ABSTRACT
The present study was carried out to study the effect of chilling stress on some metabolic products and antioxidative system in mung bean (Vigna radiata L.) plant and try to alleviate chilling injuries by using paclobutrazol, abscisic acid and H2O2. Twenty-day-old seedlings were randomly separated into equal three groups, the first group was left in green house at 35°C, the second one was chilled at 5°C for 5 or 10 h and the last one was treated with 0.0, 25 and 50 mg L-1 paclobutrazol, 0.5 and 1 mM abscisic acid or 0.0, 0.1 and 0.5 mM H2O2 before transforming to 5°C. The results showed that, chilling stress induced significant increased lipid peroxidation, membrane leakage and hydrogen peroxide level, while activities of catalase, peroxidase and ascorbate peroxidase were decreased. In addition, total chlorophyll, total carbohydrates, protein content and proline level decreased following exposure to 5°C. Paclobutrazol, abscisic acid and H2O2 treatments ameliorated the chilling injuries by lowering lipid peroxidation, membrane leakage and H2O2 level and increasing in total chlorophyll, carbohydrates, protein content, proline level and antioxidant enzymes activities. Based on these results, it was presumed that the stress protection caused by paclobutrazol, abscisic acid and H2O2 probably contributes to some extent to the enhanced activity of the free-radical scavenging systems.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajpp.2007.318.332
URL: https://scialert.net/abstract/?doi=ajpp.2007.318.332
INTRODUCTION
Cultivated plants are often subjected to different types of environmental stress during their growth in the field, which could result in reduction of their yield. Low temperature stress (chilling) induces considerable changes in biochemistry and physiology of plants (Katterman, 1990). Exposure of watermelon seedlings to low temperatures retards growth, delays flowering, reduces total yields and quality and even kills the plants (Korkmaz and Dufault, 2001).
Electrolyte leakage indicator as chilling injuries increased in Stylosanthes guianesis under chilling conditions due to damage of the membrane system (Zhou et al., 2005). In addition, prolonged exposure to low temperatures increased the leakage of solutes in mung bean seedlings, such as soluble sugars and free amino acids (Chang et al., 2001). Chilling-enhanced lipid peroxidation, as indexed by malondialdehyde (MDA) content, in maize cells (Chen and Li, 2002) and wheat seedlings (Berova et al., 2002). These deteriorative symptoms in wheat seedlings were ameliorated by the paclobutrazol (PBZ) treatment (Berova et al., 2002) due to enhancing the activity of the free-radical scavenging systems.
Chilling induces oxidative stress (Prasad et al., 1994) during which Reactive Oxygen Species (ROS), including hydrogen peroxide (H2O2), are accumulated in concentrations higher than necessary for normal metabolism. High levels of H2O2 could promote lipid peroxidation in the membranes (Senaratna et al., 1987) in the absence of increased antioxidant mechanism. The H2O2 excess can be removed by catalase and the ascorbate-glutathione pathway (Apel and Hirt, 2004). The higher activities of defense enzymes and higher content of antioxidant under stress were associated with tolerance to chilling (Huang and Guo, 2005). Activities of catalase, ascorbate peroxidase, glutathione reductase and superoxide dismutase were decreased in chilling sensitive cultivar of rice (Huang and Guo, 2005), cucumber (Heelee and Bumlee, 2000; Saropulos and Drennan, 2002) and Stylosanthes guianesis (Zhou et al., 2005) under chilling stress.
Many plant species accumulate free proline in response to salinity, drought and cold (Delauney and Verma, 1993; Hare and Cress, 1997; Yoshiba et al., 1997). Positive correlations between the accumulation of endogenous proline and improved cold tolerance have been found in maize (Zhou et al., 2002), rye (Koster and Lynch, 1992), wheat (Dörffling et al., 1997), grapevine (Ait-Barka and Audran, 1997), potato (Swaaij et al., 1986) and Arabidopsis thaliana (Nanjo et al., 1999). Proline has been suggested to play multiple roles in plant stress tolerance. It acts as a mediator of osmotic adjustment (Yoshiba et al., 1997), a stabilizer of proteins and membranes (Rudolph et al., 1986), an inducer of osmotic stress-related genes (Iyer and Caplan, 1998) and a scavenger of Reactive Oxygen Species (ROS) (Saradhi et al., 1995).
In general, chilling injury is characterized by reduction of chlorophyll levels in wheat plant (Berova et al., 2002) and inhibition of the photosynthetic process (Hodgson and Raison, 1989). Maize leaves grown at low temperature are characterized by a very low photosynthetic performance (Haldimann et al., 1996), changes in the content and the composition of the pigments (Haldimann et al., 1996; Haldimann, 1998), changes in the activities of several enzymes of photosynthetic carbon assimilation (Stamp, 1987) and reduced activities of photosystem I and photosystem II (Robertson et al., 1993).
Paclobutrazol, which is a member of the triazole family of plant growth regulators, has been found to protect severa1 crops from various environmental stresses, including drought, chilling, heat and UV-B radiation (280-320 nm) (Davis and Curry, 1991; Lurie et al., 1994; Pinhero and Fletcher, 1994; Kraus et al., 1995; Paliyath and Fletcher, 1995). Even though paclobutrazol-induced stress tolerance is reported to be due to increased antioxidant enzymes (Paliyath and Fletcher, 1995; Kraus et al., 1995), increased levels of proline (Mackay et al., 1990) and chlorophyll content (Berova et al., 2002). Foliar applications of uniconazole reduced electrolyte leakage and malondialdehyde accumulation caused by freezing stress (Berova et al., 2002). Enhanced chilling tolerance in triazole-treated cucumber (Upadhyaya et al., 1989) and tomato (Senaratna et al., 1988) was associated with increased antioxidant enzyme concentrations. Triazole-induced tolerance to low temperature stress has been associated with increased levels of endogenous abscisic acid (Fletcher et al., 2000), which has increased free proline (Chen and Li, 2002) and scavenging systems (Fath et al., 2001; Zhou et al., 2002).
