities.
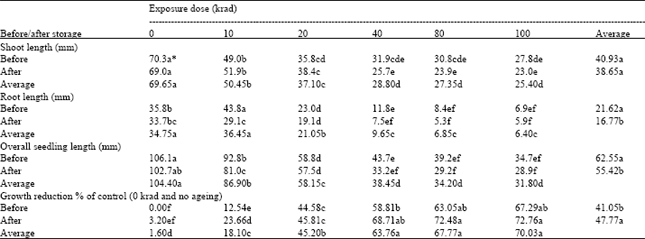
Seedling length: Shoot length was not influenced by the storage treatment. However, it was significantly affected by the radiation doses and their interaction with storage. As the radiation dose increased from 0 to 40 krad, shoot length decreased significantly. After 40 krad no significant effect of radiation dose was found. As far as interaction between the two factors is concerned, maximum shoot lengths were recorded in seedlings resulting from the un-irradiated seeds, either subjected to storage treatment or not. As radiation dose increased, shoot length decreased. The minimum shoot length was recorded in the seedlings resulting from the seed irradiated at higher doses and subjected to storage treatment (Table 3).
Root length was significantly affected by all the three factors; radiation doses, storage treatment and their interaction. Root length decreased with the increase in radiation dose, especially up to 40 krad, showing the inhibiting effect of radiation administered. The storage treatment also resulted in a significant decrease in root length. The means of interaction show that the maximum root length was recorded in the seedlings resulted from the seed irradiated at the lower radiation dose of 10 krad and not subjected to storage treatment followed by the control (un-irradiated and not subjected to storage), but both of these differed significantly with each other. This suggests that the lowest dose of gamma radiation (10 krad) have stimulating effect on root growth. This might be attributed to an increase in auxin levels, resulting in enhanced root growth. Minimum root length was recorded when seed was irradiated at higher doses and subjected to storage treatment followed by those not subjected to storage treatment (Table 3).
Overall seedling length was also affected significantly by all the three factors, i.e. radiation doses, storage treatment and their interaction. Overall seedling length decreased significantly as the radiation dose increased up to 40 krad. However, after this dose, the response was non-significant. As root length was reduced significantly by the storage treatment, the overall seedling length was also decreased significantly. Regarding the interaction between two factors of study, the maximum seedling lengths were recorded in un-irradiated seed either subjected or not, to the storage treatment. Seedling lengths decreased with increase in radiation dose. The minimum seedling lengths were recorded in case of the seedlings resulting from the seeds exposed to higher doses of gamma radiation and subjected to storage treatment followed by those resulting from the seeds exposed to higher doses of gamma radiation but not subjected to storage treatment (Table 3).
As the data of growth reduction was derived from the data on overall seedling lengths, therefore, as the seedling lengths decreased, reduction percentage in growth increased. Measurements of seedling growth after post-irradiation storage show that reductions in height occurred with increase in radiation dose. When the un-irradiated control in this post-irradiation storage is compared with the control in the irradiation only (without storage), it can be seen that these grew to roughly the same height (approximately 102 to 106 mm). This is interesting to note that the radiation doses higher than 40 krad did not cause further reductions in seedling growth. Reduction in growth due to seed irradiation has already been recorded by different workers in various crops e.g. winged bean (Veeresh et al., 1995), barley (Aggarwal and Kaul, 1998) etc. The reduction in plant growth at higher doses of radiation has been explained by Srivastava and Tyagi (1986) on the basis of auxin destruction, changes in ascorbic acid content and physiological and biochemical disturbance.
By comparing the data of immediate testing after irradiation with that followed by post-irradiation storage, there appears to be no increases in damage shown by seed viability, germination potential or speed of germination. However, the storage treatment appeared to have significantly increased the numbers of abnormal seedlings and reduced seedling growth.
REFERENCES
- Al-Safadi, B. and P.W. Simon, 1996. Gamma irradiation-induced variation in carrots (Daucus carota L.). J. Am. Soc. Hortic. Sci., 121: 599-603.
Direct Link - Conger, A.D., 1961. Biological after-effect and long-lived free radicals in irradiated seeds. J. Cell. Comp. Physiol., 58: 27-32.
Direct Link - Coolbear, P., A. Francis and D. Grierson, 1984. The effect of low temperature pre-sowing treatment on the germination performance and membrane integrity of artificially aged tomato seeds. J. Exp. Bot., 35: 1609-1617.
Direct Link - Curtis, H.J., N. Delihas, R.S. Caldecott and C.F. Konzak, 1958. Modification of radiation damage in dormant seeds by storage. Radiat. Res., 8: 526-534.
Direct Link - Kawamura, Y., N. Suzuki, S. Uchiyama and Y. Saito, 1992. Germination test for identification of gamma-irradiated wheat. Radiat. Phys. Chem., 40: 17-22.
CrossRef - Mishra, K. and S.S. Raghuvanshi, 1989. Cytogenetic effects of gamma irradiated stored seeds of Trigonella foenum-graecum. Cytologia, 54: 33-36.
Direct Link - Punjabi, B. and R.N. Basu, 1982. Control of age-and irradiation-induced seed deterioration in lettuce (Lactuca sativa L.) by hydration-dehydration treatments. Proc. Indian Natl. Sci. Acad., 48: 242-250.
Direct Link








