Research Article
Production of Glucoamylase Enzyme by a Rhizopus oryzae Strain
Department of Biology, Duba University College, University of Tabuk, Tabuk, Saudi Arabia
Glucoamylase (GA) is a hydrolyzing enzyme. It can degrade both amylose and amylopectin by hydrolyzing both α-1,4 and α-1, 6 glycosidic links of starch and produce glucose (Ono et al., 1964; Elegado and Fujio, 1993). Hence, glucoamylase can convert starch completely to glucose. Now a days, glucoamylase is one of the most important enzymes in food industries (Cock, 1982; Beuchat, 1987; Soccol et al., 1992) as, it is used for the production of glucose and fructose syrup from liquefied starch (Dale and Langlois, 1940; Nigam and Singh, 1995; Nguyen et al., 2002). It is also employed in baking, juice, beverage pharmaceuticals and many fermented foodstuffs industries for commercial production (Hesseltine, 1965) in some cases textile, leather and detergents industries (Whistler et al., 1984; Reed and Rhem, 1987). Due to its increasing demand, the production technique of glucoamylase and α-amylase has been studied in detail.
The enzyme was reported to produce by many fungi like Aspergillus awamori, A. saitoi, A. oryzae, Rhizopus spp., Mucor spp., Penicillium spp. and Yeast (Tsujisaka et al., 1958; Sen and Chakarabarty, 1984). Among these, Rhizopus spp. are considered good producers of amylolytic enzyme (Takahashi et al., 1994; Jin et al., 1999). On the basis of the importance of glucoamylase, the present study has been taken to optimize the glucoamylase production by Rhizopus spp. in liquid culture.
Isolation and identification of microorganisms: All general chemicals were of Analar grade or equivalent. Rhizopus spp. were isolated from different Sudanese, cereal grains, rotten fruits, food materials and were also obtained from previous collections. Cereal grains and pieces of rotten fruits were surface disinfected with 4% sodium hypochlorite for 2 min (King et al., 1986). The surface disinfected grains and pieces of rotten fruits and also the untreated bread were plated on sterilized glass petri dishes each with three layers of moistened filter papers. Then they were incubated at 25°C for 7 days.
Amylase activities of Rhizopus isolates producing the starch digesting amylase was detected on plates by the index of amylolytic activity according to the method described by Hankin and Anagnostakis (1975). Rhizopus colonies producing large clear zone were picked up and purified by streaking on PDA. Pure cultures were maintained in PDA media at 4°C and were sub cultured at 30 days interval. The Rhizopus isolates were characterized using the Manual of Taxonomical Studies on the Genus Rhizopus (Inui et al., 1965).
Screening of glucoamylase: Rhizopus isolates were selected for the production of the enzyme on the basis of producing larger clear zone. The growth medium contained 0.5% g w/v of rice flour, 100 mL of distilled water in 500 mL Erlenmeyer flasks and then was sterilized by autoclaving at 121°C and 15 lb/square inch for 15 min. A loop full of spores of the chosen Rhizopus isolates (from a 7 days old culture grown in PDA slant) was inoculated separately in the flask. These cultures propagated in a rotary incubator shaker (100 rpm) for 24 h at 30°C. 5 mL of these vegetative inocula were then inoculated in 250 mL flasks containing 45 mL of the growth medium. These cultures propagated in a rotary incubator shaker (100 rpm) for 24 h at different temperatures 30/35/40/45/50°C.
Samples were taken from cultures for each temperature at different time intervals 24, 48, 72 and 96 h. The samples were then centrifuged at 5000 rpm to remove mycelia. The supernatant fluids were collected and used for assaying of amylase activity.
Based on the result of this experiment, two strains were selected for subsequent studies.
Culture condition and glucoamylase production: Growth and incubation period was quantified along with the effect of carbon, nitrogen source, phosphorus salts on production of the enzyme. To determine the incubation period the liquid culture media with inoculum were incubated up to 4 days. For determining the effect of temperature on enzyme production, various temperatures ranging from 30-50°C were used. The pH values of initial cultures were adjusted to 4, 5, 6, 7 and 8 to find its optimum value. Ammonium sulphate (NH4)2SO4/ammonium chloride NH4Cl/sodium nitrate NaNO3/peptone/beef extract/yeast extract/urea, were used as the nitrogen sources to determine the better nitrogen source. Diammonium hydrogen phosphate (NH4)2HPO4/sodium dihydrogen phosphate NaH2PO4/disodium hydrogen phosphate Na2HPO4/potassium dihydrogen phosphate KH2PO4/dipotassium hydrogen phosphate K2HPO4 were used as phosphorus salts to determine the effect of different phosphorus salts. Crude enzymes were extracted and assayed after 24 h of inoculation starting from the 2nd day up to the 4th day of incubation. Each of the parameters described above was made separately with three replicates.