High levels of endogenous abscisic acid (ABA) are related to increase chilling tolerance and an increase in ABA content before low temperature exposure might be an essential step in activating a protection mechanism against chilling (Zhang et al., 1986). ABA-decreased chilling injuries in tomatoes (King et al., 1982), cucumbers (Yamazaki et al., 1995) and Stylosanthes guianesis (Zhou et al., 2002) is partially associated with enhanced scavenging systems and increases in endogenous free proline content in rice (Chou et al., 1991), maize (Chen and Li, 2002) and cucumber plants (Flores et al., 1988). However, ABA-treated maize cells displayed less lipid peroxidation and accumulated proline in response to chilling and enough to improve chilling tolerance (Chen and Li, 2002).
H2O2 can play dual roles in plants exposed to chilling stress, inducing not only oxidative damage, but also acting as a signal for induction of defenses, resulting in chilling tolerance (Prasad et al., 1994). Exogenously supplied of H2O2 induced chilling tolerance in maize plant exposed to 4°C (Prasad et al., 1994) and mung bean seedlings (Chih-Wen et al., 2003) by increasing antioxidant enzymes such as catalase and peroxidase and lowering electrolyte leakage.
Thus, the objective of this research was to carried out to examine the effect of low temperature on some metabolic activities and antioxidant enzymes of mung bean (Vigna radiata L.) plant and trying to alleviate the injuries caused by low temperature by using paclobutrazol (PBZ), Abscisic acid (ABA) and hydrogen peroxide (H2O2).
MATERIALS AND METHODS
Plant Material and Growth Conditions
Mung bean (Vigna radiata L.) seeds were obtained from the Agricultural Research Center (ARC), Giza, Egypt. Seeds were sterilized with 2.5% sodium hypochlorite for 15 min and washed with distilled water. Seeds were immersed in running water overnight and then grown in plastic pots (25 cm in height and 20 cm in diameter) which equally filled with beat-moss. All pots were kept in a green house under natural photoperiod and a temperatures regime of 35/22°C (day/night) and irrigated regularly every two days until chilling treatment. Thinning was carried out after 10 days from germination so that six seedlings of symmetrical growth rates were left per pot. This study was carried out at Botany Department, Faculty of Science, Suez Canal University, Ismailia, Egypt.
Paclobutrazol, Abscisic Acid or Hydrogen Peroxide Treatments
Twenty-day-old, seedlings were used for chilled treatments are separated into three groups. The first group was left in green house at 35°C (negative control). Seedlings of the second group were transferred to 5°C and 45 μmol m-2 sec-1 in an light incubator for various lengths of time (5 and 10 h) (positive control). The root portions of the whole seedlings of the third group were immersed in 0.0, 25 and 50 mg L-1 paclobutrazol or 0.5 and 1 mM ABA for 24 h or in 0.0, 0.1 and 0.5 mM H2O2 for 4 h at 27°C in darkness. The treated seedlings were washed with distilled water, transplanted in peat moss and further incubated at 27°C for an additional 12 h before transforming to 5°C (5 and 10 h). At the end of the chilling period, the treated plants were returned to the pre-experimental conditions in the greenhouse, where they have recovered for 5 days. Treatments were replicated using 4 pots. 25-day-old plants were harvested, weighed, frozen and stored in deep-freezer for further analysis.
Measurement of Chilling Injury
The conductivity of electrolyte leakage in the cell sap from leaf discs was used as a measure parameter of chilling injury (Sukumaran and Weiser, 1972).
Determination of Malondialdehyde Content
Malondialdehyde (MDA) content, an indicator of lipid peroxidation, was assayed spectrophotometrically using TBA-MDA assay (Minotti and Aust, 1987).
Assay of H2O2
H2O2 concentration in the incubation medium of treated leaf explants was measured by the FOX I method (Jiang et al., 1990; Wolf, 1994) based on the peroxide-mediated oxidation of Fe2+ with xylenol orange.
Antioxidant Enzymes
Enzymes extracts were prepared in 20 mL chilled extraction buffer (pH 7.5). Extracts were then centrifuged at 6000 rpm for 20 min at 5°C. Enzyme assays were conducted immediately following extraction.
Determination of Catalase Activity
Catalase activity was assayed in a method following Aebi (1983). Activity was determined by following the decomposition of H2O2 at 240 nm.
Determination of Peroxidase Activity
Peroxidase activity was determined by following the dehydrogenation of guaicol at 436 nm (Malik and Singh, 1980).
Determination of Ascorbate Peroxidase (ASPX) Activity
ASPX activity was determined using the method of Nakano and Asada (1987). Activity was determined by following the H2O2 dependent decomposition of ascorbate at 290 nm.
Estimation of Proline
Proline was assayed colorimetrically using the method of Bates et al. (1973) at wave length 520 nm.
Determination of Total Protein
The colorimetric method for the determination of protein according to Lawry et al. (1951) at wave length of 500 nm was used.
Estimation of Chlorophyll Content
Total chlorophylls were estimated in the fresh plant leaves according to the procedure of Lichtenthaler (1987).
Determination of Total Carbohydrates
The total available carbohydrate content were extracted according to Smith et al. (1964) and estimated colorimetrically by the Phenol-Sulphuric Acid Method as described by Dubois et al. (1951).
Statistical Analyses
Analysis of variance (ANOVA) and student t-test was performed on all data using SPSS program (Version 11.0).
RESULTS
Electrolyte Leakage
Chilling stress induced a significant increase in electrolyte leakage percentage of mung bean seedlings when compared to the negative control (Fig. 1). It was also found that increasing time of chilling stress increased electrolyte leakage. In addition, electrolyte leakage of paclobutrazol, abscisic acid or hydrogen peroxide-treated plants, which chilled for 5 h, decreased to the negative control level after the plants were recovered for 5 days.
Lipid Peroxidation (LP) Level
Lipid peroxidation, expressed as MDA levels, in mung bean seedlings was significantly increased with increasing chilling time. The percentage of increasing was 88.83 and 131.7% in the seedlings exposed to 5°C for 5 or 10 h, respectively, relative to the negative control (Fig. 2). Lipid peroxidation level was significantly decreased after treating the mung bean seedlings with paclobutrazol, abscisic acid or hydrogen peroxide. The decrease was more pronounced at 1 mM ABA and 0.5 mM H2O2 in the seedlings exposed to 5°C for 5 h.
