ABSTRACT
The effects of 0, 2.5, 5 and 10 μ (Cd+2) [Cd( NO3)2x4H2O] and 0, 10, 25, 50 and 100 mM NaCl on certain parameters of growth and photosynthesis and different ionic contents in maize (Zea mays L. single cross) were studied. With increasing Cd+2 and NaCl alone to Hogland nutrient solution, the contents of chlorophyll (Chl) and starch content decreased. Interaction effect of salinity and cadmium caused decrease of negative effects of two stress and the results indicate that plants whose growth has been retarded by a mild level of one stress factor (in this case 25 and 50 mM NaCl) may become more tolerant to a second stress factor (Cd). The addition of cadmium caused a partial elimination of salinity effects on roots and shoots and growth parameters, ions accumulation and photosynthetic parameters were improved at mild concentrations of two stresses.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2003.196.201
URL: https://scialert.net/abstract/?doi=ajps.2003.196.201
INTRODUCTION
Salinity is one of the most sever problems in worldwide agricultural production, in arid and semi-arid regions; salinity is the major and usual abiotic stress for plants (Umezawa et al., 2000). On the other hand heavy metals are natural elements that are found at various high background levels at different places throughout the world due to various concentrations in the bedrock (Temmerman et al., 1984 and Forstner and Wittman, 1974). Many studies deals with effects of Cd+2 on the growth of crop plants. Inouhe et al. (1994) compared the growth responses of various monocot and dicot species to cadmium. Root growth, most dicot species was more severely inhibited at Cd treatments then monocot species. Under Cd stress leaf weights of strawberry plants were more reduced than root weights (Cieslinski et al., 1996). Increased Cd concentrations in shoot of Triticum plants under Cd stress were accompanied by declining concentrations of K, Ca, Mg and Fe (Ouzounidou et al., 1997).
Heavy metals usually decreased the total chl and chl a/b ratio in higher plants (Sheoran et al., 1990). Total soluble sugars and starch contents of soybean plants reduced with increasing of Cd concentration in different media (Ghorbanli et al., 1999). Responses of plants to salt stress have been studied using anatomical, ecological, physiological and molecular approaches (Hurkman, 1992). Ewing (1981) reported that the addition of NaCl to the nutrient solution caused a marked increase in soluble sugars contents, protein contents and decrease in growth and Chl contents. Several factors may contribute to the reduction in growth exhibited by plants under salinity stress. One significant factor may be related to inhibition of vascular tissue production under stress (Shininger, 1979 and Ewing, 1981). Plants undergo one or more forms of stress during various stages of their life cycle. Although most of the research has been focused on the responses of plants to single stress factors, plants in nature often encounter multiple stress factors whose interacting effect may be far from additive (Chapin et al., 1987). In some cases however, preconditioning to one stress factor may even increased the tolerance of plants to a different stress factor imposed simultaneously or at a letter time (King and Nelson, 1987; Mozafar and Oertli, 1990).
This study was thus undertaken to investigate the combined effects of salinity and cadmium on growth, ionic componts and photosynthesis of Zea mays.
MATERIALS AND METHODS
Seeds of Zea mays were germinated for 8 days in pots filled with LECA (light expanded clay aggregate) grains (03). Prior to potting, the LECA grains were washed with distilled water and pots were rinsed with distilled water everyday until germination. 8-day-old seedlings were transferred to 500 cm3 plastic containers (3 plants per container) with nutrient solution containing 5 mM KNO3, 5 mM Ca (NO3) 2x 4 H2O, 2 mM MgSO4 ,4 H2O, 1 mM KH2PO4, 0.09 mM NH4Fe (SO4)2 and micronutrients. The plants were treated with 0 (control), 2.5, 5 and 10 μ (Cd+2) with adding 0 (control), 10, 25, 50 and 100 mM NaCl for 8 days. All solutions were kept at pH 5.7 and aerated daily throughout the experimental period. The amount of water was adjusted daily using distilled water. The environmental conditions were 16 h photoperiod with irradiance of 700 Fmol m-2 s-1 (PAR) at 27±3 °C. The plants of three container sets (replicates), each container holding three 16-day-old plants were harvested for growth using the methods of Reynolds et al. (2001).