Enzyme assay: The enzyme assayed by the method of Bernfeld (1951) as described by Pestana and Castillo (1985) using starch as substrate. One unit of enzyme activities was expressed as 1 mg of glucose liberated per one milliliter. of culture supernatant at pH 7.5, 40°C and in 1 min. Extensive screening was carried out by measuring residual glucose and glucoamylase activity. The protein content was determined by the method of Bradford (1976) with bovine serum albumin as the protein standard. The specific activity of amylolytic enzyme was taken as units/mg protein. Among 6 isolates, 2 isolates showed relatively highest enzyme activity and were selected for subsequent studies.
Enzyme activity measurements: Amylase activity was determined by the method of Bernfeld (1951) as described by Pestana and Castillo (1985) as follows: The assay mixture consist of 1 mL of 1% soluble starch as substrate, 1 mL of 16 mM acetate buffer (pH 6.0) and 0.9 mL of distilled water. At zero time 100 μL of crude enzyme preparation was added and the mixture was shaken and incubated at 20°C in a water bath. After 30 min. the reaction was stopped by adding 2 mL of 3,5 DNS and the tube was heated for 5 min in boiling water. After cooling the absorbance was read spectrophotometrically at 546 nm against a blank consisting of 100 μL of buffer instead of crude enzyme preparation. The amount of reducing sugar was calculated from standard curve constructed by using glucose (Fig. 1a).
Thin layer chromatography: The products of starch hydrolysis by the crude enzyme, as described in the assay method, were identified by Thin Layer Chromatography (TLC) using n-butanol:ethanol:water in a ratio of 4:2.2:2 as a solvent and maltose and glucose as standard sugars. Sugars and oligosaccharides were detected as described by Trevelyan et al. (1950). The sugar spots appeared as dark brown spots. Identification of the sugars was done by comparing the Relative fraction (Rf) values of the samples with that of the standards.
Species isolated from different sources (crop seeds and bread) are screened for amylase in starch agar plate. Based on the index of amylolytic activity (Hankin and Anagnostakis, 1975) two Rhizopus isolates were chosen for further studies.
Assay of enzyme production was carried out at various temperature ranging from 25-50°C. Samples for the determination of the amylase activity were taken for each temperature at different time intervals of 24, 48, 72 and 96 h. Rhizopus oryzae (R2) has shown maximum amylolytic enzyme production at 30°C (specific activities were 4.25) retaining 22 of its enzyme activities at 35°C (Fig. 1a, b). Rhizopus oryzae (R3) showed maximum performance of enzyme production at 45°C with specific activities 2.5 U mg–1 protein, retaining 95 of its activities at 50°C.
In fact this result for Rhizopus oryzae (R3), which showed maximum amylolytic enzyme production 45°C could be of importance industrially as it is always cheaper not to cool the production. Accordingly, it is chosen for further studies.
Similar results for production of glucoamylase by Rhizopus spp. at 45°C was reported by Nahar et al. (2008). Previous reports depict that 35°C temperature shows maximum enzyme activity by Rhizopus delemar (Soccol et al., 1994) and Aspergillus niger (Feroza et al., 1998).
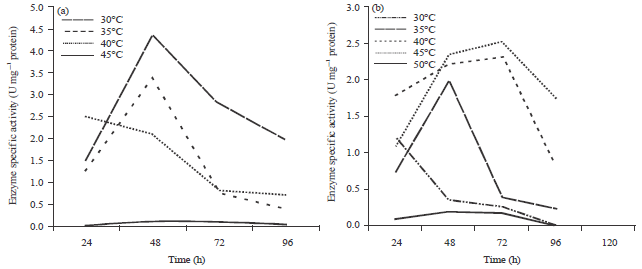 | |
| Fig. 1(a-b): | Effect of temperature and time intervals on amylase production by (a) Rhizopus oryzae (R2) and (b) Rhizopus oryzae (R3) |
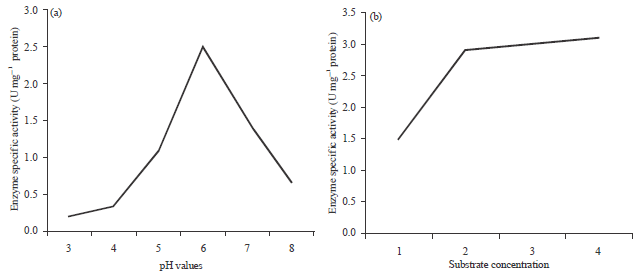 | |
| Fig. 2(a-b): | Effect of (a) pH and (b) Substrate concentration on amylase production by Rhizopus oryzae (R3) |
Increase in the incubation period resulted in a decrease in the production of amylase by culture of Aspergillus spp. It may be due to the fact that after maximum production of amylase enzyme (maximum incubation time), the production of other by products result in the depletion of nutrients. These byproducts inhibited the growth of fungi and hence enzyme formation (Feroza et al., 1998). Optimum pH for maximum enzyme production was found to be 6.0 (Fig. 2a). According to James and Lee (1997) the range of glucoamylase pH is between 3.7 and 7.4 (20) and reported that Rhizopus-RFF showed maximum enzyme activity at pH 4.5. Maximum enzyme production of enzyme occurred at pH 4-6, very little growth was observed without enzyme production in medium at initial pH 3-4 (Hankin and Anagnostakis, 1975).