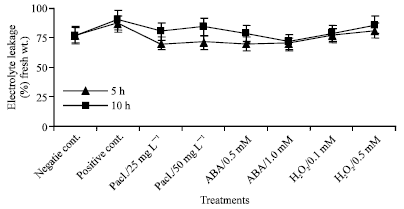 | |
| Fig. 1: | Electrolyte leakage expressed as (%) fresh wt. in the leaf extract of mung bean seedlings pretreated with 0.0, 25 and 50 mg L-1 Pacloputrazol, 0.0, 0.5 and 1 mM ABA or 0.0, 0.1 and 0.5 mM H2O2. The seedlings then subjected to chilling stress at 5°C for 5 or 10 h. Then, the treated plants were transferred to the pre-experimental conditions in the green house, where they have recovered for 5 days. Data represented are mean of 3 replica±SE |
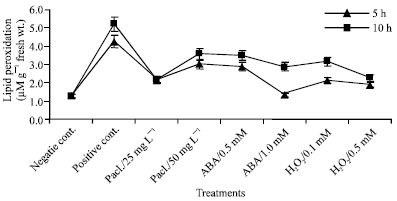 | |
| Fig. 2: | Lipid peroxidation as malondialdehyde content (MDA) expressed as μmole g-1 fresh wt. in the leaf extract of mung bean seedlings pretreated with 0.0, 25 and 50 mg L-1 pacloputrazol, 0.0, 0.5 and 1 mM ABA or 0.0, 0.1 and 0.5 mM H2O2. The seedlings then subjected to chilling stress at 5°C for 5 or 10 h. Then, the treated plants were transferred to the pre-experimental conditions in the green house, where they have recovered for 5 days. Data represented are mean of 3 replica±SE |
H2O2 Level
It was noticed that chilling stress at 5°C induced a highly significant increase in H2O2 content comparing with unchilled plants as shown in Fig. 3. In addition, increasing time of chilling stress increased H2O2 content when compared to the negative control. The percentage of increasing was 86.76 and 237.86% in the seedlings exposed to 5°C for 5 or 10 h respectively, compared to the negative control. Pretreatment with paclobutrazol, abscisic acid and H2O2 lowered H2O2 level of mung bean seedlings. The highest reduction in H2O2 level was observed at 25 mg L-1 paclobutrazol (70.1%) and 1.0 mM ABA (66.85%) in seedlings chilled for 5 h, in comparison with the positive control.
Catalase Activity
The results in Fig. 4 revealed that low temperature stress induced a significant decrease in catalase activity when compared to the negative control. This reduction increased with increasing time of stress.
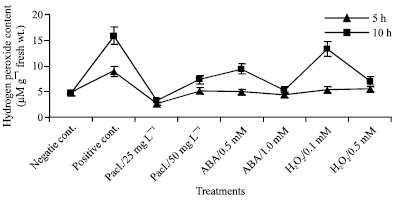 | |
| Fig. 3: | H2O2 content expressed as μM g-1 fresh wt. in the leaf extract of mung bean seedlings pretreated with 0.0, 25 and 50 mg L-1 pacloputrazol, 0.0, 0.5, 1 mM ABA or 0.0, 0.1 and 0.5 mM H2O2. The seedlings then subjected to chilling stress at 5°C for 5 or 10 h. Then, the treated plants were transferred to the pre-experimental conditions in the green house, where they have recovered for 5 days. Data represented are mean of 3 replica±SE |
 | |
| Fig. 4: | Catalase activity expressed as unit g-1 fresh wt. in the leaf extract of mung bean seedlings pretreated with 0.0, 25 and 50 mg L-1 pacloputrazol, 0.0, 0.5 and 1 mM ABA or 0.0, 0.1 and 0.5 mM H2O2. The seedlings then subjected to chilling stress at 5°C for 5 or 10 h. Then, the treated plants were transferred to the pre-experimental conditions in the green house, where they have recovered for 5 days. Data represented are mean of 3 replica±SE |
Paclobutrazol, abscisic acid or hydrogen peroxide treated seedlings showed an enhancement catalase activity at all concentrations used in both seedlings stressed for 5 or 10 h over that of the positive control. The highest increasing in catalase activity was observed at 25 mg L-1 paclobutrazol (70.90 and 178.88%), 1.0 mM ABA (43.99 and 134.95%) and 0.5 mM H2O2 (15.58 and 88.60%) in seedlings chilled for 5 h, in comparison with the negative and the positive control, respectively.
Peroxidase Activity
After exposure of mung bean seedlings to chilling stress, the peroxidase activity decreased as shown in Fig. 5. In addition, prolonged exposure to chilling stress decreased peroxidase activity. Treatment with different concentrations of paclobutrazol, abscisic acid or hydrogen peroxide significantly increased peroxidase activity over that of the negative and positive control. Paclobutrazol and abscisic acid, however, introduced the highest peroxidase activity.
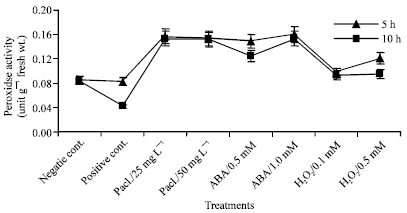 | |
| Fig. 5: | Peroxidase activity expressed as unit g-1 fresh wt. in the leaf extract of mung bean seedlings pretreated with 0.0, 25 and 50 mg L-1 pacloputrazol, 0.0, 0.5 and 1 mM ABA or 0.0, 0.1 and 0.5 mM H2O2. The seedlings then subjected to chilling stress at 5°C for 5 or 10 h. Then, the treated plants were transferred to the pre-experimental conditions in the green house, where they have recovered for 5 days. Data represented are mean of 3 replica±SE |
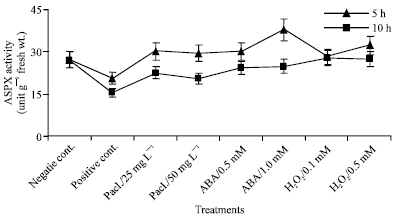 | |
| Fig. 6: | ASPX activity expressed as unit g-1 fresh wt. in the leaf extract of mung bean seedlings pretreated with 0.0, 25 and 50 mg L-1 pacloputrazol, 0.0, 0.5 and 1 mM ABA or 0.0, 0.1 and 0.5 mM H2O2. The seedlings then subjected to chilling stress at 5°C for 5 or 10 h. Then, the treated plants were transferred to the pre-experimental conditions in the green house, where they have recovered for 5 days. Data represented are mean of 3 replica±SE |
Ascorbate Peroxidase (ASPX) Activity
The ASPX activity was determined in leaves of mung bean seedlings chilled at 5°C for 5 or 10 h as shown in Fig. 6. The data showed that chilling stress induced a significant decline in ASPX activity when compared to the negative control. Treating of mung bean seedlings with paclobutrazol, abscisic acid or hydrogen peroxide significantly increased ASPX activity. Paclobutrazol and abscisic acid, however, introduced the highest ASPX activity in seedlings chilled for 5 h, in comparison with the positive control.