Chlorophylls were extracted with 80 % acetone under a dim green irradiation and determined spectrophotometrically (Arnon, 1949). The contents of total soluble sugars and starch were measured by the method of Kochert (1978). The contents of Na+, K+ were determined by flame photometer (Model 410, Sherwood Scientific Ltd company made in UK) and Ca+2, Mg+2 and Cd+2 were analyzed with an atomic absorption spectrophotometer (Spectra AA200, Varian). Some of the given results represent means ±SE and others due to their formula represent only one observation.
The results were analyzed with the SPSS (Version 10.0) statistical package.
RESULTS AND DISCUSSION
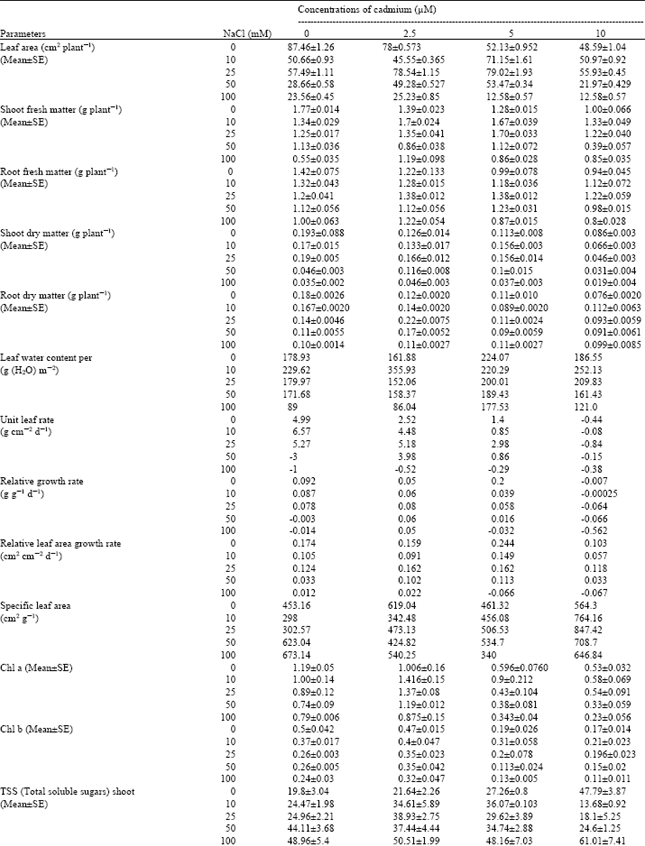
Analyses of data (Table 1) showed that salinity and cadmium affect all growth parameters alone but interaction of Cd and salinity positively affected growth rate and reduced their poisonous effects at some concentration on maize plants. With increasing of salinity, leaf area, shoot and root dry matter and shoot and root fresh matter of Zea mays plants decreased. The data indicated that increasing Cd had a negative effect on different parameters of growth. At moderate concentration of salinity (25, 50 mM NaCl) and Cd (2.5, 5μ Cd) different growth parameters were improved then other treatment but their contents were decreased then control (0) treatments. Some reports indicate that conditioning of plants to drought and salinity stress may render then more tolerant to later damage by environmental pollutants (King and Nelson, 1987 and McBirde, 1987). The data reported (Table 1) however, indicate that plants whose growth has been retarded by a mild level of one stress (in case 25 and 50 mM NaCl) may become more tolerant to a second stress (Cadmium). Thus, in mild level of salt resistant to Cd+2 increased and growth responses would be better, for example, unite leaf rate, relative growth rate and relative leaf area growth rate would be better than control and other treatments. Results showed that some adaptation mechanisms for coping with multiple environmental stress factors similar to those observed by King and Nelson (1987) may exist in maize plants in this study.
Chlorophylls contents in Zea mays plants were decreased at different treatments of salinity and cadmium but interaction effect of salinity and cadmium caused increasing in chlorophylls contents. Cadmium is certainly an effective inhibitor of plant metabolism, particularly of photosynthetic processes and chloroplast development in higher plants (Rascio et al., 1993). Effect of Cd+2 on chlorophyll and chloroplast development act synergistically and inhibit starch production (Stobart et al., 1985).
The contents of total soluble sugars were increased and starch content and unit leaf rate decreased with increasing of cadmium and salinity alone. Prado et al. (2000) believed that it was also possible that the high soluble sugars contents in Chenopodium quinoa at salt stress was due to a low Invertase activity under such conditions. Interaction effects of cadmium and salinity increased starch and unite leaf rate contents and improved photosynthesis via improvement of chlorophyll contents and affecting enzymes of photosynthetic carbon reduction cycle (Prasad, 1995).