It is clear that 2% rice flour concentration seems to be the concentration that gave maximum production of amylase (Fig. 2b). Rhizopus oryzae (R3) gave lower increase in production with specific activity 2.9 U mg–1 protein. This indicates that the enzyme is inducible. Ali et al. (1989) working on A. terreus; Pestana and Castillo (1985) working on A. awamori and Michelena and Castillo (1984) working on A. foetidus reached the same conclusion.
Nitrogen sources have a great effect for microbial growth and in the production of extra cellular enzymes by R3. The results of the different combinations of organic nitrogen source are presented in Fig. 3a. The maximum enzyme activity was found by using ammonium sulfate (R3). Sodium nitrate and ammonium chloride have shown slightly inhibitory effect towards the production of amylase enzyme by R. oryzae (R3). This agree with Ali et al. (1989), who reported that addition of ammonium sulfate, peptone, yeast extract or beef extract to the culture medium stimulated glucoamylase production by A. terreus, also with Goto et al. (1998), who reported an increase of glucoamylase production by A. niger when the basal culture medium was supplemented with ammonium sulphate. On the other hand, Lagzouli et al. (2007) reported that ammonium sulphate and ammonium chloride reduced the final yield of amylase enzyme produced by A. foetidus, while sodium nitrate stimulated the enzyme production. Previous study reported that the increase of glucoamylase production by A. awamori NRRL by addition of urea to the culture medium.
The isolate of Rhizopus oryzae (R3) was only affected by the monosodium hydrogen phosphate, which significantly increased the amylase production (Fig. 3b).
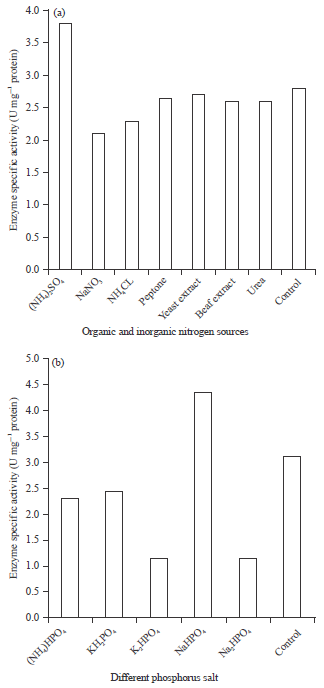 | |
| Fig. 3(a-b): | Effect of (a) Organic and inorganic nitrogen sources and (b) Different phosphorous salt on amylase production by Rhizopus oryzae (R3) |
Lagzouli et al. (2007) working on Aspergillus foetidus, Goto et al. (1998) working on A. niger and Ali et al. (1989) working on A. terreus reported the repression of glucoamylase synthesis by disodium and potassium phosphate salts while, monosodium and potassium phosphate increased its production by these fungi. However, Ramachandran et al. (1979) reported that phosphorus salts did not affect glucoamylase production by A. awamori.
Thin Layer Chromatography (TLC): Products of starch hydrolysis by the enzymes from the Rhizopus oryzae (R3) and as identified by thin layer chromatography is seen to be only glucose by comparison with the authentic markers. Accordingly, it is concluded the amylase is glucoamylase. These results are similar to those published by Ali et al. (1989) and Ramachandran et al. (1979) using A. terreus, A. awamori and A. flavus var. columnaris, respectively.
The results of this study show that highest glucoamylase activity was found in shake flasks provided by a medium containing 2% rice flour concentration, at temperature 45°C and initial pH value of 6.0. Rice flour suspension can be utilized for growth Rhizopus oryzae (R3) which is used in this work and production of enzyme, although supplementation with nitrogen sources and phosphorus salts is needed in order to increase enzyme level.