Proline Content
Control mung bean seedlings had low proline content after chilling for 5 or 10 h at 5°C in comparison with unchilled control (Fig. 7). Under chilling conditions, paclobutrazol, abscisic acid and H2O2-treated plants had higher proline content than positive and negative control but abscisic acid introduced the highest proline content.
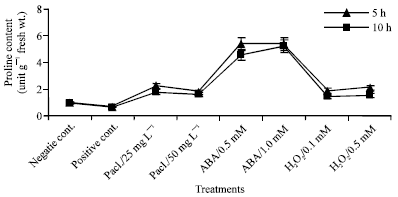 | |
| Fig. 7: | Proline content expressed as μM g-1 fresh wt. in the leaf extract of mung bean seedlings pretreated with 0.0, 25 and 50 mg L-1 pacloputrazol, 0.0, 0.5 and 1 mM ABA or 0.0, 0.1 and 0.5 mM H2O2. The seedlings then subjected to chilling stress at 5°C for 5 or 10 h. Then, the treated plants were transferred to the pre-experimental conditions in the green house, where they have recovered for 5 days. Data represented are mean of 3 replica±SE |
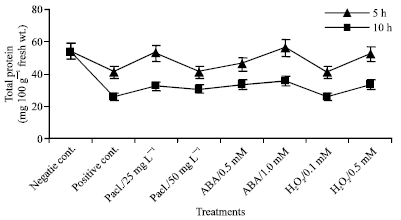 | |
| Fig. 8: | Total protein expressed as mg/100 g fresh wt. in the leaf extract of mung bean seedlings pretreated with 0.0, 25 and 50 mg L-1 pacloputrazol, 0.0, 0.5 and 1 mM ABA or 0.0, 0.1 and 0.5 mM H2O2. The seedlings then subjected to chilling stress at 5°C for 5 or 10 h. Then, the treated plants were transferred to the pre-experimental conditions in the green house, where they have recovered for 5 days. Data represented are mean of 3 replica±SE |
Total Protein
The results of Fig. 8 show that chilling stress induced a significant decrease in total protein content when compared to the negative control. This reduction increased with increasing time of stress. The percentage reduction being 24.38 and 52.59% in the plants chilled at 5 and 10 h, respectively, compared to the negative control. Paclobutrazol, abscisic acid and H2O2 treatments enhanced protein contents over that of positive control, but still lower than the negative control.
Total Chlorophyll
The data revealed that low temperature stress for 5 and 10 h induced a significant decrease in total chlorophyll content in mung bean seedlings when compared to negative control (Table 1). Treating mung bean seedlings with paclobutrazol, abscisic acid and H2O2 induced a significant increase in total chlorophyll content when compared to the positive control level but still decreased than the negative control.
| Table 1: | Effect of chilling time on total chlorophyll and carbohydrate contents of mung bean |
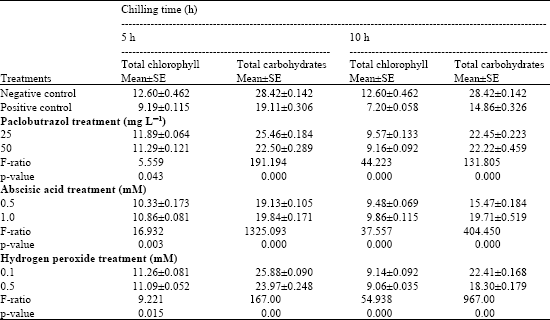 | |
| Total chlorophyll content expressed as μg g-1 dry weight and total carbohydrates expressed as mg glucose/100 g dry wt. in mung bean seedlings pretreated with 0.0, 25 and 50 mg L-1 pacloputrazol, 0.0, 0.5 and 1 mM ABA or 0.0, 0.1 and 0.5 mM H2O2. The seedlings then subjected to chilling stress at 5°C for 5 or 10 h. Then, the treated plants were transferred to the pre-experimental conditions in the green house, where they have recovered for 5 days. Data represented are mean of 3 replica±SE | |
Total Carbohydrates
The data in Table 1 showed that increasing time of chilling led to decreasing in total carbohydrates when compared to the negative control. Increasing in total carbohydrates content after the chilling stress was higher in paclobutrazol, abscisic acid and H2O2–treated seedlings in comparison with the positive control, but still lower than the negative control. The increase was much more pronounced at paclobutrazol and H2O2 treatments.
DISCUSSION
When the plants are exposed to unfavorable conditions, Activated Oxygen Species (AOS) can react with pigments, membranes, enzymes and nucleic acids, thereby modifying their functions (Sun and Leopold, 1995). Autocatalytic peroxidation of membrane lipids can be triggered by AOS, resulting in loss of membrane semipermeability (Basaga, 1989), one of the primary mechanisms of stress injury. Electrolyte leakage and lipid peroxidation level in mung bean plant increased under chilling conditions (Fig. 1, 2). This result is in agreement with Zhou et al. (2002), who reported the membrane system of Stylosanthes guianensis was damaged under chilling stress due to the induction of oxidative damage is related to the imbalance of ROS production (Bowler et al., 1992). ROS are highly reactive and have the potential to damage membrane lipids, proteins, chlorophyll and nucleic acids, thus disrupting the homeostasis of the organism (Scandalios, 1993). Lyons (1973) proposed that membranes of the plant under low temperature become less fluid, their protein components can no longer function normally, causing water and soluble materials to leak out into the intercellular spaces. Paclobutrazol, abscisic acid or H2O2 treatments decreased membrane leakage and lipid peroxidation in mung bean plant (Fig. 1, 2). This result is in agreement with Berova et al. (2002) and Chen and Li (2002), they found that the lipid peroxidation content in paclobutrazol-treated wheat and maize seedlings was reduced. The ability of ABA-treated plants to protect cellular membranes during chilling may be attributed, in part, to higher activities of reactive oxygen scavenging enzymes such as catalase, ascorbate peroxidase and superoxide dismutase, which are known to be up-regulated by ABA (Fath et al., 2001). The data of antioxidant enzymes support this suggestion as showed in Fig. 4-6.