Salinity also adversely affected water status of the plants (Reynold et al. 2001). On the other hand according to Hatch et al. (1988), Cd+2 influence transpiration more by affecting water flow through the roots than by changing stomatal aperture. These results were shown decreasing of water contents of plants at some treatments and increasing at other treatments.
Concentrations of Ca, Mg and K in treated plants were decreased by salinity and cadmium alone, an observation was also made by Lynch and Lauchli (1985) and Ouzounidou et al. (1997). Subjecting the plants to cadmium treatments however caused a lesser reduction of cations at most intermediate salinity levels. With increasing of salinity in cultured medium, the absorption of Na was increased significantly in shoots and roots of maize plants but accumulation of Na in roots more than shoot. Salinity induced Na-transporters in roots more than shoots (Hasegawa et al., 2000). Increasing of cadmium in culture solutions reduced others cations contents such as Ca and Mg. The reduction of other cations absorption by plants in presence of cadmium is due to competition between cadmium and other cations in cultured solution (Karez et al., 1990). Ehret et al. (1990) believed that despite tendency for salt exclusion, the mineral nutrition of none halophytes in influence by the presence of salt, often as a consequence of ion interactions and salinity induced low calcium levels in plants or calcium deficiency in none halophytic plants.
| Table 1: | The interaction of Cd (μM) and NaCl (mM) treatments on different parameters of Zea mays |
 | |
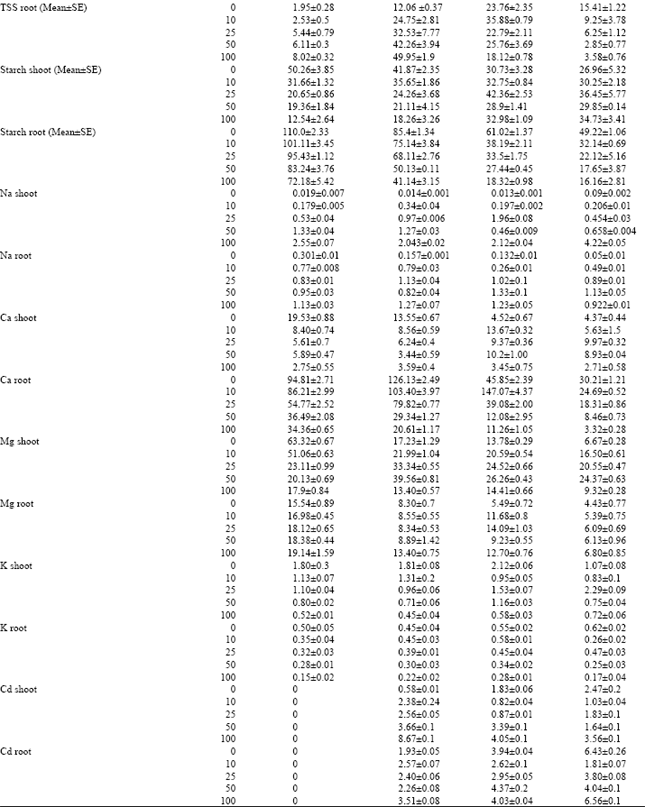 | |
The cadmium uptake by maize plants was decreased by also increasing of salinity. It has been shown in water culture that when increasing the salinity the uptake of cadmium in plants decreased (Greger et al., 1995). This may be due to CdClx complexes were formed in the water and these complexes are hard for the plant to take up (Prasad and Hagemeyer, 1999). For this reason, uptake of cadmium with increasing of salinity decreased and from the other hand the negative effects of salinity on uptake of cations such as Ca, Mg, K and Na, on different parameters of growth, sugars and chlorophyll contents were decreased and improvement in responses (Table 1).
In conclusion, the presence of cadmium in the nutrient solution with salinity affected the responses of plants. At mild levels of cadmium and salinity different growth parameters and photosynthesis were better than other treatments (salinity and cadmium alone). The entrance of cadmium to plants decreased in presence of salinity and the poisonous effects of salinity and cadmium reduced in interaction of these two factors.
REFERENCES
- Arnon, D.I., 1949. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol., 24: 1-15.
CrossRefPubMedDirect Link - Cieslinski, G., G.H. Neilsen and E.J. Hogue, 1996. Effect of soil cadmium application and pH on growth and cadmium accumulation in roots, leaves and fruit of strawberry plants (Fragaria xananassa Duch). Plant Soil, 180: 267-276.
