Research Article
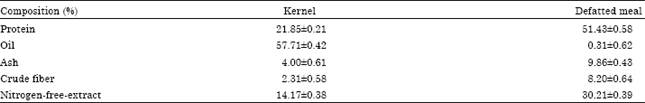
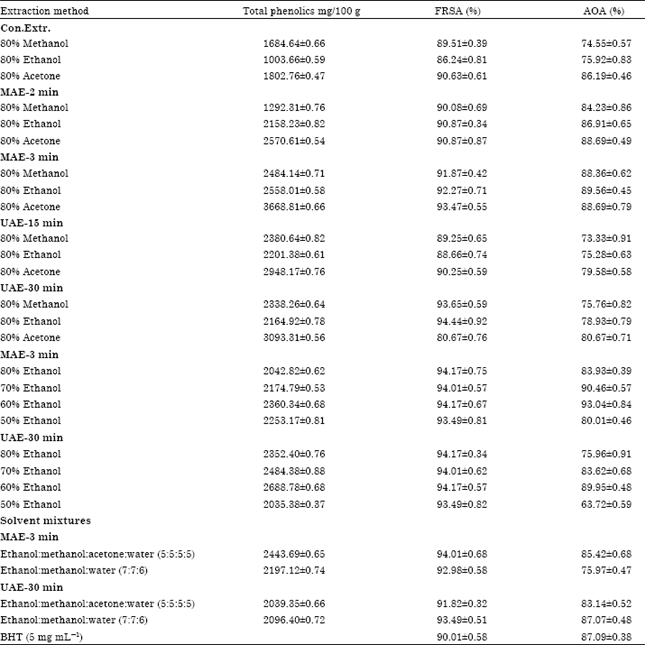
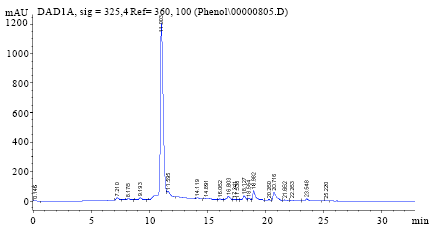
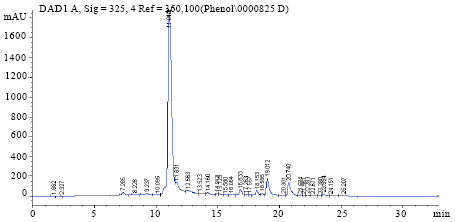
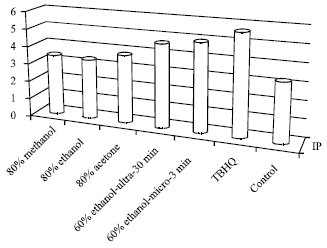
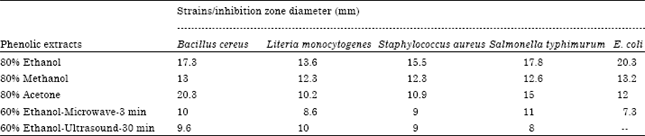
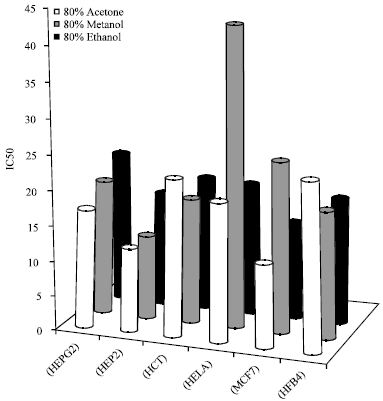
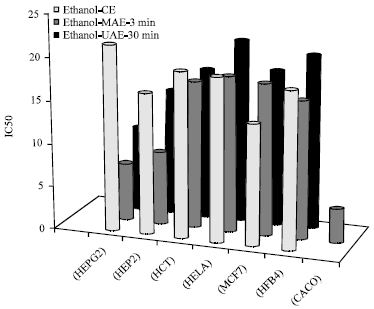
Optimization of the Extraction of Total Phenolic Compounds from Sunflower Meal and Evaluation of the Bioactivities of Chosen Extracts
Department of Fats and Oils, National Research Centre, Dokki, Cairo, Egypt
G.F. Mohamed
Department of Food Technology,National Research Centre, Dokki, Cairo, Egypt
S.H. Mohamed
Department of Dairy, National Research Centre, Dokki, Cairo, Egypt
S.S. Mohamed
Department of Fats and Oils, National Research Centre, Dokki, Cairo, Egypt
M.M. Kamil
Department of Food Technology,National Research Centre, Dokki, Cairo, Egypt