One of the reactive oxygen species that accumulates in plant tissues during cold stress is hydrogen peroxide (H2O2) (Kang et al., 2003). The results of the present study revealed that chilling stress induced a significant increase in hydrogen peroxide (H2O2) level of mung bean seedlings (Fig. 3). These data are in according of these obtained by Aroca et al. (2005) who stated that Chilling can cause H2O2 accumulation in Zea mays plant could be attribute to decrease activity of key antioxidant enzymes (catalase and superoxide dismutase) activity (Scandalios et al., 1997; Polle, 1997). The results of the present study showed that, H2O2 decreased after treatment of mung bean seedlings with paclobutrazol, abscisic acid or H2O2 (Fig. 3). This result is in agreement with Parasad et al. (1994), who reported that maize seedlings pretreated with H2O2 or ABA and exposed to 4°C induced chilling tolerance could attribute to increase the antioxidant enzymes. Paclobutrazol, H2O2 and ABA treatments protect wheat from oxidative stress by enhancing antioxidant enzyme activity (Kraus et al., 1995) or other factors involved in membrane stability (Prasad et al., 1994).
Higher plants possess several enzymatic and non-enzymatic scavenging systems to minimize deleterious effects of Reactive Oxygen Species (ROS) including H2O2. Hydrogen peroxide is scavenged by catalases, peroxidase and ascorbate peroxidase (Kim et al., 2005). The results of the present work showed that chilling stress induced a significant decrease in catalase, peroxidase and ascorbate peroxidase activities in mung bean seedlings (Fig. 4-6). A large body of evidence has shown that the antioxidant enzyme systems are altered under abiotic stresses, including chilling (Kim et al., 2005). The chilling treatment decreased significantly both the catalase and the ascorbate peroxidase activities in rice (Huang and Guo, 2005) and Stylosanthes guianesis (Zhou et al., 2002). Mung bean seedlings treated with paclobutrazol, abscisic acid or H2O2 had higher activity of catalase, peroxidase and ascorbate peroxidase (Fig. 4-6). Yong et al. (2003) suggests a higher CAT activity in rice plant more efficient scavenging of H2O2, which would result in better protection against this toxic molecule during chilling and thus protect rice leaves from chilling injury. ABA pretreated increased activities of antioxidant enzymes in maize seedlings and reduces the degrees of oxidative damage under stressful conditions (Zhou et al., 2002). In addition, paclobutrazol and ABA-induced chilling tolerance due to increased antioxidant enzymes and protected the membranes from oxidative stress (Prasad et al., 1994; Paliyath and Fletcher, 1995).
In the present study, the results in Fig. 7 revealed that low temperature stress induced significant decreased in proline content in mung bean seedlings, while pretreatment with paclobutrazol, ABA or H2O2 induced a significant increase in its content. However, proline content in the present study decreased with increasing time of chilling stress. The present results are supported by that of Chen and Li (2002), who reported most of the accumulated proline in maize cells, was lost to the medium during a prolonged chilling treatment. The leakage might be attributed to membrane damage (Chen and Li, 2002), as evidenced by the chilling-enhanced lipid peroxidation (Fig. 2). It has been shown that plant membrane damage during chilling is related to the peroxidation of membrane lipid due to the stress-induced accumulation of free radicals (Wise and Naylor, 1987). Whether proline prevents membrane lipid peroxidation in plant tissue by acting as an antioxidant to counteract the chilling-induced free radicals (Xin and Li, 1993). ABA induces chilling tolerance in maize plant by increasing free proline content (Chen and Li, 2002). Heber et al. (1973) showed that proline is capable of preventing freezing-induced inactivation of membrane activities. Whether proline prevents membrane lipid peroxidation in plant tissue by acting as an antioxidant to counteract the chilling-induced free radicals is under investigation. These results suggest that increased chilling tolerance by overexpression of reactive oxygen scavenging enzymes in plants (Mckerise et al., 2000) might be due to the prevention of proline leakage. From these results, it appears that paclobutrazol, ABA and H2O2 may influence chilling tolerance in mung bean by increasing free proline content (Xin and Li, 1993).
As showed in the present study (Fig. 8), chilling stress induced a significant decline in total protein content in mung bean seedlings. Paclobutrazol, abscisic acid and hydrogen peroxide significantly increased protein content in the plant under investigation. The decline in protein content may be due to extensive damage of protein synthesizing system in various crop (Hallman et al., 1973) and tree (Espindola et al., 1994) or synthesis or activation of large quantities of proteolytic enzymes (Bewley and Black, 1982) as protease (Krishna et al., 2000). Adva and Waisel (1975) reported that at temperatures of 0 to 10°C, hydrophobic bonding in proteins decreases, causing conformation changes in protein structure and irreversible injury to plants. Plants synthesize a spectrum of new proteins on exposure to different environmental stresses including cold stress (Sutton et al., 1992). Proteins induced by stress fall into three categories: (i) those inducible by stress and ABA; (ii) specifically induced by stress but not by ABA and (iii) inducible by ABA. Therefore, it is quite evident that many of these stress-responsive proteins.
AOS generated under environmental stresses are highly reactive and have the potential to damage membrane lipids, proteins, chlorophyll and nucleic acids, thus disrupting the homeostasis of the organism (Rao et al., 1996). Low temperatures generally cause a decrease in the entire metabolism and in the photochemical steps of photosynthesis (Oliveira et al., 2002). In mung bean plant, chilling stress induced a significant decline in total chlorophyll content, while paclobutrazol, abscisic acid and hydrogen peroxide treatments induced significantly increased total chlorophyll content when compared to the positive control (Table 1). Leaf chlorophyll content was reduced significantly after plants were subjected to freezing stress and foliar sprays of paclobutrazol retarded the degradation of chlorophyll (Zhou and Leul, 1998). It was established that paclobutrazol prevent the decline in total chlorophyll content in corn plants after exposure to chilling temperature (Pinhero and Fletcher, 1994). Paclobutrazol-induced freezing tolerance was accompanied by increased activities of various antioxidant enzymes, including superoxide dismutase, catalase and peroxidase (Zhou and Leul, 1998). In addition, the protection of photosynthesis can be realized by reducing either superoxide or H2O2 levels, thereby reducing the possibility of hydroxyl radical formation (Payton et al., 2001).