Direct Link - Chapin III, F.S., A.J. Bloom, C.D. Field and R.H. Waring, 1987. Plant responses to multiple environmental factors. BioScience, 37: 49-57.
CrossRefDirect Link - Ehret, D.L., R.E. Redmann, B.L. Harvey and A. Cipywnyk, 1990. Salinity-induced calcium deficiencies in wheat and barley. Plant Soil, 128: 143-151.
CrossRefDirect Link - Greger, M., L. Kautsky and T. Sandberg, 1995. A tentative model of Cd uptake in Potomogeton pectinatus in relation to salinity. Environ. Exp. Bot., 35: 215-225.
Direct Link - Hasegawa, P.M., R.A. Bressan, J.K. Zhu and H.J. Bohnert, 2000. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Physiol. Plant Mol. Biol., 51: 463-499.
CrossRefDirect Link - Hatch, D.J., L.H.P. Jones and R.G. Burau, 1988. The effect of pH on the uptake of cadmium by four plant species grown in flowing solution culture. Plant Soil, 105: 121-126.
CrossRef - Hurkman, W.J., 1992. Effect of salt stress on plant gene expression: A review. Plant Soil, 146: 145-151.
CrossRef - Inouhe, M., S. Ninomiya, H. Tohoyama, M. Joho and T. Murayama, 1994. Different characteristics of roots in the cadmium-tolerance and Cd-binding complex formation between mono and dicotyledonous plants. J. Plant Res., 107: 201-207.
CrossRef - Karez, C.S., D. Allemand, G.D.E. Renzis, M. Gnassia-Barelli, M. Romeo and S. Puiseux-Dao, 1990. Ca-Cd interaction in prymnesiophyte Cricosphaera elongata. Plant Cell Environ., 13: 483-487.
CrossRef - King, D.A. and W.L. Nelson, 1987. Assessing the impacts of soil moisture stress on Regional soybean yield and its sensitivity to ozone. Agric. Ecosyst. Environ., 20: 23-35.
CrossRef - Lynch, J. and A. Lauchli, 1985. Salt stress disturbs the calcium nutrition of barley (Hordeum vulgare L.). New Phytol., 99: 345-354.
CrossRefDirect Link - Mozafar, A. and J.J. Oertli, 1990. Multiple stress and growth of barley: Effect of salinity and temperature shock. Plant Soil, 128: 153-160.
CrossRef - Ouzounidou, G., M. Moustakas and E.P. Eleftheriou, 1997. Physiological and ultrastructural effects of cadmium on wheat (Triticum aestivum L.) leaves. Arch. Environ. Contam. Toxicol., 32: 154-160.
CrossRef - Prado, F.E., C. Boero, M. Gallardo and J.A. Gonzalez, 2000. Effect of NaCl on germination, growth and soluble sugar content in Chenopodium quinoa willd. seeds. Bot. Bull. Acad. Sin., 41: 27-34.
Direct Link - Rascio, N., F.D. Vecchia, M. Ferretti, L. Merlo and R. Ghisi, 1993. Some effects of cadmium on maize plants. Arch. Environ. Contam. Toxicol., 25: 244-249.
CrossRef - Reynolds, C.E., G. Houleand and C. Marquis, 2001. Light and salinity affect growth of the salt marsh plant Aster Laurentianus. New Phytol., 149: 441-448.
Direct Link - Sheoran, I.S., H.R. Singal and R. Singh, 1990. Effect of cadmium and nickel on photosynthesis and enzymes of the photosynthetic carbon reduction cycle in pigeonpea Cajanus cajan L.). Photosynthesis Res., 23: 345-351.
Direct Link - Stobart, A.K., W.T. Griffiths, I. Ameen-Bukhari and R.P. Sherwood, 1985. The effect of Cd2+ on the biosynthesis of chlorophyll in leaves of barley. Physiol. Plant, 63: 293-298.
CrossRefDirect Link - Temmerman, L.O., M. Hoenig and P.O. Scokart, 1984. Determination of normal levels and upper limit values of trace elements in soils. Z. Pflanzenernahr Bodenkd, 147: 687-694.
CrossRefDirect Link - Umezawa, T., K. Shimizu, M. Kato and T. Ueda, 2000. Enhancement of salt tolerance in soybean with NaCl pretreatment. Physiol. Plant., 110: 59-63.
CrossRef