As seen in results (Table 1), mung bean showed a significant decline in carbohydrates content when faced a chilling stress, but treating mung bean seedlings with paclobutrazol, abscisic acid and hydrogen peroxide significantly increased carbohydrates content when compared to the positive control. The stress effect on photosynthetic activity is manifested in many different ways, leading to efficiency loss in the photosynthetic apparatus, such as decreased use of photons for NADPH, ATP and carbohydrate production (Oliveira et al., 2002). Haldimann (1999) observed in maize leaves grown at low temperature is likely to be related to the fact that such leaves display reduced proportions of reaction centre core complexes to light harvesting antenna complexes in photosystem I and photosystem II (Nie and Baker, 1991). This feature is associated with reduced electron transport activities in both photosystems (Nie et al., 1995). Low temperatures generally cause a decrease in the entire metabolism and in the photochemical steps of photosynthesis, which are interdependent on the biochemical phase and, as expected, more directly influenced by low temperatures (Öquist, 1983). In addition, cold stress can affect photosynthesis rates by inhibiting the light and dark reactions of photosynthesis (Katterman, 1990) and change in the activities of several enzymes of photosynthetic carbon assimilation (Stamp, 1987).
In conclusion, mung bean plant is sensitive to low temperature. The result of the present investigation suggested that paclobutrazol, abscisic acid and H2O2 improved chilling injuries by decreasing lipid peroxidation, electrolyte leakage and H2O2 level and increasing catalase, peroxidase, ascorbate peroxidase activities and free proline content.
REFERENCES
- Ait-Barka, E. and J.C. Audran, 1997. Response of champenoise grapevine to low temperatures: Changes of shoot and bud proline concentrations in response to low temperatures and correlations with freezing tolerance. J. Hortic. Sci., 72: 577-582.
Direct Link - Apel, K. and H. Hirt, 2004. Reactive oxygen species: Metabolism, oxidative stress and signal transduction. Annu. Rev. Plant Biol., 55: 373-399.
CrossRefPubMedDirect Link - Aroca, R., G. Amodeo, S. Fernandez-Illescas, E. Herman, F. Chaumont and M. Chrispeels, 2005. The role of aquaporins and membrane damage in chilling and hydrogen peroxide induced changes in the hydraulic conductance of maize roots. Plant Physiol., 137: 341-353.
CrossRefDirect Link - Basaga, H.S., 1990. Biochemical aspects of free radicals. Biochem. Cell Biol., 68: 989-998.
CrossRefDirect Link - Bates, L.S., R.P. Waldren and I.D. Teare, 1973. Rapid determination of free proline for water-stress studies. Plant Soil, 39: 205-207.
CrossRefDirect Link - Berova, M., Z. Zlatev and N. Stoeva, 2002. Effect of paclobutrazol on wheat seedlings under low temperature stress. Bulg. J. Plant Physiol., 28: 75-84.
Direct Link - Bowler, C., M.V. Montagu and D. Inze, 1992. Superoxide dismutase and stress tolerance. Annu. Rev. Plant Physiol. Mol. Biol., 43: 83-116.
CrossRefDirect Link - Chang, M., S. Chen, S. Lee and Y. Chen, 2001. Cold-acclimation and root temperature protection from chilling injury in chilling-sensitive mung bean (Vigna radiata L.) seedlings. Bot. Bull. Acad. Sin., 42: 53-60.
Direct Link - Chen, W.P. and P.H. Li, 2002. Membrane stabilization by abscisic acid under cold aids proline in alleviating chilling injury in maize (Zea mays L.) cultured cells. Plant Cell Environ., 25: 955-962.
Direct Link - Yu, C.W., T.M. Murphy and C.H. Lin, 2003. Hydrogen peroxide-induced chilling tolerance in mung beans mediated through ABA-independent glutathione accumulation. Plant Biol., 30: 955-963.
CrossRefDirect Link - Chou, I.T., C.T. Chen and C.H. Kao, 1991. Characteristics of the induction of the accumulation of proline by abscisic acid and isobutyric acid in detached rice leaves. Plant Cell Physiol., 32: 269-272.
Direct Link - Davis, T.D., E.A. Curry and G.L. Steffens, 1991. Chemical regulation of vegetative growth. Crit. Rev. Plant Sci., 10: 204-216.
CrossRefDirect Link - Delauney, A.J. and D.P.S. Verma, 1993. Proline biosynthesis and osmoregulation in plants. Plant J., 4: 215-223.
CrossRef - Dorffling, K., H. Dorffling, G. Lesselich, E. Luck, C. Zimmermann, G. Melz and H.U. Jurgens, 1997. Heritable improvement of frost tolerance in winter wheat by in vitro-selection of hydroxyproline-resistant proline overproducing mutants. Euphytica, 93: 1-10.
Direct Link - Dubois, M., K. Gilles, J.K. Hamilton, P.A. Rebers and F. Smith, 1951. A colorimetric method for the determination of sugars. Nature, 168: 167-167.
CrossRefPubMedDirect Link - Fath, A., P.C. Bethke and R.L. Jones, 2001. Enzymes that scavenge reactive oxygen species are down-regulated prior to gibberellic acid-induced programmed cell death in barley aleurone. Plant Physiol., 126: 156-166.
Direct Link - Fletcher, R.A., A. Gilley, N. Sankhla and T.D. Davis, 1999. Triazoles as Plant Growth Regulators and Stress Protectants. In: Horticultural Reviews, Janick, J. (Ed.)., John Wiley & Sons, Inc., Hoboken, New Jersey, ISBN: 9780470650776, pp: 55-138.
CrossRefDirect Link - Flores, A., A. Grau, F. Laurich and K. Dorffling, 1988. Effect of new terpenoid analogues of abscisic acid on chilling and freezing resistance. Plant Physiol., 132: 362-369.
CrossRefDirect Link - Haldimann, P., Y. Fracheboud and P. Stamp, 1996. Photosynthetic performance and resistance to photoinhibition of Zea mays L. leaves grown at sub-optimal temperature. Plant Cell Environ., 19: 85-92.
Direct Link - Haldimann, P., 1998. Low growth temperature-induced changes to pigment composition and photosynthesis in Zea mays genotypes differing in chilling sensitivity. Plant Cell Environ., 21: 200-208.
Direct Link - Haldimann, P., 1999. How do changes in temperature during growth affect leaf pigment composition and photosynthesis in Zea mays genotypes differing in sensitivity to low temperature? J. Exp. Bot., 50: 543-550.
Direct Link - Hallam, N.D., B.E. Roberts and D.J. Osborne, 1973. Embryogenesis and germination in rye (Secale cereale L.): III. Fine structure and biochemistry of the non-viable embryo. Planta, 110: 279-290.
Direct Link - Hare, P.D. and W.A. Cress, 1997. Metabolic implications of stress-induced proline accumulation in plants. Plant Growth Regul., 21: 79-102.
CrossRefDirect Link - Heber, U., L. Tyankova and K.A. Santarius, 1973. Effects of freezing on biological membranes in vivo and in vitro. Biochim. Biophys. Acta, 291: 23-37.
CrossRef - Lee, D.H. and C.B. Lee, 2000. Chilling stress-induced changes of antioxidant enzymes in the leaves of cucumber: In gel enzyme activity assays. Plant Sci., 159: 75-85.
CrossRefDirect Link - Hodgson, R.A.J. and J.K. Raison, 1989. Inhibition of photosynthesis by chilling in moderate light: A comparison of plants sensitive and insensitive to chilling. Planta, 178: 545-552.
CrossRefDirect Link - Huang, M. and Z. Guo, 2005. Responses of antioxidative system to chilling stress in two rice cultivars differing in sensitivity. Biologia Planta, 49: 81-84.
Direct Link - Iyer, S. and A. Caplan, 1998. Products of proline catabolism can induce osmotically regulated genes in rice. Plant Physiol., 116: 203-211.
Direct Link - Jiang, Z.Y., A.C.S. Woollard and S.P. Wolff, 1990. Hydrogen peroxide production during experimental protein glycation. FEBS Lett., 268: 69-71.
Direct Link - Kang, G., C. Wang, G. Sun and Z. Wang, 2003. Salicylic acid changes activities of H2O2-metabolizing enzymes and increase the chilling tolerance of banana seedlings. Environ. Exp. Bot., 50: 9-15.
Direct Link - Kim, S., J. Lim, M. Park, Y. Kim, T. Park, Y. Seo and S. Yun, 2005. Enhanced antioxidant enzymes are associated with reduced hydrogen peroxide in barley roots under saline stress. J. Biochem. Mol. Biol., 38: 218-224.
Direct Link - King, A.I., M.S. Reid and B.D. Patterson, 1982. Diurnal changes in the chilling sensitivity of seedlings. Plant Physiol., 70: 211-214.
CrossRefDirect Link - Korkmaz, A. and R.J. Dufault, 2001. Developmental consequences of cold temperature stresses at transplanting on seedling and field growth and yield: I. Watermelon. J. Am. Soc. Hortic. Sci., 126: 404-409.
Direct Link - Koster, K.L. and D.V. Lynch, 1992. Solute accumulation and compartmentation during the cold acclimation of Puma rye. Plant Physiol., 98: 108-113.
Direct Link - Kraus, T., B. McKersie and R. Fletcher, 1995. Paclobutrazol-induced tolerance of wheat leaves to paraquat may involve increased antioxidant enzyme activity. J. Plant Physiol., 145: 570-576.
Direct Link - Krishna, C., K.S. Keshavkant and S. Naithani, 2000. Changes in total protein and protease activity in dehydrating recalcitrant sal (Shorea robusta) seeds. Silva Fennica, 34: 71-77.
Direct Link - Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem., 193: 265-275.
CrossRefPubMedDirect Link - Lichtenthaler, H.K., 1987. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol., 148: 350-382.
CrossRefDirect Link - Lurie, S., R.Z. Lipsker and B. Aloni, 1994. Effects of paclobutrazol and chilling temperatures on lipids, antioxidants and ATPase activity of plasma membrane isolated from green bell pepper fruits. Physiol. Plant, 91: 593-598.
Direct Link - Mackay, C., J. Hall, G. Hofstra and R. Fletcher, 1990. Uniconazole-induced changes in abscisic acid, total amino acids and proline in Phaseolus vulgaris. Pesticide Biochem. Physiol., 37: 74-82.
Direct Link - McKersie, B.D., J. Murnaghan, K.S. Jones and S.R. Bowle, 2000. Iron-superoxide dismutase expression in transgenic alfalfa increases winter survival without a detectable increase in photosynthetic oxidative stress tolerance. Plant Physiol., 122: 1427-1437.
Direct Link - Nakano, A. and K. Asada, 1987. Purification of ascorbate peroxidase in spinach chloroplasts; its inactivation in ascorbate-depleted medium and reactivation by monodehydroascorbate radical. Plant Cell Physiol., 28: 131-140.
CrossRefDirect Link - Nanjo, T., M. Kobayashi, Y. Yoshida, Y. Kakubari, K. Yamaguchi-Shinozaki and K. Shinozaki, 1999. Antisense suppression of proline degradation improves tolerance to freezing and salinity in Arabidopsis thaliana. Febs Lett., 461: 205-210.
Direct Link - Nie, G.Y. and N.R. Baker, 1991. Modification to thylakoid composition during development of maize leaves at low growth temperatures. Plant Physiol., 5: 184-191.
Direct Link - Nie, G.Y., E.J. Robertson, M.J. Fryer, R.M. Leech and N.R. Baker, 1995. Response of photosynthetic apparatus in maize leaves grown at low temperature to transfer to normal growth temperature. Plant Cell Environ., 18: 1-12.
Direct Link - Oliveira, G., P. Alves and A. Magalhaes, 2002. The effect of chilling on the photosynthetic activity in coffee (Coffea arabica L.) seedlings. The protective action of chloroplastid pigments. Braz. J. Plant Physiol., 14: 95-104.
Direct Link - Oquist, G., 1983. Effects of low temperature on photosynthesis. Plant Cell Environ., 6: 281-300.
CrossRefDirect Link - Payton, P., R. Webb, D. Kornyeyev, R. Allen and A. Holaday, 2001. Protecting cotton photosynthesis during moderate chilling at high light intensity by increasing chloroplastic antioxidant enzyme activity. J. Exp. Bot., 52: 2345-2354.
Direct Link - Pinhero, R.G. and R.A. Fletcher, 1994. Paclobutrazol and ancymidol protect corn seedlings from high and low temperature stresses. Plant Growth Regul., 15: 47-53.
CrossRefDirect Link - Prasad, K., D. Marc and R. Stewart, 1994. Acclimation, hydrogen peroxide and abscisic acid protect mitochondria against lrreversible chilling lnjury in maize seedlings. Plant Physiol., 105: 619-627.
Direct Link - Rao, M.V., G. Paliyath and D.P. Ormrod, 1996. Ultraviolet-B- and ozone-induced biochemical changes in antioxidant enzymes of Arabidopsis thaliana. Plant Physiol., 110: 125-136.
CrossRefDirect Link - Robertson, E.J., N.R. Baker and R.M. Leech, 1993. Chloroplast thylakoid protein changes induced by low growth temperature in maize revealed by immunocytology. Plant Cell Environ., 16: 809-818.
Direct Link - Rudolph, A.S., J.H. Crowe and L.M. Crowe, 1986. Effects of three stabilizing agents-proline, betaine and trehalose-on membrane phospholipids. Arch. Biochem. Biophys., 245: 134-143.
PubMedDirect Link - Saradhi, P.P., S. AliaArora and K.V.S.K. Prasad, 1995. Proline accumulates in plants exposed to uv radiation and protects them against uv-induced peroxidation. Biochem. Biophys. Res. Commun., 209: 1-5.
CrossRefDirect Link - Saropulos, A.S. and D.S.H. Drennan, 2002. Chlorophyll photooxidation and changes in the antioxidant protective enzymes in leaves of Capsicum annuum and Cucumis sativus seedlings subjected to chilling at high light intensity. Acta Hortic., 579: 239-244.
Direct Link - Scandalios, J.G., 1993. Oxygen stress and superoxide dismutases. Plant Physiol., 101: 7-12.
CrossRefPubMedDirect Link - Senaratna, T., B.D. McKersie and A. Borochov, 1987. Desiccation and free radical mediated changes in plant membranes. J. Exp. Bot., 38: 2005-2014.
CrossRefDirect Link - Senaratna, T., C.E. Mackay, B.D. McKersie and R. Fletcher, 1988. Uniconazole-induced chilling tolerance in tomato and its relationship to antioxidant content. J. Plant Physiol., 133: 56-61.
CrossRefDirect Link - Smith, D., G.M. Poulsen and C.A. Roguse, 1964. Extraction of total available carbohydrates from grass and legume tissues. Plant Physiol., 39: 960-962.
Direct Link - Stamp, P., 1987. Photosynthetic traits of maize genotypes at constant and fluctuating temperature. Plant Physiol. Biochem., 25: 729-733.
Direct Link - Sun, W.Q. and A.C. Leopold, 1995. The Millard reaction and oxidative stress during aging of soybean seeds. Physiol. Plant., 94: 94-104.
Direct Link - Sutton, F., X. Ding and, D.G. Kenefic, 1992. Group 3 LEA gene HVA1 regulation by cold acclimation and deacclimation in two barley cultivars with varying freeze resistance. Plant Physiol., 99: 338-340.
Direct Link - Kocsy, G., M. Brunner, A. Ruegsegger, P. Stamp and C. Brunold, 1996. Glutathione synthesis in maize genotypes with different sensitivities to chilling. Planta, 198: 365-370.
Direct Link - Upadhyaya, A., T.D. Davis, R.H. Walser, A.B. Galbraith and N. Sankhla, 1989. Uniconazole-induced alleviation of low-temperature damage in relation to antioxidant activity. Hortic. Sci., 24: 955-957.
Direct Link - Wise, R.R. and A.W. Naylor, 1987. Chilling-enhanced photooxidation. The peroxidative destruction of lipids during chilling injury to photosynthesis and ultrastructure. Plant Physiol., 83: 272-277.
PubMedDirect Link - Wolff, S.P., 1994. Ferrous ion oxidation in presence of ferric ion indicator xylenol orange for measurement of hydroperoxides. Methods Enzymol., 233: 182-189.
CrossRefDirect Link - Xin, Z. and P. Li, 1993. Relationship between proline and abscisic acid in the induction of chilling tolerance in maize suspension-cultured cells. Plant Physiol., 103: 607-613.
Direct Link - Yamazaki, H., T. Nishijima and M. Koshioka, 1995. Effects of (+)-S-abscisic acid on the quality of stored cucumber and tomato seedlings. Hortic. Sci., 30: 80-82.
Direct Link - Yong, K., S. Shin, R. Nilda., T. Hwang, O. Han, B. Cho, S. Jung and O. Guh, 2003. Antioxidative enzymes offer protection from chilling damage in rice plants. Crop Sci., 43: 2109-2117.
Direct Link - Yoshiba, Y., T. Kiyosue, K. Nakashima, K. Yamaguchi-Shinozaki and K. Shinozaki, 1997. Regulation of levels of proline as an osmolyte in plants under water stress. Plant Cell Physiol., 38: 1095-1102.
Direct Link - Zhang, L.X., P.H. Li and M.L. Brenner, 1986. Relationship between mefluidide treatment and abscisic acid metabolism in chilled corn leaves. Plant Physiol., 81: 699-701.
Direct Link - Zhou, W. and M. Leul, 1998. Uniconazole-induced alleviation of freezing injury in relation to changes in hormonal balance. Enzyme activities and lipid peroxidation in winter rape. Plant Growth Regul., 26: 41-47.
Direct Link - Zhou, B.Y., Y.B. Li, J.Z. Chen, Z.L. Ji and Z.Q. Hu, 2002. Effects of low temperature stress and ABA on flower formation and endogenous hormone of litchi. Acta Hortic. Sinica, 29: 577-578.
Direct Link - Zhou, B., Z. Guo and Z. Liu, 2005. Effects of abscisic acid on antioxidant systems of Stylosanthes guianensis under chilling stress. Crop Sci., 45: 599-605.
Direct Link








